Published online May 10, 2015. doi: 10.5317/wjog.v4.i2.24
Peer-review started: December 21, 2014
First decision: January 20, 2015
Revised: January 31, 2015
Accepted: April 10, 2015
Article in press: April 14, 2015
Published online: May 10, 2015
Processing time: 130 Days and 12.6 Hours
The ongoing research on pioneering drug candidates for the overactive bladder (OAB) aimed to overcome the limitations of currently licensed pharmacotherapies, such as antimuscarinics, β3-adrenergic agents, and botulinum neurotoxin, has been reviewed performing a systematic literature review and web search. The review covers the exploratory agents alternative to available medications for OAB and that may ultimately prove to be therapeutically useful in the future management of OAB patients based on preclinical and early clinical data. It emerges that many alternative pharmacological strategies have been discovered or are under investigation in disease-oriented studies. Several potential therapeutics are known for years but still find obstacles to pass the clinical stages of development, while other completely novel compounds, targeting new pharmacological targets, have been recently discovered and show potential to translate into clinical therapeutic agents for idiopathic and neurogenic OAB syndrome. The global scenario of investigational drugs for OAB gives promise for the development of innovative therapeutics that may ultimately prove effective as first, combined or second-line treatments within a realistic timescale of ten years.
Core tip: The forefront of global research scenario of investigational drug candidates for the management of patients with overactive bladder and detrusor overactivity was reviewed. Among a huge amount of exploratory compounds with completely new mechanisms of action, some promising pharmacological principles show potential to translate into novel therapeutics to be clinically used as first-line alternative treatments, or in combination with established drugs, or as second-line treatments in refractory patients.
- Citation: Sacco E, Recupero S, Bientinesi R, Palermo G, D’Agostino D, Currò D, Bassi P. Pioneering drugs for overactive bladder and detrusor overactivity: Ongoing research and future directions. World J Obstet Gynecol 2015; 4(2): 24-39
- URL: https://www.wjgnet.com/2218-6220/full/v4/i2/24.htm
- DOI: https://dx.doi.org/10.5317/wjog.v4.i2.24
Overactive bladder (OAB), defined as urinary urgency with or without urgency urinary incontinence, usually associated with increased urinary frequency and nocturia[1], is a very bothersome and debilitating chronic condition that severely affects the patient's quality of life[2]. The socioeconomic burden is very high due to the aging population, the OAB-associated comorbidities and the increased risk of hospitalization[3]. The pathophysiology is largely unknown, although multiple causes have been proposed, such as a primary detrusor dysfunction, observed as detrusor overactivity (DO) during urodynamic studies, an overactivity of the afferent arm of the micturition reflex, a primary dysfunction of higher central nervous system (CNS) inhibitory centers[4]. OAB is underdiagnosed and undertreated, however, the increase in patient awareness, the rise in the geriatric population, and the availability of more pharmacological principles have triggered a significant growth in the OAB market with a estimated market size of over $2 billion in 2012.
Pharmacological treatment has been based for years on antimuscarinic agents, but recently other two pharmacological principles have been approved for OAB by the United States Food and Drug Administration (FDA): the first β3-adrenergic agent, mirabegron (Myrbetriq®, Astellas, approved in June 2012), and the botulinum neurotoxin (Botox®, Allergan, approved in January 2013)[5,6]. Although many novel antimuscarinic and β3-adrenergic agents, and alternatives to the botulinum neurotoxin are under development, the ideal medication for the cure of OAB with an optimal profile in terms of safety, tolerability and efficacy is still to be discovered. A huge amount of preclinical studies is ongoing exploring the therapeutic potential of many novel compounds some of which already advanced to the clinical phases of development, which mixed results[7,8].
This review provides an extensive update on the exploratory drugs, alternative to available medications for OAB that may ultimately prove to be therapeutically useful in the future treatment of lower urinary tract symptoms (LUTS) and OAB.
A systematic literature review search of peer-reviewed English-language full papers published by November 2014 has been performed. Medline databank was searched employing both “MeSH” and “free text” protocols, and using a combination of the following search terms: “urinary bladder, overactive”, “urinary incontinence, urge”, ”lower urinary tract symptoms” AND “drug therapy”. Scopus and ISI Web of Science databanks were also searched using the same search terms. A search of articles related to each specific compound was also performed. A hand search of reference lists of retrieved articles was performed in order to identify further studies not captured by the above used terms. Clinical trials and pharmaceutical companies’ websites were also searched for pipeline projects. All the investigational pharmacological principles with at least preclinical evidence of activity against OAB/DO have been discussed.
Compounds with agonist activity on γ-aminobutyric acid (GABA) receptors in the CNS exploit the inhibitory effect of this neurotransmitter on micturition reflex[9]. Baclofen, a GABAB-receptors agonist, is used for the treatment of neurological spasticity, particularly in the lower limb. Baclofen is pumped directly into the subarachnoid space by means of a programmable pump via a catheter system. Preclinical studies showed that intrathecal baclofen was effective in attenuating oxyhemoglobin-induced DO[10] and, in rats with spinal cord injury (SCI), produced a dose-dependent inhibition of non-voiding bladder contractions and a decrease in micturition pressure[11]. Clinical studies based on urodynamic evaluations showed that the continuous intrathecal baclofen pump infusion is effective in the management of patients with medically-refractory neurogenic DO (NDO) and decreased bladder compliance[12]. Although baclofen gained approval for treatment of NDO in SCI patients, the narrow therapeutic window and the tolerability profile limited its widespread clinical use. ADX71441, a novel, potent and selective agonist of GABAB-receptor, showed efficacy in rodent models of OAB and may allow further exploitation of this central inhibitory mechanism[13].
The inhibition of the GABA transporters (GAT) that are thought to provide a GABA inactivation mechanism in the CNS, has been explored as a possible pharmacological principle aimed to treat DO. Tiagabine, an anticonvulsants that selectively inhibits the GABA re-uptake via the GABA transporter GAT1, given intravenous or intrathecal in rats, improved storage phase parameters suggesting a potential utility for OAB treatment[14].
Gabapentin is a putative GABA analog crossing the blood-brain barrier originally developed to treat epilepsy, and currently used for neuropathic pain and other conditions. Its mechanism of action has not been fully elucidated although it appears to have inhibitory activity on afferent C-fibers likely by binding to α-2-delta subunit of voltage-dependent calcium channel[15]. Carbone et al[16] reported in a pilot study that gabapentin improved both symptoms and urodynamic parameters in NDO patients. Kim et al[17] reported that the drug was well tolerated and improved symptoms in 14 out of 31 antimuscarinics-refractory OAB patients. Recently, beneficial clinical effects of gabapentin as an add-on therapy have been reported also in 16 out of 30 children with antimuscarinics-refractory OAB[18]. Phase II trials are ongoing comparing gabapentin with solifenacin in OAB patients[19] and evaluating the efficacy and tolerability of a combination of low doses of gabapentin and oxybutynin[20].
Pregabalin (Lyrica®, Pfizer) is an anticonvulsant drug mainly used for neuropathic pain and fibromyalgia. Like gabapentin, pregabalin binds to the α-2-delta subunit of the voltage-dependent calcium channel in the CNS, leading to decreased release of several neurotransmitters[21]. A phase II trial is ongoing in patients with idiopathic OAB comparing pregabalin with tolterodine and their combination[22]. Furthermore, preclinical data showed significant improvement on the urodynamic parameters of an animal model of NDO providing a rationale for future proof-of-concept clinical trial on NDO patients[23].
Levetiracetam is an antiepileptic drug with a mechanism of action not yet clarified, although the drug binds to the synaptic vesicle glycoprotein SV2A and inhibits presynaptic calcium channels reducing neurotransmitter release and acting as a neuromodulator[24]. Experimental findings in spinal cord transected rats have shown that levetiracetam, administered by subcutaneous osmotic minipump, improved urodynamic parameters in this animal model of NDO[25].
An alternative strategy is based on increasing GABA in the spinal cord via viral-mediated gene delivery. Injection in SCI rats of HSV-GAD (replication-defective herpes simplex virus vectors encoding genes of glutamic acid decarboxylase, the GABA synthesis enzyme) significantly decreased the number and amplitude of non-voiding contractions compared with controls, without blunting micturition pressure likely via the inhibition of the afferent limb of the micturition reflex[26]. Thus, GAD gene therapy gives promise to become a novel therapy of urinary dysfunctions in SCI patients.
Glycine is a major inhibitory neurotransmitter in the spinal cord. Animal studies suggest that glycinergic neurons have an important inhibitory effect on the spinobulbospinal micturition reflexes at the level of the lumbosacral cord[9].
The extracellular concentration of glycine at synapses is regulated by two types of glycine transporters (GlyTs): GlyT1 and GlyT2[27]. In rats, GlyT2 plays a major role in the clearance of extracellular glycine in the spinal cord and its inhibition leads to amelioration of cyclophosphamide-induced DO and pain behavior[28]. As a result, activation of glycinergic inhibitory mechanisms by GlyT2 inhibitors has been suggested as a novel therapeutic strategy for OAB and bladder pain syndrome.
Inhibitory effects on micturition are known side-effects of drugs with inhibitory action on the monoamine reuptake, including tricyclic antidepressants. Furthermore, depression is more common in patients with OAB and a shared deficiency of monoamine (serotonin and noradrenaline) behind both depression and OAB has been suggested[29].
Imipramine, a tricyclic antidepressants, improves storage LUTS and DO at the cost of not negligible side-effects. Antidepressants selective serotonin reuptake inhibitors, such as escitalopram, are under evaluation for efficacy in OAB patients[30].
Duloxetine, an antidepressant acting as a selective serotonin-norepinephrine reuptake inhibitor (SNRI) and approved for the treatment of stress urinary incontinence for its stimulatory activity on external urethral sphincter, demonstrated significant efficacy compared to placebo in relieving urinary symptoms in women with OAB[31]. However, the side-effects of this compound significantly limit patient’s compliance.
Based on animal experiments showing that besipirdine, a SNRI that interacts also with α1 (agonist) and α2 (antagonist) receptors, significantly and dose-dependently improves storage function and external urethral sphincter activity[32], a human proof-of-concept study has been initiated by UroGene in patients with storage LUTS[33].
Increasing evidence indicates that serotonin [5-hydroxytryptamine (5-HT)] is involved in a complex way in the control of micturition at central and peripheral sites, with both inhibitory and facilitatory effects[34-41], although the serotonergic pathway generally enhances urine storage by facilitating the vesical sympathetic reflex pathway and inhibiting the parasympathetic voiding pathway.
The 5-HT1A receptor agonist 8-OH-DPAT has been investigated in alpha-chloralose anesthetized or conscious chronic SCI cats[37]. This compound significantly increased the bladder volume threshold for eliciting a large amplitude micturition contraction, but only slightly reduced the amplitude of the contractions, indicating that drugs that activate 5-HT1A receptors might be useful in treating NDO after SCI. 8-OH-DPAT also improved voiding efficiency and maximum intravesical pressure, and enhanced the external urethral sphincter tonic and bursting activity in a rat model of incomplete cauda equina/conus medullaris injury[38].
5-HT2 and 5-HT3 receptors mediate excitatory effects on sympathetic and somatic reflexes to increase outlet resistance, and preclinical studies have shown that 5-HT2C and 5-HT3 receptors play an inhibitory role on micturition reflex suggesting that agonists at this site may have potential as candidate drugs for OAB[36].
