Published online Mar 27, 2016. doi: 10.4240/wjgs.v8.i3.179
Peer-review started: August 11, 2015
First decision: September 22, 2015
Revised: October 24, 2015
Accepted: December 29, 2015
Article in press: January 4, 2016
Published online: March 27, 2016
Processing time: 225 Days and 22.1 Hours
Lymph node metastasis informs prognosis and is a key factor in deciding further management, particularly adjuvant chemotherapy. It is core to all contemporary staging systems, including the widely used tumor node metastasis staging system. Patients with node-negative disease have 5-year survival rates of 70%-80%, implying a significant minority of patients with occult lymph node metastases will succumb to disease recurrence. Enhanced staging techniques may help to identify this subset of patients, who might benefit from further treatment. Obtaining adequate numbers of lymph nodes is essential for accurate staging. Lymph node yields are affected by numerous factors, many inherent to the patient and the tumour, but others related to surgical and histopathological practice. Good lymph node recovery relies on close collaboration between surgeon and pathologist. The optimal extent of surgical resection remains a subject of debate. Extended lymphadenectomy, extra-mesenteric lymph node dissection, high arterial ligation and complete mesocolic excision are amongst the surgical techniques with plausible oncological bases, but which are not supported by the highest levels of evidence. With further development and refinement, intra-operative lymphatic mapping and sentinel lymph node biopsy may provide a guide to the optimum extent of lymphadenectomy, but in its present form, it is beset by false negatives, skip lesions and failures to identify a sentinel node. Once resected, histopathological assessment of the surgical specimen can be improved by thorough dissection techniques, step-sectioning of tissue blocks and immunohistochemistry. More recently, molecular methods have been employed. In this review, we consider the numerous factors that affect lymph node yields, including the impact of the surgical and histopathological techniques. Potential future strategies, including the use of evolving technologies, are also discussed.
Core tip: The number of lymph nodes in surgical resection specimens is influenced by numerous factors. Good practice by surgeons and pathologists is essential to maximize lymph node yields, but there are non-modifiable factors related to patient and tumour. Extended lymphadenectomy, extra-mesenteric lymph node dissection, high arterial ligation and complete mesocolic excision, all increase lymph node yields, but a definite benefit in prognosis is not proven and the optimal extent of surgical resection remains contentious. Conversely, further development in sentinel lymph node biopsy techniques could allow selective lymphadenectomy, whilst providing appropriate information to guide adjuvant therapy.
- Citation: Ong MLH, Schofield JB. Assessment of lymph node involvement in colorectal cancer. World J Gastrointest Surg 2016; 8(3): 179-192
- URL: https://www.wjgnet.com/1948-9366/full/v8/i3/179.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v8.i3.179
Lymph node metastasis (LNM) informs prognosis and is a key factor in deciding further management, particularly adjuvant chemotherapy. As such, lymph node metastasis has had a role in colorectal cancer staging from the earliest classification systems. Its importance in prognosis has been borne out by successive classification systems and is reflected in all contemporary staging systems, in particular the widely used tumor node metastasis (TNM) staging system, developed and maintained by the Union for International Cancer Control and American Joint Committee on Cancer (AJCC).
Patients with node-negative disease have 5-year survival rates of 70%-80% in contrast to 30%-60% in those with node-positive disease. Survival is improved in the latter group by adjuvant chemotherapy. The 20%-30% disease recurrence in apparently completely excised tumours without lymph node metastases is thought to be due to occult lymph node disease. If this subset could be identified by better lymph node staging, they might also benefit from adjuvant chemotherapy.
There are several prognostic factors other than lymph node disease status that also identify patients who might benefit from adjuvant treatment. These include venous invasion, peri-neural invasion, tumour perforation, serosal involvement and incomplete resection[1,2]. However, lymph node assessment remains a mainstay of deciding adjuvant chemotherapy. To achieve accurate staging, surgeons and pathologists must exercise due diligence in their respective practices. Most authorities recommend examination of a minimum of 12 lymph nodes, although the evidence base for this is weak. Behind this apparently simple number are numerous complex issues, many without clear solutions. In this review, we consider the factors that affect lymph node yields including the influence of surgical and histopathological techniques. Evolving concepts and technologies that are not in widespread use, such as sentinel lymph node evaluation, are also discussed.
