Copyright
©The Author(s) 2015.
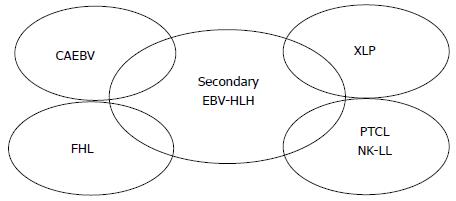
Figure 1 Underlying or other diseases overlapping with Epstein-Barr virus-related hemophagocytic lymphohistiocytosis.
Although the majority of cases of EBV-HLH due to secondary HLH develop without any apparent immunodeficiency, some cases may develop in association with CAEBV (see also Figure 2), XLP (type 1 or type 2), FHL (types 2-5), or EBV-positive peripheral T cell lymphoma, or NK cell leukemia or lymphoma. EBV-HLH: Epstein-Barr virus-related hemophagocytic lymphohistiocytosis; CAEBV: Chronic active EBV infection; XLP: X-linked lymphoproliferative disease; FHL: Familial HLH; NK: Natural killer.
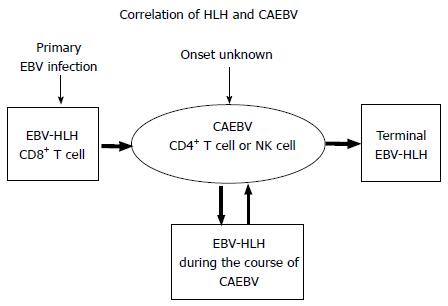
Figure 2 Correlations between hemophagocytic lymphohistiocytosis and chronic active Epstein-Barr virus infection status.
CAEBV status may occur without apparent onset of symptoms or may develop following initial acute onset EBV-HLH. During the course of CAEBV, HLH episodes may develop, and if it is not adequately treated by transplantation, most patients eventually succumb to terminal HLH or to lymphoid malignancies. CD8+ T cells play a major role in initial acute onset HLH, whereas CD4+ T cells or NK cells play a role in the status of CAEBV and in CAEBV-related HLH. CAEBV: Chronic active EBV infection; EBV-HLH: Epstein-Barr virus-related hemophagocytic lymphohistiocytosis; NK: Natural killer.
Figure 3 Treatment regimens for Epstein-Barr virus-related hemophagocytic lymphohistiocytosis.
Prior to commencement of treatment, determination of EBV genome copy numbers (D), EBV serology (anti-VCA-IgM, -IgG,-EADR-IgG and anti-EBNA) (E), flow cytometry (F), and, if possible, EBV tropism in subsets (G) of PB are required. Furthermore, cytogenetics (H) of PB or bone marrow cells are recommended. It is also recommended to determine EBV genome copy numbers (D) following 4, 8, and 24 wk of treatment to observe treatment response, and following 24 wk, 12, 18, and 24 mo of treatment to determine whether the disease progresses to the status of CAEBV. Screening tests for XLP or FHL are ideally required (I) for any cases demonstrating a PR/NR to the A1 and B regimens until 8 wk of treatment. Treatment comprises a window period of 2 wk commencing with PSL (A1; 2 mg/kg per day). Once a GR is attained, PSL with tapering (A2) is administered. Following 5 wk of PSL given, treatment is discontinued in patients who attain a CR. Patients with a PR/NR to A1 and those relapsed with A2 are to commence the B regimen, which comprises a weekly dose of etoposide (100 mg/m2), PSL (2 mg/kg), and CSA (trough levels, 80-150). If EBV is found to reside in B cells, three doses of rituximab (375 mg/m2 per dose) are then added to the B regimen. Patients that do attain a CR becomes off therapy at the end of 8 wk, while who do not attain a CR with B arm treatment are to commence the C regimen, which comprises a once every 2 wk dose of etoposide (100 mg/m2), PSL (2 mg/kg), and CSA (trough levels, 80-150). Patients who relapse after CR with B arm also go to C regimen. If the patient attains a CR following 24 wk of treatment, the total cumulative dose of etoposide is 2200 mg/m2. Patients that remain at PR/NR following a total 24 wk of treatment are to undergo salvage therapy or HSCT. GR: Good response; PR: Poor response; NR: No response; CR: Complete response; HSCT: Hematopoietic stem cell transplantation; PSL: Prednisolone; CSA: Cyclosphorin A.
- Citation: Imashuku S. Treatment of Epstein-Barr virus-related hemophagocytic lymphohistiocytosis: Study protocol of a prospective pilot study. World J Hematol 2015; 4(4): 69-75
- URL: https://www.wjgnet.com/2218-6204/full/v4/i4/69.htm
- DOI: https://dx.doi.org/10.5315/wjh.v4.i4.69