The peripheral excitatory function of serotonin is increased in disorders known to be associated with DO, such as bladder outlet obstruction (BOO) and diabetes[39]. The facilitatory action on micturition reflex of the 5-HT2A receptor has been demonstrated in rats and its overexpression observed in BOO rat bladder[36]. Accordingly, sarpogrelate (a 5-HT2A selective antagonist) counteracted in diabetic rat bladder the increased contractile response to 5-HT in a dose-dependent manner[39]. Accordingly, Takimoto et al[40] reported a symptomatic benefit in patients with diabetes and refractory OAB treated with sarpogrelate.
5-HT4 and 5-HT1A receptors have been also involved in micturition control and their selective antagonists such as piboserod and WAY100635, respectively, potently inhibited the micturition reflex in animal models and human detrusor[34,41]. However, disappointing results have been reported with Rec-0545, a potent and selective antagonist of the 5-HT1A receptor evaluated by Recordati in a proof-of-concept trial for the treatment of OAB patients[42]. The combination of WAY100635 with duloxetine has been evaluated in a cat model of DO with promising results[43].
5-HT3 receptor is another candidate target for the development of novel drugs for the OAB according to recent preclinical findings[44]. Dynogen Pharmaceuticals, Inc. is developing a drug (DDP225) with both 5-HT3 receptor-antagonist and noradrenaline reuptake inhibitor properties for the treatment of OAB in patients who are not incontinent[45].
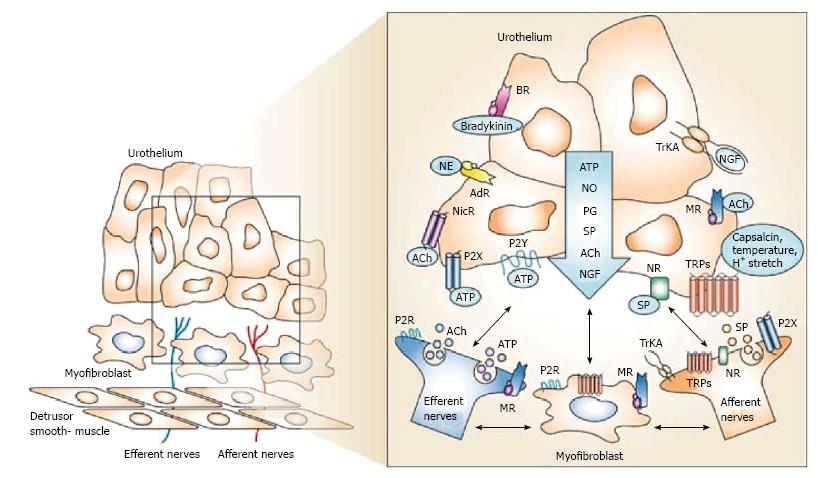
Several pharmacological approaches have been driven for a more in depth understanding of the physiology of the “mucosal bladder network” (the functional unit consisting of the urothelium, interstitial cells and afferent nerves) (Figure 1). An interesting hypothesis-driven approach for the future treatment of OAB is represented by the antagonism of purinergic receptors, namely P2X1 and P2X3/P2X2/3[46]. Thus, several studies suggested that the adenosine 5’-triphosphate (ATP) and purinergic ionotropic (P2X) receptors are involved in DO[47-49]. This is not surprising taking into account that purinergic transmission has been found on both afferent and efferent signalling pathways within the lower urinary tract and appears to be abnormally enhanced with aging[50] and DO[51]. In particular, P2X3 receptors on sensory nerve terminals are involved in voiding dysfunctions of conscious chronic SCI rats, raising the possibility that P2X3 receptor antagonists might be useful for the treatment of NDO[46]. In human bladders with DO an increase in P2X3 receptor expression was observed[52].
The growing appreciation for the role of purinergic receptors in mediating nociceptive neurotransmission prompted the development of P2X receptor-selective antagonists as potential therapeutics for pain management[53]. The novel P2X3/P2X2/3 receptor antagonists possess attributes that offer the potential for optimization into candidate drug molecules not only for inflammatory and painful bladder conditions but also for OAB, in particular the recently developed RO3 compound (Roche Palo Alto) and the AF-742 (Afferent Pharmaceuticals), which is ongoing a phase II trial for bladder pain syndrome[54]. Finally, P2X3 antisense oligonucleotides and RNA interference-mediated treatment, that appear to be promising for neuropathic pain management, might open up new avenues for therapeutic OAB strategies[55].
Substance P (SP) and neurokinin A (NKA) are neuropeptides with the highest affinity for NK1 and NK2 receptors, respectively. NK-receptors have been demonstrated in CNS regions involved in micturition control[56]. Many experimental observations are available indicating that spinal and supraspinal NK1 and NK2 receptors may modulate the micturition reflex[57-59].
Tachykinins are also released from urothelial/suburothelial capsaicin-sensitive afferents and are able to stimulate muscle tone and bladder contractions (NKA > NKB > SP), and to influence vascular tone and permeability (“neurogenic inflammation”)[60,61]. Intravenous NK1 and NK2 receptor selective antagonists reduced DO in rat with SCI[61,62]. Perfusion of bladder with a NK1 receptor antagonist improved DO in rats wit cyclophosphamide-induced cystitis[48].
Mixed clinical results have been reported on some compound in this class. Aprepitant (Merck Sharp and Dohme Corp.) is a CNS-penetrating NK-1 antagonist used to treat chemotherapy-induced nausea. A pilot, proof-of-concept randomized controlled trial (RCT) including 125 post-menopausal women with urge or mixed (urge-predominant) incontinence reported satisfactory tolerability and efficacy of aprepitant over placebo in ameliorating OAB symptoms[63]. Serlopitant (MK0594, Merck Sharp and Dohme Corp.) has been evaluated in a RCT and, although it significantly decreased the primary endpoint of daily micturitions, no advantages in efficacy have been found vs tolterodine[64]. Netupitant (by Helsinn Healthcare) is another potent and selective NK1 receptor antagonist that failed to demonstrate superiority over placebo in a phase II trial[65].
Several “Transient receptor potential” (TRP) neuroreceptors have been involved in nociception and mechanosensory transduction in various organ systems as well as in storage bladder function and DO, offering the possibility to target bladder dysfunctions at the level of sensory signal initiation (Figure 1)[66].
“Transient receptor potential vanilloids 1” (TRPV1) is the principal transduction channel for nociception. TRPV1 is also found in myelinated Aδ-fibres and sensory unmyelinated C-fibres located in the pelvic nerve afferents and in a sub/intraurothelial plexus; it is sensible to bladder filling, bladder contractions and noxious stimuli[67]. TRPV1 is expressed also by the urothelial cells themselves[67].
C-fibres are normally silent but have been found to become active and to convey signal to the spinal cord in pathological situation such as OAB, NDO and SCI, resulting in the bothersome sensation of urinary urgency[68].
“Vanilloids” such as capsaicin are the best-known natural TRPV1 agonists and several trials showed that, given intravesically, they could cause a sustained activation of the TRPV1 receptor resulting in a desensitization of C-fibers with beneficial effects in patients with neurogenic or idiopathic DO, but at the cost of nonnegligible side-effects[69,70]. Resiniferatoxin is at least as effective as capsaicin, without the local side-effects although formal RCTs are needed to determine its appropriate use and dosage[71,72].
Potent orally-available small-molecule TRPV1 antagonists are undergoing clinical trials for chronic pain, but the lack of bladder-selectivity and potential effects on thermoregulation may be serious barriers for the clinical development[73,74]. XEN-D0501 (Provesica Ltd.) is a highly potent oral TRPV1 antagonist that was found to improve storage bladder function and reduce the intensity of capsaicin-induced bladder contractions in animal models; a phase I study reported a satisfactory tolerability and safety[75]. XEN-D0501 is currently being assessed for efficacy in OAB in an international phase II dose-ranging trial. JTS-653 (Japan Tobacco), MCP-101 (Mt. Cook Pharma) and SAF312 (Novartis Pharmaceuticals)[76], are other compounds in this class under investigation for the treatment of NDO.
Other TRP channels are expressed in the lower urinary tract (TRPV2, TRPV4, TRPM8, and TRPA1), and based on recent preclinical observations, TRPA1[77,78] and TRPV4[79-82] appear to have a critical role in bladder storage function and overactivity. Selective antagonists for these ion channels are already available making the superfamily of TRP channels a very interesting class of potential targets for drugs aimed to treat LUTS/OAB/DO.
μ-opioid receptor-agonists
μ-opioid receptor (MOR) agonists are known for decades for their analgesic efficacy and excellent tolerability. Tramadol, an effective and safe analgesic, is a weak MOR-agonist, but its metabolites have a stronger MOR-agonist effect and also inhibit the reuptake of noradrenaline and 5-HT and elicit effects by indirectly activating serotonergic and α2-adrenergic receptors[83]. Promising clinical results in OAB patients were published on tramadol by Safarinejad et al[84] but the study has been retracted due to statistical errors. Singh et al[85] evaluated urodynamic effects of epidural tramadol in 15 subjects reporting that it increased bladder capacity and compliance and delayed filling sensations without affecting voiding phase, even for those with BOO.
Tramadol-like compounds with less incidence of nausea might have a treatment potential in patients with NDO and the development of novel MOR-agonists is ongoing. KN203 (KeyNeurotek Pharma) is the first compound of this class to be developed against OAB, and the results of a proof-of-concept study are expected to clarify its role in this clinical setting[86].
δ-receptor agonists
A growing volume of information supports a role for the δ-receptor in the regulation of bladder activity[87]. In contrast to μ-agonists, δ-receptor agonists present with lower toxicity and no addiction, their most crucial safety aspect being the incidence of seizure-like convulsions in rodents. MCP-202 is a compound in this class and in the development pipeline of Mt Cook Pharma for the treatment of OAB. A novel nonpeptide, orally bioavailable δ-receptor agonist (DPI-221) with satisfactory safety profile and high potency in extending micturition interval in mice has been recently developed[88].
Nociceptin or orphanin FQ (N/OFQ) is the endogenous ligand of opioid-like receptor-4 (or NOP receptor)[89]. N/OFQ has a variety of effects both in the CNS and peripherally and there is evidence suggesting that N/OFQ inhibits the micturition reflex in rats by acting on the afferent bladder signalling and on supraspinal micturition sites[60,90], although a peripheral excitatory effect was also detected[90].
Lazzeri et al[91] reported that N/OFQ given intravesically was able to elicit an acute inhibitory effect on voiding reflex in 9 patients with NDO but not in 5 normal subjects. A RCT by the same authors including 14 NDO patients found that N/OFQ, but not placebo, increased significantly bladder capacity and reflex volume[92] and the results were replicated in a multicenter study[93]. Further investigations are required in order to establish if available selective NOP receptor agonists may become a new pharmacological way of treatment of NDO.
The Cannabis Sativa (marijuana) plant contains a group of biologically active substances, termed cannabinoids (CBs). The endocannabinoid system comprises the cannabinoid receptors (CB1 and CB2), their endogenous (“endocannabinoids”) and exogenous (‘‘exocannabinoids’’, such as plant-derived and synthetic cannabinoids) ligands, and related enzymes for biosynthesis and degradation, such as fatty acid amide hydrolase (FAAH)[94]. Recently, an orphan human G-protein coupled receptor, GPR55, was claimed to be a novel cannabinoid receptor[95].