In order to identify and maximise the diagnostic information from lymph nodes within a specimen, it is important to understand the factors that influence the lymph node harvest (LNH). This relates to a range of different factors: The pathologist, the surgeon and factors inherent to the patient and tumour. While tumour and patient characteristics cannot be changed, the pathologist can employ various techniques to maximise both the LNH and gain additional diagnostic information from enhanced study of the lymph node. The surgeon can modify the surgical procedure to excise more tissue or use ancillary techniques to aid selection and examination of lymph nodes by the pathologist.
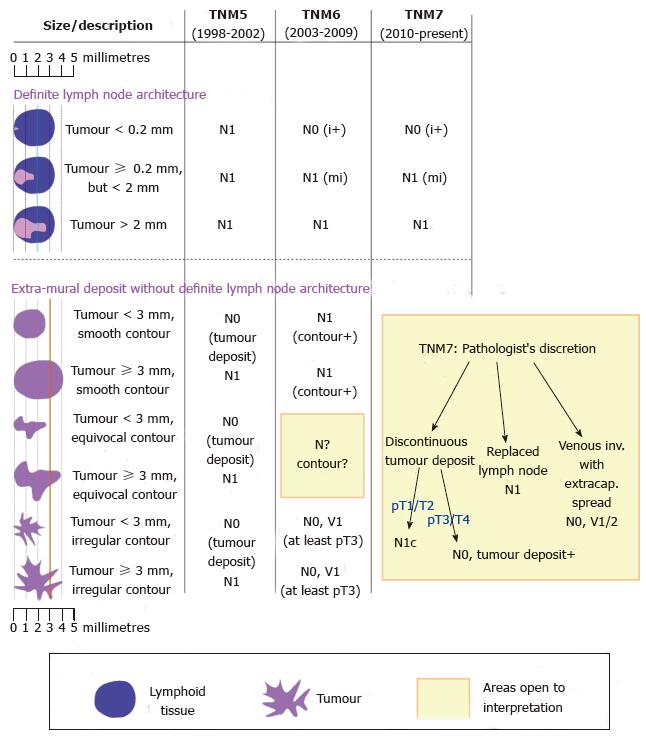
There are several tumour staging systems, of which the TNM staging system is the most widely used internationally. It seems self-evident that lymph node metastasis indicates the presence of tumour cells within a lymph node. However, precise definition of different types of burden is crucial. Metastatic disease is often sub-classified into isolated tumour cells (ITCs, < 0.2 mm), micrometastases (defined as > 0.2 mm but < 2 mm) and macrometastases (≥ 2 mm). More recently, the concept of molecular positivity has been introduced. The classification of nodal disease (N-stage) under the current 7th edition of the TNM staging system (TNM7) is summarised in Table 1.
| N Stage | Description |
| NX | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastasis |
| N0 (i-) | No regional lymph node metastases histologically, negative IHC |
| N0 (i+) | Isolated tumour cells, identified by H&E and/or IHC |
| N0 (mol-) | No regional lymph node metastases histologically, negative molecular findings (RT-PCR) |
| N0 (mol+) | Positive molecular findings (RT-PCR), but no regional lymph node metastases detected by histology or IHC |
| N1mi | Micrometastases |
| N1 | Metastasis in 1-3 regionl lymph nodes |
| N1a | Metastasis in 1 regional lymph node |
| N1b | Metastasis in 2-3 regional lymph nodes |
| N1c | Tumor deposit(s) in the subserosa, mesentery, or nonperitonealized pericolic or perirectal tissues without regional nodal metastasis |
| N2 | Metastasis in four or more regional lymph nodes |
| N2b | N2b Metastasis in seven or more regional lymph nodes |
| N2a | N2a Metastasis in 4-6 regional lymph nodes |
A universally agreed definition of what constitutes lymph node metastasis is important for communication between all parties involved in treating, diagnosing and researching colorectal cancer. It facilitates uniformity for the purposes of entry to clinical trials, subsequent applicability of the ensuing results and interpretation of historical trends. Any criteria should be objective, reproducible, evidence-based and met with broad agreement. However, significant changes to the criteria in successive editions of TNM have been criticised for lacking some of the above qualities.