These components have been located to animal and human lower urinary tract tissues (detrusor, bladder afferent nerves, and, particularly, urothelium) and have been involved in regulation of bladder function and bladder inflammation[94,96-100]. Intravesical, intrathecal and systemic administered CB-agonists are reported to inhibit bladder afferent signalling in animal models of bladder inflammation and improve urodynamics parameters in naive and DO animals models[98,101-103]. Plasticity of the endocannabinoid system in the spinal cord has been reported in rats with BOO-induced DO[104].
In patients with MS, cannabis extracts and delta-9-tetrahydrocannabinol (THC) were found to reduce OAB symptoms in open-label[105] and randomized trials[106], respectively. Nabiximols (Sativex, GW Pharmaceuticals), a standardazid mixture of compounds (mainly THC and cannabidiol) derived from Cannabis plants, failed to achieve primary endpoint (incontinence episodes) in a RCT including MS patients with OAB, however significantly improved other OAB symptoms (e.g., voids per 24 h, nocturia, and bladder symptom severity)[107].
Neurological side-effects of CB1-agonists, together with the unknown consequences of long-term use of such drugs, generated concern about their safety[108]. However, the intravesical administration of CB-agonists, the possible exploitation of CB2 (mainly peripheral) receptors and the inhibition of the FAAH by systemic, intravesical or intratecal-administered inhibitors may be alternative approaches to target the endocannabinoid system averting CNS side-effects[94,97,104,109,110].
Gonadotropin releasing hormone receptor (GnRH-R) antagonists have been reported to have beneficial effects on LUTS in patients with benign prostatic hyperplasia (BPH)[111], although they are considered still investigational in this setting, especially in light of the disappointing results of a phase III RCT on cetrorelix[112].
Treatment with subcutaneous degarelix (Ferring), a long-acting GnRH-R antagonist, improved experimental DO in rats and also promoted more efficient bladder emptying; isolated detrusor from degarelix-treated rats responded with larger carbachol-contractions than controls[113]. Another compound in this class, ganirelix, given systemically counteracted experimental DO in rats[114]. Interestingly, intravesical ganirelix and degarelix improved urodynamic parameters in rats[113,114]. Based on these results and since the GnRH-R is expressed in the rat bladder[113], a local intravesical administration of this class of drugs may be considered.
Phosphodiesterases (PDE) are enzymes that degrading cyclic nucleotides (cAMP and cGMP), can counteract the detrusor relaxation[115]. Among eleven PDE isoforms so far identified, PDE1-5 are described in the bladder and preclinical studies showed that PDE inhibitors (PDE-Is) are able to reverse the cholinergic-induced contraction of human detrusor and to enhance cAMP/cGMP-mediated detrusor relaxation[116]. Selective inhibitors of the different PDE types have been showed to can counteract DO[117].
Although a pilot study suggested a possible role for vinpocetine, a PDE1-inhibitor, in the treatment of refractory OAB[118], in a multicentre, placebo-controlled RCT in patients with DO, vinpocetine showed a statistically significant superiority over placebo for only one parameter[115].
Rolipram, a PDE4-I, has been showed to inhibit phasic myogenic contractile activity of human detrusor[119]. Other PDE4-I have been showed to reduced DO in rats with BOO, without affecting the voiding phase, suggesting that PDE4-Is might represent an alternative strategy for the treatment of the OAB[120,121].
A PDE9-I (ASP4901) is also under evaluation in a phase II trial by Astellas Pharma enrolling male patients with BPH[122].
Sildenafil, a PDE5-I, reversed the tonic cholinergic-induced contraction of human detrusor smooth muscle and produced relaxation via activation of cGMP- and cAMP-dependent pathways, K+ channels and the hydrogen sulfide [H(2)S] signaling pathway[123,124]. A series of RCTs provided substantive evidence of the efficacy and tolerability of PDE5-Is (sildenafil, tadalafil, vardenafil, and United Kingdom-369003) for the treatment of LUTS in male patients with or without erectyle dysfunction, confirmed by meta-analyses[125,126]. Tadalafil received the FDA approval in October 2011 for the treatment of males with LUTS secondary to BPH or concurrent LUTS and ED.
PDE-Is require further evaluations in order to better define their mechanism and site of action in lower urinary tract, their role and optimal dosage in different group of patients and in women, long-term safety and efficacy and cost-effectiveness.
Nitric oxide (NO) is a potent biological messenger that promotes detrusor relaxation, likely via the elevation of intracellular cGMP.
HCT-1026 (nitroflurbiprofen, by NicOx SA) is a NO-releasing derivative of the nonsteroidal anti-inflammatory drugs (NSAID) flurbiprofen[127]. Nitroflurbiprofen combines the anti-inflammatory activity of flurbiprofen with the smooth muscle relaxant activity of the NO moiety and promising preclinical (internal report of NicOx SA) and phase II clinical efficacy results have been announced in the treatment of NDO patients and women with OAB, providing a rationale for phase III trials[128,129].
Several lines of evidence suggest an important role of prostaglandins (PGs) in the modulation of the bladder function[130]. PGF2α, PGE1, and PGE2 slowly contract isolated animal and human detrusor and a role of PGs in the maintenance of detrusor tone and in the modulation of efferent and afferent neurotransmission has been suggested[131]. Release of PGE2, which acts via mainly EP receptors, is elevated in DO due to SCI[132] or to BOO[133,134]. The intravesical instillation of PGE2 also induces DO, urgency and decreases bladder capacity in humans[135].
PGs are locally synthesized in human bladder by constitutive (COX-1) and inducible (COX-2) cyclooxygenase[130]. Several factors including stretch, nerve stimulation, injury, exposure to ATP and other inflammatory mediators may induce the synthesis of PGs[131].
COX-inhibitors such as NSAID were found to be able to increase bladder capacity and prolong micturition interval without affecting voiding phase in rats, and favorable clinical effects have been reported in OAB patients treated with aspirin, indometacin, flurbiprofen, ketoprofen and loxoprofen[136,137]. Other preclinical findings have also indicated COX-2-selective inhibitors as potential drugs aimed to treat OAB, also by intravesical instillation[138,139]. It seems that NSAIDs might open a novel treatment opportunity for OAB although clinical evidence of efficacy of COX-inhibitors in OAB patients remains scarce and side-effects are important issues with these drugs[140].
The use of selective antagonists of PG receptor subtypes has been explored as a possible way to treat OAB. EP1 and EP2 PGs receptors have been demonstrated in the mucosal bladder network where they may trigger DO by eliciting bladder afferent activity during inflammation (possibly through TRPV1) and likely through the activation of interstitial cells[141,133]. There are data showing that EP3 receptors also participate in PGE2-induced DO[142].
Encouraging observations reported with novel EP1 antagonist compound (e.g., ONO-8539) in animal models[134] prompted their evaluation in a clinical proof-of-concept trial with disappointing results[143], thus reducing the likelihood of an oncoming introduction of EP1 receptor antagonists in the clinical management of OAB.
The Ras homologue family member A (RhoA) is a guanosine triphosphate hydrolase (GTPase) that, together with one of its downstream effectors, the type I and type II Rho-kinase (ROCK), have been shown to play an important role in calcium-independent pathway of smooth muscle contraction (the so-called “calcium-sensitization”) both in animal and human bladder[144-147]. The upregulation of RhoA pathway has been implicated in cystopathy associated to diabetes, BOO and DO[148].
Nonclinical in vitro studies showed that Y-27632 and HA-1077 (fasudil), ROCK1 and ROCK 2 inhibitors, respectively, significantly blocked carbachol-induced contractions and caused concentration-dependent relaxation of human detrusor strips[146]. It has been showed in pig urinary bladder tissues that this effect involved both urothelium-dependent and independent pathways[149].
Inhibition of Rho-kinase activity with Y-27632 produced a significant suppression of DO in spontaneously hypertensive rats (SHR) that also showed significantly higher RhoA protein expression in bladder tissues[150]. Treatment with oral fasudil partly but significantly ameliorated the development of DO in a rat model of BOO[151].
ROCK inhibitors may be a new pharmacological approach to treat OAB/DO if novel bladder-selective ROCK-inhibitors will be discovered in order to overcome the hypotensive side-effects of nonselective compounds.
Vitamin D3 receptor (VDR) is expressed in prostate and bladder tissues and BKL-628 (elocalcitol, BioXell), an agonist of vitamin D3-receptors, entered the pipeline for the therapy of BPH[152,153]. Elocalcitol is able to counteract the RhoA/ROCK pathway in the prostate and in both rat bladder strips and human bladder cells[154]. Elocalcitol appears to modulate bladder contractility by decreasing calcium sensitization and increasing L-type-mediated calcium entry[154,155]. The oral treatment with elocalcitol suppressed DO in two animal models of OAB and exerted strong suppressive effect on urinary bladder sensory signaling during filling in mice[156]. Encouraging results of a proof-of-concept clinical study prompted a phase IIb RCT including 308 OAB women[157]. The primary endpoint was not met but a favourable efficacy/tolerability profile and the statistically significant improvement of relevant secondary endpoints in the elocalcitol group vs placebo make this compound worthy of future reappraisal.
It has been suggested that selective inhibitors of nerve growth factor (NGF) may be a new way to treat OAB[158]. Several findings corroborate this hypothesis: urinary NGF levels decreased after successful treatment of OAB with antimuscarinics or BoNT[159,160]; NGF overexpression in the bladder and bladder afferent pathways has been reported to be involved in the emergence of hyperexcitability in bladder C-fiber sensory pathways[161]; the intrathecal administration of NGF antibodies decreased NGF levels in bladder afferent pathways and normalized bladder/urethral function in SCI rats[162].
There is nonclinical evidence that the local instillation of antisense oligonucleotides against the NGF, suppresses DO and the expression of NGF and chemokines[163]. In particular, the intravesical liposome-delivered antisense NGF-suppressing therapy could be an attractive approach for OAB, avoiding the toxicity of systemic nonspecific blockade[163].
Potassium channel opening drugs (KCOs) cause hyperpolarization and reduction in intracellular calcium concentration, promoting detrusor relaxation[164,165]. Furthermore, these agents may inhibit overactive bladder afferent pathways or influence the release of various urothelial mediators[166].
Many types of potassium channels have been demonstrated in the detrusor smooth muscle[167]: (1) big, intermediate and small calcium-activated channels (BK or maxi-K, IK and SK, respectively); (2) voltage-dependent (KV) channels; (3) inward-rectifying ATP-dependent channels (KATP, also known as Kir6 channels, a subtype of the Kir channels family); and (4) two-pore-domain (K2P) channels (also known as “leak potassium channels”).
BK channels (also called Maxi-K or slo1) have been extensively studied in animal and human detrusor smooth muscle and are arguably the most important physiologically relevant potassium channels regulating detrusor muscle cells action potential, resting membrane potential, and contractility[168-170]. Convincing data suggest that BK channels are also involved in mediating the relaxing effects of β3-ARs stimulation[171]. An important role of BK channels has been also advocated in etiopathogenesis of DO based on experimental in vitro observations in animal and human bladder tissues[170-173].
The role of KV channels in normal and pathological detrusor activity remains controversial and largely unexplored[174]. A reduction in potassium currents through KV channels has been involved in the hyperexcitability of the afferent neurons[175,176]. The structural diversity and the variety of the KV channels may allow for the identification of bladder-specific channels paving the way for the development of bladder-selective agent and genetic therapies for OAB[167,177].
Promising preclinical data from both in vitro and in vivo studies have been published supporting the possibility to restore normal detrusor function with openers of BK channel[168,178] and A-type KV channel[179,180]. Although their role is still debated, interesting preclinical observations are also available on openers of KATP channel[181-184], SK channel[185,186], combined SK/IK channel[187] and putative TREK-1 K2P channel[188].
Unfortunately, clinical trials with some of these drugs (e.g., ELB245 and ZD0947) were disappointing because of failure to demonstrate superiority vs placebo for the treatment of OAB[189], or because of side-effects leading to early termination[190]. Nevertheless, there is an ongoing effort to develop new classes of more potent and selective KCOs that may lead to the development of bladder-selective agent in the future.
Tetrodotoxin-resistant sodium channels (NaV1.8 subtype) are expressed in primary afferent capsaicin-responsive neurons innervating the bladder and their blockade by antisense oligodeoxy-nucleotide reduced the frequent voiding evoked by acetic acid-induced irritation of the bladder[191]. Ralfinamide (NW-1029) is a sodium channel blocker that suppresses tetrodotoxin-resistant sodium currents in C-type dorsal root ganglia neurons[192]. Via selective inhibition of capsaicin-responsive nociceptive neurons expressing tetrodotoxin-resistant sodium channels, ralfinamide is thought to elicit anti-nociceptive effects in animal models of inflammatory and neuropathic pain, as well as beneficial effects in DO[193].
Mechanosensitive ion channels, such as degenerin family/epithelial, amiloride-sensitive, sodium channel (ENaC) and TRP channel superfamily, have been recently demonstrated to play key roles in the mechanosensory signalling of the urinary bladder[194]. Acid-sensing (voltage-insensitive) cation channels (ASIC) are a subgroup of neuronal ENaC channels highly expressed also in the urothelium and suburothelial nerve plexus[195]. An increase in intrabladder pressure or upregulation of these channels may trigger afferent signalling during bladder filling[196]. ASIC channels seem involved in nociception in various pathological conditions including human bladder inflammation[197,198]. Consequently, ENaC/ASIC ion channels may become novel targets for the pharmacological treatment of inflammatory and overactive bladder conditions[194].
The complex neurophysiological control of the micturition reflex at both central and peripheral level, and the emerging recognition of the role of the different cell types involved in bladder physiopathology, prompted the development of many lines of research mostly aimed to the discovery of new pharmacological principles using receptor ligands as starting point. However, it appears that very few candidate agents, discovered starting from ligands-like compounds have passed the proof-of-concept stage in patient-oriented studies.
The pharmacological manipulation of central micturition circuitry is supported by the growing evidence on the central origin of OAB, although side-effects limit the use of currently available neuropharmacological agents and clinical results with selective antiserotoninergic are disappointing. It emerges a growing appreciation at preclinical level for the role of purinergic receptors as new targets for the treatment of OAB. Although the first clinical data are disappointing, NK-1 antagonists have attracted the interest of several companies and proof-of-concept studies are ongoing evaluating other compounds in this pharmacological class. Proof-of-concepts data are awaited also on novel opioids receptors agonists. Based on the recent evidence on the key role of the mucosal bladder network in the regulation of bladder function, many novel pharmacological principles targeting urothelium and afferent nerve fibers are under development. Although unsatisfactory clinical results have been reported with compounds based on this strategy (PG receptor antagonists, KCOs, elocalcitol), many other investigational agents show promise such as TRPV1-antagonists, modulators of endocannabinoid system, COX-2 inhibitors, ENaC/ASIC ion channels modulators. Intravesical strategies using N/OFQ, GnRH-R antagonist and liposome-delivered targeting NGF also deserve future investigations. Another strategy that seems encouraging is based on the modulation of second messangers by using PDE and ROCK inhibitors, and NO-donor drugs. Although the exciting expectations rose from gene therapy still need to be realized, the advances in this field are promising also in the clinical setting of OAB.
It is likely that the future will provides the clinicians with a variety of drugs, with distinctive mechanism of actions, to be used in combination or sequentially, and in groups of patients with different clinical phenotypes.
P- Reviewer: Khajehei M, Rovas L, Sonoda K, Yokoyama Y S- Editor: Ma YJ L- Editor: A E- Editor: Lu YJ
| 1. | Abrams P, Cardozo L, Fall M, Griffiths D, Rosier P, Ulmsten U, van Kerrebroeck P, Victor A, Wein A. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Neurourol Urodyn. 2002;21:167-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5099] [Cited by in RCA: 4669] [Article Influence: 203.0] [Reference Citation Analysis (1)] |
| 2. | Kelleher CJ, Kreder KJ, Pleil AM, Burgess SM, Reese PR. Long-term health-related quality of life of patients receiving extended-release tolterodine for overactive bladder. Am J Manag Care. 2002;8:S616-S630. [PubMed] |
| 3. | Sacco E, Tienforti D, D’Addessi A, Pinto F, Racioppi M, Totaro A, D’Agostino D, Marangi F, Bassi P. Social, economic, and health utility considerations in the treatment of overactive bladder. Open Access J Urol. 2010;2:11-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 4. | Sacco E. [Physiopathology of overactive bladder syndrome]. Urologia. 2012;79:24-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 5. | Sacco E, Bientinesi R, Tienforti D, Racioppi M, Gulino G, D’Agostino D, Vittori M, Bassi P. Discovery history and clinical development of mirabegron for the treatment of overactive bladder and urinary incontinence. Expert Opin Drug Discov. 2014;9:433-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 6. | Duthie JB, Vincent M, Herbison GP, Wilson DI, Wilson D. Botulinum toxin injections for adults with overactive bladder syndrome. Cochrane Database Syst Rev. 2011;CD005493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 87] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 7. | Sacco E, Pinto F, Bassi P. Emerging pharmacological targets in overactive bladder therapy: experimental and clinical evidences. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:583-598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 8. | Sacco E, Bientinesi R. Innovative pharmacotherapies for women with overactive bladder: where are we now and what is in the pipeline? Int Urogynecol J. 2015;26:629-640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 9. | Miyazato M, Yoshimura N, Nishijima S, Sugaya K. Roles of Glycinergic and Gamma-aminobutyric-ergic Mechanisms in the Micturition Reflex in Rats. Low Urin Tract Symptoms. 2009;1:S70-S73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 10. | Pehrson R, Lehmann A, Andersson KE. Effects of gamma-aminobutyrate B receptor modulation on normal micturition and oxyhemoglobin induced detrusor overactivity in female rats. J Urol. 2002;168:2700-2705. [PubMed] |
| 11. | Miyazato M, Sasatomi K, Hiragata S, Sugaya K, Chancellor MB, de Groat WC, Yoshimura N. GABA receptor activation in the lumbosacral spinal cord decreases detrusor overactivity in spinal cord injured rats. J Urol. 2008;179:1178-1183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Steers WD, Meythaler JM, Haworth C, Herrell D, Park TS. Effects of acute bolus and chronic continuous intrathecal baclofen on genitourinary dysfunction due to spinal cord pathology. J Urol. 1992;148:1849-1855. [PubMed] |
| 13. | Kalinichev M, Palea S, Haddouk H, Royer-Urios I, Guilloteau V, Lluel P, Schneider M, Saporito M, Poli S. ADX71441, a novel, potent and selective positive allosteric modulator of the GABA(B) receptor, shows efficacy in rodent models of overactive bladder. Br J Pharmacol. 2014;171:995-1006. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 14. | Pehrson R, Andersson KE. Effects of tiagabine, a gamma-aminobutyric acid re-uptake inhibitor, on normal rat bladder function. J Urol. 2002;167:2241-2246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 15. | Maneuf YP, Gonzalez MI, Sutton KS, Chung FZ, Pinnock RD, Lee K. Cellular and molecular action of the putative GABA-mimetic, gabapentin. Cell Mol Life Sci. 2003;60:742-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 100] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 16. | Carbone A, Palleschi G, Conte A, Bova G, Iacovelli E, Bettolo CM, Pastore A, Inghilleri M. Gabapentin treatment of neurogenic overactive bladder. Clin Neuropharmacol. 2006;29:206-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 17. | Kim YT, Kwon DD, Kim J, Kim DK, Lee JY, Chancellor MB. Gabapentin for overactive bladder and nocturia after anticholinergic failure. Int Braz J Urol. 2004;30:275-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 25] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 18. | Ansari MS, Bharti A, Kumar R, Ranjan P, Srivastava A, Kapoor R. Gabapentin: a novel drug as add-on therapy in cases of refractory overactive bladder in children. J Pediatr Urol. 2013;9:17-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 19. | Ansari MS; St. Luke’s Medical Center, Philippines. Efficacy and Safety of Gabapentin in Treating Overactive Bladder (OAB). In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). Available from: https://clinicaltrials.gov/ct2/show/NCT01486706. |
| 20. | Ansari MS; DDP-200. DrugBank. Available from: http://www.drugbank.ca/drugs/DB05919 Accession Number: DB05919. |
| 21. | Micheva KD, Taylor CP, Smith SJ. Pregabalin reduces the release of synaptic vesicles from cultured hippocampal neurons. Mol Pharmacol. 2006;70:467-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 84] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 22. | Micheva KD; Pfizer. Investigation of Tolterodine, Pregabalin and a Tolterodine - Pregabalin Combination for the Treatment of Overactive Bladder. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). Available from: http://clinicaltrials.gov/show/NCT00746681 NLM Identifier: NCT00746681. |
| 23. | Loutochin O, Al Afraa T, Campeau L, Mahfouz W, Elzayat E, Corcos J. Effect of the anticonvulsant medications pregabalin and lamotrigine on urodynamic parameters in an animal model of neurogenic detrusor overactivity. Neurourol Urodyn. 2012;31:1197-1202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 24. | Vogl C, Mochida S, Wolff C, Whalley BJ, Stephens GJ. The synaptic vesicle glycoprotein 2A ligand levetiracetam inhibits presynaptic Ca2+ channels through an intracellular pathway. Mol Pharmacol. 2012;82:199-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 82] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 25. | Elzayat EA, Campeau L, Karsenty G, Blok B, Padjen AL, Corcos J. Effect of antiepileptic agent, levetiracetam, on urodynamic parameters and neurogenic bladder overactivity in chronically paraplegic rats. Urology. 2009;73:922-927. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 26. | Miyazato M, Sugaya K, Goins WF, Wolfe D, Goss JR, Chancellor MB, de Groat WC, Glorioso JC, Yoshimura N. Herpes simplex virus vector-mediated gene delivery of glutamic acid decarboxylase reduces detrusor overactivity in spinal cord-injured rats. Gene Ther. 2009;16:660-668. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 34] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 27. | Eulenburg V, Armsen W, Betz H, Gomeza J. Glycine transporters: essential regulators of neurotransmission. Trends Biochem Sci. 2005;30:325-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 239] [Cited by in RCA: 255] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 28. | Yoshikawa S, Oguchi T, Funahashi Y, de Groat WC, Yoshimura N. Glycine transporter type 2 (GlyT2) inhibitor ameliorates bladder overactivity and nociceptive behavior in rats. Eur Urol. 2012;62:704-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 29. | Kafri R, Kodesh A, Shames J, Golomb J, Melzer I. Depressive symptoms and treatment of women with urgency urinary incontinence. Int Urogynecol J. 2013;24:1953-1959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 30. | Samsung Medical Center. Efficacy and Safety of Selective Serotonin Reuptake Inhibitor (SSRI) in Overactive Bladder Patients. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). Available from: http: //clinicaltrials.gov/show/NCT00902421 NLM Identifier: NCT00902421. |