Detailed analysis of the changes wrought by the two most recent TNM editions is presented elsewhere[3-5]. The main changes are summarised diagrammatically in Figure 1, but a few points warrant discussion. In the 6th edition (TNM6)[6] of the TNM staging system, isolated tumour cells became classed as N0 for the purposes of grouping tumours into AJCC stage I to IV, in contrast to N1 in the 5th edition (TNM5)[7]. Secondly, extra-mural deposits are difficult to classify. In a study of 69 tumour deposits, step sections were performed on what were initially diagnosed as tumour deposits. A significant proportion were found to represent other patterns of tumour spread[8]. The ‘‘3 mm rule’’ stipulated in TNM5 was not based on published data, but had the advantage of being objective and reproducible[9], in contrast to the assessment of ‘‘contour’’ introduced in the 6th edition (TNM6)[10]. The ‘‘contour rule’’ was dropped in the 7th edition (TNM7), but explicit criteria were not provided to replace it. Left to the discretion of the pathologist, classification of extra-mural tumour is fraught with inter-observer variability[11]. Unsurprisingly, there has been stage migration as a result of these changes, making it difficult to compare historical data. Data from the Surveillance, Epidemiology and End Results population-based registries showed that 10% of colorectal cancer cases had ‘‘tumour deposits’’, of which 30%-40% occurred without concomitant lymph node metastases. Compared to TNM6, this represented up-staging of 2.5% of colon and 3.3% of rectal cases to N1c, a significant stage migration from stage I to stage III[12]. There have also been misgivings over the use of TNM7 following neoadjuvant treatment, where patchy tumour regression may give the false appearance of lymph node metastasis or discontinuous tumour deposit. Finally, the changes in definition tend to reduce lymph node counts[13], a concern where LNH is being used as a marker of “quality’’. It is hoped than the 8th of edition, due to published this year, will resolve some of these issues.
In many pathology laboratories, macroscopic examination and dissection of colorectal cancer specimens is delegated to trainee pathologists, sometimes with limited experience and expertise. These large resection specimens tend to be left to the end of the ‘‘cut-up’’ session when time may be limited. Even in experienced hands, the detection of minute lymph nodes in mesenteric fat by palpation and dissection is pain-staking and time-consuming. Marked variation in the assessment of colorectal cancer in the pathology laboratory, particularly in lymph node yields, is not a new issue[14,15], but there is now more awareness of the crucial role of dissection. Results from staff pathologists[16] and non-pathologist dissectors[17-19] may be superior, but it is likely that a major factor is not the expertise of the operator, but rather the time devoted to searching for lymph nodes. de Burlet et al[20] studied LNH in gastrointestinal tumour resection specimens. Twenty minutes was allocated to an initial lymph node search, followed by an extra 5 and 10 min, which increased yields by 12% and 20% respectively. Twenty additional minutes added a mean of 6 lymph nodes, albeit with a diminishing rate of lymph node discovery. The United Kingdom Royal College of Pathologists’ Guidelines on Staffing and Workload allocates 8 points for cutting-up a colorectal resection, corresponding to an anticipated time spent of 31-50 min[21]. This would appear to underestimate the time required for a thorough job if de Burlet et al[20] findings are correct. Often little thought is given to the ergonomics around cut-up. To optimise lymph node yields, we recommend that large specimens should be dealt with first when the operator is still fresh.
Current practice in handling of lymph nodes is not uniform. The United Kingdom Royal College of Pathologist guidance recommends embedding each lymph node whole, if < 4 mm, and a central block through longest axis for larger nodes[22]. It is common practice to bisect or serially slice larger lymph nodes.
Typically, a single haematoxylin and eosin stained section is cut from each lymph node block, representing only a tiny volume of the lymph node in a single axis. Cutting more sections increases detection of lymph node metastases, including up-staging of several cases[23], but the workload implications for the laboratory and histopathologist makes routine application of this unfeasible. Similarly, identification of small deposits of tumour by immunohistochemistry increases detection, but once again, has significant cost and workload implications.