| 31. | Steers WD, Herschorn S, Kreder KJ, Moore K, Strohbehn K, Yalcin I, Bump RC; Duloxetine OAB Study Group. Duloxetine compared with placebo for treating women with symptoms of overactive bladder. BJU Int. 2007;100:337-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 78] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 32. | Pérez-Martínez FC, Vela-Navarrete R, Virseda J, Ocaña AV, Lluel P, Rekik M, Bienaymé H, Ferté J, Attali P, Palea S. Halothane-anesthetized rabbit: a new experimental model to test the effects of besipirdine and duloxetine on lower urinary tract function. Urol Int. 2011;86:210-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 33. | Pérez-Martínez FC; UROGENE. Besipirdine. Available from: http: //urogene.free.fr/besipirdine.php. |
| 34. | Yoshiyama M, Kakizaki H, de Groat WC. Suppression of the micturition reflex in urethane-anesthetized rats by intracerebroventricular injection of WAY100635, a 5-HT(1A) receptor antagonist. Brain Res. 2003;980:281-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 35. | D’Agostino G, Condino AM, Gallinari P, Franceschetti GP, Tonini M. Characterization of prejunctional serotonin receptors modulating [3H]acetylcholine release in the human detrusor. J Pharmacol Exp Ther. 2006;316:129-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 37] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 36. | Mbaki Y, Gardiner J, McMurray G, Ramage AG. 5-HT 2A receptor activation of the external urethral sphincter and 5-HT 2C receptor inhibition of micturition: a study based on pharmacokinetics in the anaesthetized female rat. Eur J Pharmacol. 2012;682:142-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 37. | Tai C, Miscik CL, Ungerer TD, Roppolo JR, de Groat WC. Suppression of bladder reflex activity in chronic spinal cord injured cats by activation of serotonin 5-HT1A receptors. Exp Neurol. 2006;199:427-437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 38. | Chang HH, Havton LA. Serotonergic 5-HT(1A) receptor agonist (8-OH-DPAT) ameliorates impaired micturition reflexes in a chronic ventral root avulsion model of incomplete cauda equina/conus medullaris injury. Exp Neurol. 2013;239:210-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 39. | Kodama M, Takimoto Y. Influence of 5-hydroxytryptamine and the effect of a new serotonin receptor antagonist (sarpogrelate) on detrusor smooth muscle of streptozotocin-induced diabetes mellitus in the rat. Int J Urol. 2000;7:231-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 40. | Takimoto Y, Kodama M, Sugimoto S, Hamada T, Fuse T, Kawata N, Hirakata H, Hosokawa H. [The effect of 5-HT 2 antagonist for urinary frequency symptom on diabetes mellitus patients]. Nihon Hinyokika Gakkai Zasshi. 1999;90:731-740. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 41. | Darblade B, Behr-Roussel D, Gorny D, Lebret T, Benoit G, Hieble JP, Brooks D, Alexandre L, Giuliano F. Piboserod (SB 207266), a selective 5-HT4 receptor antagonist, reduces serotonin potentiation of neurally-mediated contractile responses of human detrusor muscle. World J Urol. 2005;23:147-151. [PubMed] |
| 42. | Recordati S. p.A. Available from: http://www.recordati.it/~/media/Files/R/Recordati-V2/investors/presentations/2005it/2005-09-22.pdf. |
| 43. | Reese J, Xiao Z, Schwen Z, Matsuta Y, Shen B, Wang J, Roppolo JR, de Groat WC, Tai C. Effects of duloxetine and WAY100635 on pudendal inhibition of bladder overactivity in cats. J Pharmacol Exp Ther. 2014;349:402-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 44. | Schwen Z, Matsuta Y, Shen B, Wang J, Roppolo JR, de Groat WC, Tai C. Involvement of 5-HT3 receptors in pudendal inhibition of bladder overactivity in cats. Am J Physiol Renal Physiol. 2013;305:F663-F671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 45. | 2-(1-piperazinyl) Thieno[2,3-d]pyrimidine Derivatives Having 5-HT3 Receptor Antagonist Activity and NorAdrenaline Reuptake Inhibitor (NARI) Activity; Nocturia or Overactive Bladder. Patent US7115606 B2. 2006; Available from: http: //www.google.it/patents/US7115606. |
| 46. | Burnstock G. Therapeutic potential of purinergic signalling for diseases of the urinary tract. BJU Int. 2011;107:192-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 53] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 47. | Ruggieri MR. Mechanisms of disease: role of purinergic signaling in the pathophysiology of bladder dysfunction. Nat Clin Pract Urol. 2006;3:206-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 54] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 48. | Zhang HP, Li CL, Lu P, Zheng JC, Yu LL, Yang WM, Xiong F, Zeng XY. The function of P2X3 receptor and NK1 receptor antagonists on cyclophosphamide-induced cystitis in rats. World J Urol. 2014;32:91-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 49. | Uvin P, Boudes M, Menigoz A, Franken J, Pinto S, Gevaert T, Verplaetse R, Tytgat J, Vennekens R, Voets T. Chronic administration of anticholinergics in rats induces a shift from muscarinic to purinergic transmission in the bladder wall. Eur Urol. 2013;64:502-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 50. | Yoshida M, Homma Y, Inadome A, Yono M, Seshita H, Miyamoto Y, Murakami S, Kawabe K, Ueda S. Age-related changes in cholinergic and purinergic neurotransmission in human isolated bladder smooth muscles. Exp Gerontol. 2001;36:99-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 131] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 51. | O’Reilly BA, Kosaka AH, Knight GF, Chang TK, Ford AP, Rymer JM, Popert R, Burnstock G, McMahon SB. P2X receptors and their role in female idiopathic detrusor instability. J Urol. 2002;167:157-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 116] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 52. | Brady CM, Apostolidis A, Yiangou Y, Baecker PA, Ford AP, Freeman A, Jacques TS, Fowler CJ, Anand P. P2X3-immunoreactive nerve fibres in neurogenic detrusor overactivity and the effect of intravesical resiniferatoxin. Eur Urol. 2004;46:247-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 129] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 53. | North RA, Jarvis MF. P2X receptors as drug targets. Mol Pharmacol. 2013;83:759-769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 232] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 54. | Kaan TKY, Yip PK, Grist J, Gever JR, Cefalu JS, Nunn PA, Ford AP, Zhong Y, McMahon SB. Selective Blockade of Spinal P2X3 and P2X2/3 Receptors with a Novel and Selective Antagonist, AF-742, Reveals Central Endogenous Purinergic Regulation of Bladder Activity. Abstract 602 at. 2010;Joint Annual Meeting of the International Continence Society and the International Urogynecological Association in Toronto, Canada Available from: http://www.ics.org/Abstracts/Publish/105/000602.pdf. |
| 55. | Hemmings-Mieszczak M, Dorn G, Natt FJ, Hall J, Wishart WL. Independent combinatorial effect of antisense oligonucleotides and RNAi-mediated specific inhibition of the recombinant rat P2X3 receptor. Nucleic Acids Res. 2003;31:2117-2126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 59] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 56. | Lecci A, Maggi CA. Tachykinins as modulators of the micturition reflex in the central and peripheral nervous system. Regul Pept. 2001;101:1-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 70] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 57. | Seki S, Erickson KA, Seki M, Nishizawa O, Igawa Y, Ogawa T, de Groat WC, Chancellor MB, Yoshimura N. Elimination of rat spinal neurons expressing neurokinin 1 receptors reduces bladder overactivity and spinal c-fos expression induced by bladder irritation. Am J Physiol Renal Physiol. 2005;288:F466-F473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 37] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 58. | Ishizuka O, Igawa Y, Nishizawa O, Andersson KE. Role of supraspinal tachykinins for volume- and L-dopa-induced bladder activity in normal conscious rats. Neurourol Urodyn. 2000;19:101-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 59. | Ishizuka O, Igawa Y, Lecci A, Maggi CA, Mattiasson A, Andersson KE. Role of intrathecal tachykinins for micturition in unanaesthetized rats with and without bladder outlet obstruction. Br J Pharmacol. 1994;113:111-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 60. | Andersson KE, Wein AJ. Pharmacology of the lower urinary tract: basis for current and future treatments of urinary incontinence. Pharmacol Rev. 2004;56:581-631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 362] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 61. | Abdel-Gawad M, Dion SB, Elhilali MM. Evidence of a peripheral role of neurokinins in detrusor hyperreflexia: a further study of selective tachykinin antagonists in chronic spinal injured rats. J Urol. 2001;165:1739-1744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 62. | Abdel-Karim AM, Barthlow HG, Bialecki RA, Elhilali MM. Effects of M274773, a neurokinin-2 receptor antagonist, on bladder function in chronically spinalized rats. J Urol. 2005;174:1488-1492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 63. | Green SA, Alon A, Ianus J, McNaughton KS, Tozzi CA, Reiss TF. Efficacy and safety of a neurokinin-1 receptor antagonist in postmenopausal women with overactive bladder with urge urinary incontinence. J Urol. 2006;176:2535-2540; discussion 2540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 67] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 64. | Frenkl TL, Zhu H, Reiss T, Seltzer O, Rosenberg E, Green S. A multicenter, double-blind, randomized, placebo controlled trial of a neurokinin-1 receptor antagonist for overactive bladder. J Urol. 2010;184:616-622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 47] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 65. | Haab F, Braticevici B, Krivoborodov G, Palmas M, Zufferli Russo M, Pietra C. Efficacy and safety of repeated dosing of netupitant, a neurokinin-1 receptor antagonist, in treating overactive bladder. Neurourol Urodyn. 2014;33:335-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 66. | Andersson KE, Gratzke C, Hedlund P. The role of the transient receptor potential (TRP) superfamily of cation-selective channels in the management of the overactive bladder. BJU Int. 2010;106:1114-1127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 90] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 67. | Birder LA, de Groat WC. Mechanisms of disease: involvement of the urothelium in bladder dysfunction. Nat Clin Pract Urol. 2007;4:46-54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 296] [Cited by in RCA: 280] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 68. | de Groat WC, Yoshimura N. Changes in afferent activity after spinal cord injury. Neurourol Urodyn. 2010;29:63-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 133] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 69. | Wiart L, Joseph PA, Petit H, Dosque JP, de Sèze M, Brochet B, Deminière C, Ferrière JM, Mazaux JM, N’Guyen P. The effects of capsaicin on the neurogenic hyperreflexic detrusor. A double blind placebo controlled study in patients with spinal cord disease. Preliminary results. Spinal Cord. 1998;36:95-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 70. | de Sèze M, Gallien P, Denys P, Labat JJ, Serment G, Grise P, Salle JY, Blazejewski S, Hazane C, Moore N. Intravesical glucidic capsaicin versus glucidic solvent in neurogenic detrusor overactivity: a double blind controlled randomized study. Neurourol Urodyn. 2006;25:752-757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 71. | Silva C, Silva J, Ribeiro MJ, Avelino A, Cruz F. Urodynamic effect of intravesical resiniferatoxin in patients with neurogenic detrusor overactivity of spinal origin: results of a double-blind randomized placebo-controlled trial. Eur Urol. 2005;48:650-655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 45] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 72. | Kuo HC, Liu HT, Yang WC. Therapeutic effect of multiple resiniferatoxin intravesical instillations in patients with refractory detrusor overactivity: a randomized, double-blind, placebo controlled study. J Urol. 2006;176:641-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 73. | Santos-Silva A, Charrua A, Cruz CD, Gharat L, Avelino A, Cruz F. Rat detrusor overactivity induced by chronic spinalization can be abolished by a transient receptor potential vanilloid 1 (TRPV1) antagonist. Auton Neurosci. 2012;166:35-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 30] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 74. | Gavva NR, Treanor JJ, Garami A, Fang L, Surapaneni S, Akrami A, Alvarez F, Bak A, Darling M, Gore A. Pharmacological blockade of the vanilloid receptor TRPV1 elicits marked hyperthermia in humans. Pain. 2008;136:202-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 352] [Cited by in RCA: 395] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 75. | Round P, Priestley A, Robinson J. An investigation of the safety and pharmacokinetics of the novel TRPV1 antagonist XEN-D0501 in healthy subjects. Br J Clin Pharmacol. 2011;72:921-931. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 50] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 76. | Novartis Pharmaceuticals. Safety and Efficacy of SAF312 in Patients With Neurogenic Detrusor Overactivity Due to Spinal Cord Lesions. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). Available from: http: //clinicaltrials.gov/show/NCT01598103 NLM Identifier: NCT01598103. |
| 77. | Juszczak K, Ziomber A, Thor PJ. Effect of partial and complete blockade of vanilloid (TRPV1-6) and ankyrin (TRPA1) transient receptor potential ion channels on urinary bladder motor activity in an experimental hyperosmolar overactive bladder rat model. J Physiol Pharmacol. 2011;62:321-326. [PubMed] |
| 78. | Andrade EL, Forner S, Bento AF, Leite DF, Dias MA, Leal PC, Koepp J, Calixto JB. TRPA1 receptor modulation attenuates bladder overactivity induced by spinal cord injury. Am J Physiol Renal Physiol. 2011;300:F1223-F1234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 62] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 79. | Mochizuki T, Sokabe T, Araki I, Fujishita K, Shibasaki K, Uchida K, Naruse K, Koizumi S, Takeda M, Tominaga M. The TRPV4 cation channel mediates stretch-evoked Ca2+ influx and ATP release in primary urothelial cell cultures. J Biol Chem. 2009;284:21257-21264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 227] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 80. | Aizawa N, Wyndaele JJ, Homma Y, Igawa Y. Effects of TRPV4 cation channel activation on the primary bladder afferent activities of the rat. Neurourol Urodyn. 2012;31:148-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 55] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 81. | Cho KJ, Park EY, Kim HS, Koh JS, Kim JC. Expression of transient receptor potential vanilloid 4 and effects of ruthenium red on detrusor overactivity associated with bladder outlet obstruction in rats. World J Urol. 2014;32:677-682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 82. | Everaerts W, Zhen X, Ghosh D, Vriens J, Gevaert T, Gilbert JP, Hayward NJ, McNamara CR, Xue F, Moran MM. Inhibition of the cation channel TRPV4 improves bladder function in mice and rats with cyclophosphamide-induced cystitis. Proc Natl Acad Sci USA. 2010;107:19084-19089. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 329] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 83. | Grond S, Sablotzki A. Clinical pharmacology of tramadol. Clin Pharmacokinet. 2004;43:879-923. [PubMed] |
| 84. | Safarinejad MR, Hosseini SY. Safety and efficacy of tramadol in the treatment of idiopathic detrusor overactivity: a double-blind, placebo-controlled, randomized study. Br J Clin Pharmacol. 2006;61:456-463. [PubMed] |
| 85. | Singh SK, Agarwal MM, Batra YK, Kishore AV, Mandal AK. Effect of lumbar-epidural administration of tramadol on lower urinary tract function. Neurourol Urodyn. 2008;27:65-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 86. | KeyNeurotek Pharma GmbH. Double-blind placebo-controlled proof of concept study with the µ-receptor agonist KN203 in patients with overactive bladder syndrome Double-blind, randomized, parallel groups prospective phase II-a study for 8 weeks with two dosages of KN203 (20 and 40 mg b.i.d.) and placebo. National Competent Authority: Germany - BfArM. Available from: http:// www.clinicaltrialsregister.eu[EudraCT Number: 2009-013334-24] EudraCT Number: 2009-013334-24. |
| 87. | Dray A, Nunan L, Wire W. Central delta-opioid receptor interactions and the inhibition of reflex urinary bladder contractions in the rat. Br J Pharmacol. 1985;85:717-726. [PubMed] |
| 88. | Holt JD, Watson MJ, Chang JP, O’Neill SJ, Wei K, Pendergast W, Gengo PJ, Chang KJ. DPI-221 [4-((alpha-s)-alpha-((2s,5r)-2,5-dimethyl-4-(3-fluorobenzyl)-1-piperazinyl)benzyl)-N,N-diethylbenzamide]: a novel nonpeptide delta receptor agonist producing increased micturition interval in normal rats. J Pharmacol Exp Ther. 2005;315:601-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 89. | Meunier JC. Nociceptin/orphanin FQ and the opioid receptor-like ORL1 receptor. Eur J Pharmacol. 1997;340:1-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 315] [Cited by in RCA: 301] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 90. | Lecci A, Giuliani S, Tramontana M, Criscuoli M, Maggi CA. Multiple sites of action in the inhibitory effect of nociceptin on the micturition reflex. J Urol. 2000;163:638-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 91. | Lazzeri M, Calò G, Spinelli M, Guerrini R, Beneforti P, Sandri S, Zanollo A, Regoli D, Turini D. Urodynamic and clinical evidence of acute inhibitory effects of intravesical nociceptin/orphanin FQ on detrusor overactivity in humans: a pilot study. J Urol. 2001;166:2237-2240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 35] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 92. | Lazzeri M, Calò G, Spinelli M, Guerrini R, Salvadori S, Beneforti P, Sandri S, Regoli D, Turini D. Urodynamic effects of intravesical nociceptin/orphanin FQ in neurogenic detrusor overactivity: a randomized, placebo-controlled, double-blind study. Urology. 2003;61:946-950. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 93. | Lazzeri M, Calò G, Spinelli M, Malaguti S, Guerrini R, Salvadori S, Beneforti P, Regoli D, Turini D. Daily intravesical instillation of 1 mg nociceptin/orphanin FQ for the control of neurogenic detrusor overactivity: a multicenter, placebo controlled, randomized exploratory study. J Urol. 2006;176:2098-2102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 94. | Hedlund P. Cannabinoids and the endocannabinoid system in lower urinary tract function and dysfunction. Neurourol Urodyn. 2014;33:46-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 52] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 95. | Zhao P, Abood ME. GPR55 and GPR35 and their relationship to cannabinoid and lysophospholipid receptors. Life Sci. 2013;92:453-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 96. | Hayn MH, Ballesteros I, de Miguel F, Coyle CH, Tyagi S, Yoshimura N, Chancellor MB, Tyagi P. Functional and immunohistochemical characterization of CB1 and CB2 receptors in rat bladder. Urology. 2008;72:1174-1178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 54] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 97. | Strittmatter F, Gandaglia G, Benigni F, Bettiga A, Rigatti P, Montorsi F, Gratzke C, Stief C, Colciago G, Hedlund P. Expression of fatty acid amide hydrolase (FAAH) in human, mouse, and rat urinary bladder and effects of FAAH inhibition on bladder function in awake rats. Eur Urol. 2012;61:98-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 98. | Tyagi V, Philips BJ, Su R, Smaldone MC, Erickson VL, Chancellor MB, Yoshimura N, Tyagi P. Differential expression of functional cannabinoid receptors in human bladder detrusor and urothelium. J Urol. 2009;181:1932-1938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 76] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 99. | Mukerji G, Yiangou Y, Agarwal SK, Anand P. Increased cannabinoid receptor 1-immunoreactive nerve fibers in overactive and painful bladder disorders and their correlation with symptoms. Urology. 2010;75:1514.e15-1514.e20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 44] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 100. | Katagigiotis S, Kavia R, Gonzales G. Is there a local bladder effect of oral cannabinoid agonists? Eur Urol Suppl. 2012;11:E370. |
| 101. | Campeau L, Fullhase C, Russo A. Intrathecal cannabinoid agonist effects in cystometric evaluation of normal rats [abstract MP-09.06]. Can Urol Assoc J. 2012;6:S65. |
| 102. | Gratzke C, Streng T, Stief CG, Downs TR, Alroy I, Rosenbaum JS, Andersson KE, Hedlund P. Effects of cannabinor, a novel selective cannabinoid 2 receptor agonist, on bladder function in normal rats. Eur Urol. 2010;57:1093-1100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 48] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 103. | Gratzke C, Streng T, Stief CG, Alroy I, Limberg BJ, Downs TR, Rosenbaum JS, Hedlund P, Andersson KE. Cannabinor, a selective cannabinoid-2 receptor agonist, improves bladder emptying in rats with partial urethral obstruction. J Urol. 2011;185:731-736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 104. | Füllhase C, Russo A, Castiglione F, Benigni F, Campeau L, Montorsi F, Gratzke C, Bettiga A, Stief C, Andersson KE. Spinal cord FAAH in normal micturition control and bladder overactivity in awake rats. J Urol. 2013;189:2364-2370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 105. | Brady CM, DasGupta R, Dalton C, Wiseman OJ, Berkley KJ, Fowler CJ. An open-label pilot study of cannabis-based extracts for bladder dysfunction in advanced multiple sclerosis. Mult Scler. 2004;10:425-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 152] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 106. | Freeman RM, Adekanmi O, Waterfield MR, Waterfield AE, Wright D, Zajicek J. The effect of cannabis on urge incontinence in patients with multiple sclerosis: a multicentre, randomised placebo-controlled trial (CAMS-LUTS). Int Urogynecol J Pelvic Floor Dysfunct. 2006;17:636-641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 140] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 107. | Kavia RB, De Ridder D, Constantinescu CS, Stott CG, Fowler CJ. Randomized controlled trial of Sativex to treat detrusor overactivity in multiple sclerosis. Mult Scler. 2010;16:1349-1359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 118] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 108. | Apostolidis A. Taming the cannabinoids: new potential in the pharmacologic control of lower urinary tract dysfunction. Eur Urol. 2012;61:107-109; discussion 109-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 109. | Aizawa N, Hedlund P, Füllhase C, Ito H, Homma Y, Igawa Y. Inhibition of peripheral FAAH depresses activities of bladder mechanosensitive nerve fibers of the rat. J Urol. 2014;192:956-963. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 110. | Walczak JS, Price TJ, Cervero F. Cannabinoid CB1 receptors are expressed in the mouse urinary bladder and their activation modulates afferent bladder activity. Neuroscience. 2009;159:1154-1163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 61] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 111. | Debruyne F, Tzvetkov M, Altarac S, Geavlete PA. Dose-ranging study of the luteinizing hormone-releasing hormone receptor antagonist cetrorelix pamoate in the treatment of patients with symptomatic benign prostatic hyperplasia. Urology. 2010;76:927-933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 112. | AEterna Zentaris. Cetrorelix Pamoate Regimens in Patients With Symptomatic Benign Prostatic Hypertrophy (BPH). In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). Available from: http: //clinicaltrials.gov/show/NCT00449150. |