Lymphatic mapping, the process of injecting tracer at the tumour site and following lymphatic flow to identify lymph nodes, has been used to identify sentinel lymph nodes (SLN). These SLNs are then subject to more intensive histopathological scrutiny, so-called ultrastaging[24], typically consisting of additional levels and/or immunohistochemistry and in some cases molecular techniques[25]. The utility of SLN ultrastaging is hampered by the limitations of current SLN procedures, namely false negatives, skip lesions and failure to identify a SLN (see later section on SLN). The significance on prognosis of isolated tumour cells identified in this way is also contentious[25] and is discussed in the later section on the size of tumour deposits.
Several other ancillary techniques have been employed to aid the LNH. Modified lymphatic mapping can be achieved by injection of India ink at the time of surgery[26,27] and, similarly, ex vivo intra-arterial injection of methylene blue can accentuate lymph nodes[28,29]. Chemical fat clearance can be performed with a variety of chemical regimens, typically a mixture of fixatives and organic solvents, such as glacial acetic acid, xylene, acetone, and alcohol. With the fat partially removed, nodes are accentuated, facilitating manual dissection and increasing yields. The clearance techniques are not in universal usage due to the slight delay introduced in finalizing a report and safety issues related to the disposal of the volumes of hazardous chemicals generated. The entire mesentery can be embedded without fat clearance, so-called entire residual mesenteric tissue examination, which also increases yields[30]. There is no doubt that many of these techniques increase LNH, but there are not currently enough data to show that they result in significant up-staging[31].
The disadvantage of conventional ultrastaging is that it still relies on examination of a tiny volume of the lymph node. Lymph nodes harvested fresh can be processed to extract nucleic acids that can be analysed using reverse transcriptase and polymerase-based technologies. Some studies have used conventional polymerase chain reaction, but loop-mediated isothermal amplification, also known as one-step nucleic acid amplification (OSNA) can be performed in less than an hour and can be used intra-operatively. The results are quantitative and should reflect mRNA copy number. Thresholds are set to give grades of molecular lymph node involvement equivalent to conventional nodal staging, typically ≥ 250 copies for micrometastases and ≥ 5000 for macrometastases, although these figures are based on work done with breast cancer cases. Typical markers including carcinoembryonic antigen, cytokeratins 19/20 and guanylyl cyclase C. OSNA can be performed on the entire node[32-34] or half of the node in combination with conventional sections[35]. While up-staging was described in most series, there have been discrepancies not entirely explained by tissue allocation, suggesting conventional methods, albeit with ultrastaging-type protocols may have superior sensitivity and specificity. The data on how OSNA results correlates with the performance of single section histopathological analysis is sparse, particularly when isolated tumour cells are not included as a molecular category. Application of the OSNA technique to all lymph nodes harvested is not currently feasible outside of the research setting and practically-speaking, its main role is likely to be for the purposes of analysing sentinel lymph nodes.
The principle of sentinel lymph node biopsy (SLNB) is well established in melanoma and breast cancer, where the aim is to avoid unnecessary and potentially morbid lymphadenectomy. Unlike these two malignancies, where lymphadenectomy is a separate procedure, lymphadenectomy in elective colorectal cancer surgery is typically performed as part of a single surgical procedure. The lymphadenectomy component carries a low, but not entirely negligible morbidity. In a review of SLNB, Cahill questions the assumption that additional surgery carries no or minimal risk, particularly if radical lymphadenectomy is performed[36]. The effects of excising unnecessary tissue are difficult to quantify. However, if SLNB can readily and reliably determine lymph node status, permitting more conservative surgery, then reduced tissue dissection, shortened operative time and better bowel function are all desirable outcomes.
Another scenario where SLNB may be informative is in early T-stage colorectal cancers, particularly pT1 polyp cancers identified by bowel cancer screening programmes. Adequate local excision of these polyp cancers is often achieved by endoscopic resection, but there is uncertainty about whether segmental resection for lymphadenectomy is indicated, a particular dilemma in patients with significant co-morbidities. While certain tumour characteristics predict lymph node metastases[37-40], a SLNB should provide a definitive answer. SLNB can be performed laparoscopically[41,42] and potentially via other minimally invasive techniques, e.g., a transcolonic approach using with natural orifice transluminal endoscopic surgery[43].