| 113. | Gandaglia G, Benigni F, La Croce G, Moschini M, Strittmatter F, Colli E, Andersson KE, Hedlund P. Degarelix reduces urodynamic changes in a rat model for experimental detrusor overactivity and causes more efficient voiding. J Urol Suppl. 2011;185:E322. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 114. | Russo A, Castiglione F, Salonia A, Benigni F, Rigatti P, Montorsi F, Andersson KE, Hedlund P. Effects of the gonadotropin-releasing hormone antagonist ganirelix on normal micturition and prostaglandin E(2)-induced detrusor overactivity in conscious female rats. Eur Urol. 2011;59:868-874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 115. | Truss MC, Stief CG, Uckert S, Becker AJ, Wefer J, Schultheiss D, Jonas U. Phosphodiesterase 1 inhibition in the treatment of lower urinary tract dysfunction: from bench to bedside. World J Urol. 2001;19:344-350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 93] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 116. | Truss MC, Uckert S, Stief CG, Forssmann WG, Jonas U. Cyclic nucleotide phosphodiesterase (PDE) isoenzymes in the human detrusor smooth muscle. II. Effect of various PDE inhibitors on smooth muscle tone and cyclic nucleotide levels in vitro. Urol Res. 1996;24:129-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 117. | Andersson KE, Uckert S, Stief C, Hedlund P. Phosphodiesterases (PDEs) and PDE inhibitors for treatment of LUTS. Neurourol Urodyn. 2007;26:928-933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 60] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 118. | Truss MC, Stief CG, Uckert S, Becker AJ, Schultheiss D, Machtens S, Jonas U. Initial clinical experience with the selective phosphodiesterase-I isoenzyme inhibitor vinpocetine in the treatment of urge incontinence and low compliance bladder. World J Urol. 2000;18:439-443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 47] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 119. | Oger S, Behr-Roussel D, Gorny D, Denys P, Lebret T, Alexandre L, Giuliano F. Relaxation of phasic contractile activity of human detrusor strips by cyclic nucleotide phosphodiesterase type 4 inhibition. Eur Urol. 2007;51:772-780; discussion 780-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 120. | Kaiho Y, Nishiguchi J, Kwon DD, Chancellor MB, Arai Y, Snyder PB, Yoshimura N. The effects of a type 4 phosphodiesterase inhibitor and the muscarinic cholinergic antagonist tolterodine tartrate on detrusor overactivity in female rats with bladder outlet obstruction. BJU Int. 2008;101:615-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 121. | Nishiguchi J, Kwon DD, Kaiho Y, Chancellor MB, Kumon H, Snyder PB, Yoshimura N. Suppression of detrusor overactivity in rats with bladder outlet obstruction by a type 4 phosphodiesterase inhibitor. BJU Int. 2007;99:680-686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 122. | Available from: http: //clinicaltrials.gov/show/NCT02038868.. |
| 123. | Oger S, Behr-Roussel D, Gorny D, Lebret T, Validire P, Cathelineau X, Alexandre L, Giuliano F. Signalling pathways involved in sildenafil-induced relaxation of human bladder dome smooth muscle. Br J Pharmacol. 2010;160:1135-1143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 63] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 124. | Fusco F, di Villa Bianca Rd, Mitidieri E, Cirino G, Sorrentino R, Mirone V. Sildenafil effect on the human bladder involves the L-cysteine/hydrogen sulfide pathway: a novel mechanism of action of phosphodiesterase type 5 inhibitors. Eur Urol. 2012;62:1174-1180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 69] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 125. | Liu L, Zheng S, Han P, Wei Q. Phosphodiesterase-5 inhibitors for lower urinary tract symptoms secondary to benign prostatic hyperplasia: a systematic review and meta-analysis. Urology. 2011;77:123-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 38] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 126. | Gacci M, Corona G, Salvi M, Vignozzi L, McVary KT, Kaplan SA, Roehrborn CG, Serni S, Mirone V, Carini M. A systematic review and meta-analysis on the use of phosphodiesterase 5 inhibitors alone or in combination with α-blockers for lower urinary tract symptoms due to benign prostatic hyperplasia. Eur Urol. 2012;61:994-1003. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 227] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 127. | Scatena R. Nitroflurbiprofen (NicOx). Curr Opin Investig Drugs. 2004;5:551-556. [PubMed] |
| 128. | Rufford J, Cardozo L, Toozs-Hobson P, Dixon A. HCT1026-A novel treatment for urgency? Heidelberg, Germany: 32nd Annual Meeting of International Continence Society, 2002. Abs 225 Available from: http: //www.ics.org/Abstracts/Publish/40/000225.pdf. |
| 129. | Available from: http://www.nicox.com/uploads/default/files//pr2002040200en.pdf. |
| 130. | Jeremy JY, Tsang V, Mikhailidis DP, Rogers H, Morgan RJ, Dandona P. Eicosanoid synthesis by human urinary bladder mucosa: pathological implications. Br J Urol. 1987;59:36-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 70] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 131. | Khan MA, Thompson CS, Mumtaz FH, Jeremy JY, Morgan RJ, Mikhailidis DP. Role of prostaglandins in the urinary bladder: an update. Prostaglandins Leukot Essent Fatty Acids. 1998;59:415-422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 132. | Masunaga K, Yoshida M, Inadome A, Iwashita H, Miyamae K, Ueda S. Prostaglandin E2 release from isolated bladder strips in rats with spinal cord injury. Int J Urol. 2006;13:271-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 133. | Schröder A, Newgreen D, Andersson KE. Detrusor responses to prostaglandin E2 and bladder outlet obstruction in wild-type and Ep1 receptor knockout mice. J Urol. 2004;172:1166-1170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 71] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 134. | Lee T, Hedlund P, Newgreen D, Andersson KE. Urodynamic effects of a novel EP(1) receptor antagonist in normal rats and rats with bladder outlet obstruction. J Urol. 2007;177:1562-1567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 56] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 135. | Schüssler B. Comparison of the mode of action of prostaglandin E2 (PGE2) and sulprostone, a PGE2-derivative, on the lower urinary tract in healthy women. A urodynamic study. Urol Res. 1990;18:349-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 60] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 136. | Cardozo LD, Stanton SL, Robinson H, Hole D. Evaluation of flurbiprofen in detrusor instability. Br Med J. 1980;280:281-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 76] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 137. | Cardozo LD, Stanton SL. A comparison between bromocriptine and indomethacin in the treatment of detrusor instability. J Urol. 1980;123:399-401. [PubMed] |
| 138. | Angelico P, Guarneri L, Velasco C, Cova R, Leonardi A, Clarke DE, Testa R. Effect of cyclooxygenase inhibitors on the micturition reflex in rats: correlation with inhibition of cyclooxygenase isozymes. BJU Int. 2006;97:837-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 139. | Jang J, Park EY, Seo SI, Hwang TK, Kim JC. Effects of intravesical instillation of cyclooxygenase-2 inhibitor on the expression of inducible nitric oxide synthase and nerve growth factor in cyclophosphamide-induced overactive bladder. BJU Int. 2006;98:435-439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 140. | Ratz PH, Speich JE, Klausner AP. COX Inhibitors and Overactive Bladder: The Potential for Future Therapy. Curr Bladder Dysfunct Rep. 2010;5:4-12. |
| 141. | Rahnama’i MS, van Koeveringe GA, Essers PB, de Wachter SG, de Vente J, van Kerrebroeck PE, Gillespie JI. Prostaglandin receptor EP1 and EP2 site in guinea pig bladder urothelium and lamina propria. J Urol. 2010;183:1241-1247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 46] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 142. | McCafferty GP, Misajet BA, Laping NJ, Edwards RM, Thorneloe KS. Enhanced bladder capacity and reduced prostaglandin E2-mediated bladder hyperactivity in EP3 receptor knockout mice. Am J Physiol Renal Physiol. 2008;295:F507-F514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 143. | Chapple CR, Abrams P, Andersson KE, Radziszewski P, Masuda T, Small M, Kuwayama T, Deacon S. Phase II study on the efficacy and safety of the EP1 receptor antagonist ONO-8539 for nonneurogenic overactive bladder syndrome. J Urol. 2014;191:253-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 144. | Teixeira CE, Jin L, Priviero FB, Ying Z, Webb RC. Comparative pharmacological analysis of Rho-kinase inhibitors and identification of molecular components of Ca2+ sensitization in the rat lower urinary tract. Biochem Pharmacol. 2007;74:647-658. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 145. | Marx JO, Basha ME, Mohanan S, Hypolite JA, Chang S, Wein AJ, Zderic SA, Laping NJ, Chacko S. Effects of Rho-kinase inhibition on myosin light chain phosphorylation and obstruction-induced detrusor overactivity. Int J Urol. 2014;21:319-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 146. | Kirschstein T, Protzel C, Porath K, Sellmann T, Köhling R, Hakenberg OW. Age-dependent contribution of Rho kinase in carbachol-induced contraction of human detrusor smooth muscle in vitro. Acta Pharmacol Sin. 2014;35:74-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 147. | Wibberley A, Chen Z, Hu E, Hieble JP, Westfall TD. Expression and functional role of Rho-kinase in rat urinary bladder smooth muscle. Br J Pharmacol. 2003;138:757-766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 133] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 148. | Christ GJ, Andersson KE. Rho-kinase and effects of Rho-kinase inhibition on the lower urinary tract. Neurourol Urodyn. 2007;26:948-954. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 76] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 149. | Tatsumiya K, Yamanishi T, Watanabe M, Masuda A, Mizuno T, Kamai T, Yoshida K. Effects of fasudil, a Rho-kinase inhibitor, on contraction of pig bladder tissues with or without urothelium. Int J Urol. 2009;16:959-966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 150. | Rajasekaran M, Wilkes N, Kuntz S, E Albo M. Rho-kinase inhibition suppresses bladder hyperactivity in spontaneously hypertensive rats. Neurourol Urodyn. 2005;24:295-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 67] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 151. | Kim JY, Han JY, Kwon TG, Choo MS. Effects of oral Rho kinase inhibitor fasudil on detrusor overactivity after bladder outlet obstruction in rats. LUTS. 2013;5:96-100. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 152. | Schröder A, Colli E, Maggi M, Andersson KE. Effects of a vitamin D(3) analogue in a rat model of bladder outlet obstruction. BJU Int. 2006;98:637-642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 153. | Adorini L, Penna G, Fibbi B, Maggi M. Vitamin D receptor agonists target static, dynamic, and inflammatory components of benign prostatic hyperplasia. Ann N Y Acad Sci. 2010;1193:146-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 50] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 154. | Morelli A, Vignozzi L, Filippi S, Vannelli GB, Ambrosini S, Mancina R, Crescioli C, Donati S, Fibbi B, Colli E. BXL-628, a vitamin D receptor agonist effective in benign prostatic hyperplasia treatment, prevents RhoA activation and inhibits RhoA/Rho kinase signaling in rat and human bladder. Prostate. 2007;67:234-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 54] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 155. | Morelli A, Squecco R, Failli P, Filippi S, Vignozzi L, Chavalmane AK, Fibbi B, Mancina R, Luciani G, Gacci M. The vitamin D receptor agonist elocalcitol upregulates L-type calcium channel activity in human and rat bladder. Am J Physiol Cell Physiol. 2008;294:C1206-C1214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 156. | Shapiro B, Redman TL, Zvara P. Effects of vitamin D analog on bladder function and sensory signaling in animal models of cystitis. Urology. 2013;81:466.e1-466.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 157. | Digesu GA, Verdi E, Cardozo L, Olivieri L, Khullar V, Colli E. Phase IIb, multicenter, double-blind, randomized, placebo-controlled, parallel-group study to determine effects of elocalcitol in women with overactive bladder and idiopathic detrusor overactivity. Urology. 2012;80:48-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 158. | Steers WD, Kolbeck S, Creedon D, Tuttle JB. Nerve growth factor in the urinary bladder of the adult regulates neuronal form and function. J Clin Invest. 1991;88:1709-1715. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 302] [Cited by in RCA: 296] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 159. | Liu HT, Chancellor MB, Kuo HC. Decrease of urinary nerve growth factor levels after antimuscarinic therapy in patients with overactive bladder. BJU Int. 2009;103:1668-1672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 83] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 160. | Liu HT, Chancellor MB, Kuo HC. Urinary nerve growth factor levels are elevated in patients with detrusor overactivity and decreased in responders to detrusor botulinum toxin-A injection. Eur Urol. 2009;56:700-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 132] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 161. | Schnegelsberg B, Sun TT, Cain G, Bhattacharya A, Nunn PA, Ford AP, Vizzard MA, Cockayne DA. Overexpression of NGF in mouse urothelium leads to neuronal hyperinnervation, pelvic sensitivity, and changes in urinary bladder function. Am J Physiol Regul Integr Comp Physiol. 2010;298:R534-R547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 130] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 162. | Seki S, Sasaki K, Fraser MO, Igawa Y, Nishizawa O, Chancellor MB, de Groat WC, Yoshimura N. Immunoneutralization of nerve growth factor in lumbosacral spinal cord reduces bladder hyperreflexia in spinal cord injured rats. J Urol. 2002;168:2269-2274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 132] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 163. | Kashyap M, Kawamorita N, Tyagi V, Sugino Y, Chancellor M, Yoshimura N, Tyagi P. Down-regulation of nerve growth factor expression in the bladder by antisense oligonucleotides as new treatment for overactive bladder. J Urol. 2013;190:757-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 164. | Abdel-Karim AM, Bialecki RA, Elhilali MM. Effects of ZD6169 and ZD0947, 2 potassium adenosine triphosphate channel openers, on bladder function of spinalized rats. J Urol. 2002;168:837-842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 165. | Pandita RK, Andersson KE. Effects of intravesical administration of the K+ channel opener, ZD6169, in conscious rats with and without bladder outflow obstruction. J Urol. 1999;162:943-948. [PubMed] |
| 166. | Tanaka M, Sasaki Y, Kimura Y, Fukui T, Hamada K, Ukai Y. A novel pyrrole derivative, NS-8, suppresses the rat micturition reflex by inhibiting afferent pelvic nerve activity. BJU Int. 2003;92:1031-1036. [PubMed] |
| 167. | Petkov GV. Role of potassium ion channels in detrusor smooth muscle function and dysfunction. Nat Rev Urol. 2012;9:30-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 124] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 168. | Meredith AL, Thorneloe KS, Werner ME, Nelson MT, Aldrich RW. Overactive bladder and incontinence in the absence of the BK large conductance Ca2+-activated K+ channel. J Biol Chem. 2004;279:36746-36752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 292] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 169. | Thorneloe KS, Meredith AL, Knorn AM, Aldrich RW, Nelson MT. Urodynamic properties and neurotransmitter dependence of urinary bladder contractility in the BK channel deletion model of overactive bladder. Am J Physiol Renal Physiol. 2005;289:F604-F610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 74] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 170. | Hristov KL, Chen M, Kellett WF, Rovner ES, Petkov GV. Large-conductance voltage- and Ca2+-activated K+ channels regulate human detrusor smooth muscle function. Am J Physiol Cell Physiol. 2011;301:C903-C912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 81] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 171. | Hristov KL, Cui X, Brown SM, Liu L, Kellett WF, Petkov GV. Stimulation of beta3-adrenoceptors relaxes rat urinary bladder smooth muscle via activation of the large-conductance Ca2+-activated K+ channels. Am J Physiol Cell Physiol. 2008;295:C1344-C1353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 71] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 172. | Chang S, Gomes CM, Hypolite JA, Marx J, Alanzi J, Zderic SA, Malkowicz B, Wein AJ, Chacko S. Detrusor overactivity is associated with downregulation of large-conductance calcium- and voltage-activated potassium channel protein. Am J Physiol Renal Physiol. 2010;298:F1416-F1423. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 173. | Hristov KL, Afeli SA, Parajuli SP, Cheng Q, Rovner ES, Petkov GV. Neurogenic detrusor overactivity is associated with decreased expression and function of the large conductance voltage- and Ca(2+)-activated K(+) channels. PLoS One. 2013;8:e68052. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 174. | Thorneloe KS, Nelson MT. Properties and molecular basis of the mouse urinary bladder voltage-gated K+ current. J Physiol. 2003;549:65-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 62] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 175. | Yoshimura N, Bennett NE, Hayashi Y, Ogawa T, Nishizawa O, Chancellor MB, de Groat WC, Seki S. Bladder overactivity and hyperexcitability of bladder afferent neurons after intrathecal delivery of nerve growth factor in rats. J Neurosci. 2006;26:10847-10855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 143] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 176. | Lu SH, Yamagata T, Atsuki K, Sun L, Smith CP, Yoshimura N, Chancellor MB, de Groat WC. Effect of KW-7158, a putative afferent nerve inhibitor, on bladder and vesico-vascular reflexes in rats. Brain Res. 2002;946:72-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 177. | Wulff H, Castle NA, Pardo LA. Voltage-gated potassium channels as therapeutic targets. Nat Rev Drug Discov. 2009;8:982-1001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 522] [Cited by in RCA: 617] [Article Influence: 38.6] [Reference Citation Analysis (0)] |
| 178. | Soder RP, Petkov GV. Large conductance Ca2+ -activated K+ channel activation with NS1619 decreases myogenic and neurogenic contractions of rat detrusor smooth muscle. Eur J Pharmacol. 2011;670:252-259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 179. | Rode F, Svalø J, Sheykhzade M, Rønn LC. Functional effects of the KCNQ modulators retigabine and XE991 in the rat urinary bladder. Eur J Pharmacol. 2010;638:121-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 52] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 180. | Streng T, Christoph T, Andersson KE. Urodynamic effects of the K+ channel (KCNQ) opener retigabine in freely moving, conscious rats. J Urol. 2004;172:2054-2058. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 59] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 181. | Foster CD, Fujii K, Kingdon J, Brading AF. The effect of cromakalim on the smooth muscle of the guinea-pig urinary bladder. Br J Pharmacol. 1989;97:281-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 74] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 182. | Andersson KE, Andersson PO, Fovaeus M, Hedlund H, Malmgren A, Sjögren C. Effects of pinacidil on bladder muscle. Drugs. 1988;36 Suppl 7:41-49. [PubMed] |
| 183. | Shieh CC, Brune ME, Buckner SA, Whiteaker KL, Molinari EJ, Milicic IA, Fabiyi AC, Daza A, Brioni JD, Carroll WA. Characterization of a novel ATP-sensitive K+ channel opener, A-251179, on urinary bladder relaxation and cystometric parameters. Br J Pharmacol. 2007;151:467-475. [PubMed] |
| 184. | Gopalakrishnan M, Buckner SA, Whiteaker KL, Shieh CC, Molinari EJ, Milicic I, Daza AV, Davis-Taber R, Scott VE, Sellers D. (-)-(9S)-9-(3-Bromo-4-fluorophenyl)-2,3,5,6,7,9-hexahydrothieno[3,2-b]quinolin-8(4H)-one 1,1-dioxide (A-278637): a novel ATP-sensitive potassium channel opener efficacious in suppressing urinary bladder contractions. I. In vitro characterization. J Pharmacol Exp Ther. 2002;303:379-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 185. | Parajuli SP, Hristov KL, Soder RP, Kellett WF, Petkov GV. NS309 decreases rat detrusor smooth muscle membrane potential and phasic contractions by activating SK3 channels. Br J Pharmacol. 2013;168:1611-1625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 186. | Soder RP, Parajuli SP, Hristov KL, Rovner ES, Petkov GV. SK channel-selective opening by SKA-31 induces hyperpolarization and decreases contractility in human urinary bladder smooth muscle. Am J Physiol Regul Integr Comp Physiol. 2013;304:R155-R163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 187. | Nielsen JS, Rode F, Rahbek M, Andersson KE, Rønn LC, Bouchelouche K, Nordling J, Bouchelouche P. Effect of the SK/IK channel modulator 4,5-dichloro-1,3-diethyl-1,3-dihydro-benzoimidazol-2-one (NS4591) on contractile force in rat, pig and human detrusor smooth muscle. BJU Int. 2011;108:771-777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 188. | Tertyshnikova S, Knox RJ, Plym MJ, Thalody G, Griffin C, Neelands T, Harden DG, Signor L, Weaver D, Myers RA. BL-1249 [(5,6,7,8-tetrahydro-naphthalen-1-yl)-[2-(1H-tetrazol-5-yl)-phenyl]-amine]: a putative potassium channel opener with bladder-relaxant properties. J Pharmacol Exp Ther. 2005;313:250-259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 50] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 189. | Chapple CR, Patroneva A, Raines SR. Effect of an ATP-sensitive potassium channel opener in subjects with overactive bladder: a randomized, double-blind, placebo-controlled study (ZD0947IL/0004). Eur Urol. 2006;49:879-886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 44] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 190. | Available from: http: //clinicaltrials.gov/show/NCT00439192. |
| 191. | Yoshimura N, Seki S, Novakovic SD, Tzoumaka E, Erickson VL, Erickson KA, Chancellor MB, de Groat WC. The involvement of the tetrodotoxin-resistant sodium channel Na(v)1.8 (PN3/SNS) in a rat model of visceral pain. J Neurosci. 2001;21:8690-8696. [PubMed] |
| 192. | Stummann TC, Salvati P, Fariello RG, Faravelli L. The anti-nociceptive agent ralfinamide inhibits tetrodotoxin-resistant and tetrodotoxin-sensitive Na+ currents in dorsal root ganglion neurons. Eur J Pharmacol. 2005;510:197-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 193. | Yamane H, de Groat WC, Sculptoreanu A. Effects of ralfinamide, a Na+ channel blocker, on firing properties of nociceptive dorsal root ganglion neurons of adult rats. Exp Neurol. 2007;208:63-72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 194. | Araki I, Du S, Kobayashi H, Sawada N, Mochizuki T, Zakoji H, Takeda M. Roles of mechanosensitive ion channels in bladder sensory transduction and overactive bladder. Int J Urol. 2008;15:681-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 43] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 195. | Waldmann R, Lazdunski M. H(+)-gated cation channels: neuronal acid sensors in the NaC/DEG family of ion channels. Curr Opin Neurobiol. 1998;8:418-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 397] [Cited by in RCA: 409] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 196. | Olsen SM, Stover JD, Nagatomi J. Examining the role of mechanosensitive ion channels in pressure mechanotransduction in rat bladder urothelial cells. Ann Biomed Eng. 2011;39:688-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 197. | Sánchez-Freire V, Blanchard MG, Burkhard FC, Kessler TM, Kellenberger S, Monastyrskaya K. Acid-sensing channels in human bladder: expression, function and alterations during bladder pain syndrome. J Urol. 2011;186:1509-1516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 198. | Homma Y, Nomiya A, Tagaya M, Oyama T, Takagaki K, Nishimatsu H, Igawa Y. Increased mRNA expression of genes involved in pronociceptive inflammatory reactions in bladder tissue of interstitial cystitis. J Urol. 2013;190:1925-1931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 76] [Article Influence: 6.3] [Reference Citation Analysis (0)] |