In this context, SLNB data specific to pT1/T2 tumours is of particular relevance, but many studies are small, typically include all T-stages or, in some studies, omit T-stage data. SLNB may have less of a role in pT3/T4 tumours as they are more likely to harbour lymph node metastases and therefore less likely to benefit from initial SLNB[44]. Additionally, an increased rate of false negatives has also been described in pT3/pT4 tumours[36].
Broader adoption of SLNB, however, is limited by the guarded results from existing studies. SLNB is beset by a number of problems: Failure to identify a SLN, false negatives and skip lesions[45-47]. Skip lesions have been hypothesised to be due to blocked lymphatic flow into involved lymph nodes, but this is not entirely explained by some data. It is unclear if the poor results are explained by technical problems, sub-optimal implementation of the technique or whether the concept is fundamentally flawed because of the inherently unpredictable pattern of lymph node involvement[36,48]. Further evaluation of these techniques is required to determine whether they should be generally adopted.
The most obvious measurable parameter relating to lymph nodes is the total LNH. Sampling as many lymph nodes as possible is ideal, but the focus on absolute counts alone ignores the complex and sometimes interacting factors that influence LNH. A detailed analysis of lymph node counts is presented later in this review, but other characteristics related to lymph nodes are discussed here.
There are two separate aspects to consider. Firstly, does lymph node size, irrespective of tumour involvement, have implications on LNH or prognosis? Secondly, if a lymph node is involved, is the size of the deposit within the lymph node significant?
Chirieac et al[49] showed that nodal size significantly predicted overall survival in patients with node-negative colorectal cancer. They also speculated that high numbers of bulky negative lymph nodes were a product of an active host immune response, which ultimately contributed to improved patient prognosis and survival.
In some studies, LNM were more likely to be found in larger lymph nodes[50,51], perhaps because they are easily palpable and therefore preferentially sampled. In node-positive disease, the size of the lymph node (as opposed to the tumour deposit) appears to have no significance on outcome[52,53]. These studies and several others have demonstrated that many, if not the majority, of LNM occur in lymph nodes < 5 mm[51,54]. The relevance of this is that small LNs are harder or impossible to palpate and are therefore less likely to be sampled during pathological dissection. Secondly, it is hard to completely separate the size of the tumour deposit from the size of the lymph node as the deposit obviously cannot exceed the size of the node. According to TNM7 rules, a positive 1.9 mm lymph node will either be involved by isolated tumour cells or micrometastases, but never a macrometastases (Figure 1).
This leads to the next question: Is the size of LN tumour deposit significant? The size of the largest lymph node tumour deposit appears to be prognostic[55], but the overall volume of lymph node tumour burden appears to be less important than the number of involved lymph nodes[56].
There is also considerable debate about the significance of isolated tumour cells. The data shows a wide variation in the incidence of isolated tumour cells and micrometastases, ranging from 11% to 59%. Some demonstrate an adverse effect on survival[57-60], but others show no significance[61-64]. The discrepancy reflects the differences in study design such as method of detection, length of follow-up and whether other confounding factors were considered. As previously discussed, more thorough scrutiny of lymph nodes with ultrastaging and/or molecular methods may increase detection of tumour, but it is unclear what significance this has on prognosis as direct comparison of data difficult. A 2014 meta-analysis suggests micrometastases have an adverse prognosis whilst isolated tumour cells do not[65], but this distinction is not always straight-forward: The size cut-off of 0.2 mm is arbitrary and other definitions in terms of total cell numbers are hard to apply consistently.
Oncological practice in the United Kingdom continues to use the TNM5 definitions of lymph node metastasis, which defines the presence of any metastatic disease as N-positive, warranting adjuvant chemotherapy. Practice in other parts of the world differs, particularly in countries that have already adopted TNM7, which classifies isolated tumour cells as N0.
Extracapsular extension is typically associated with more aggressive and infiltrative tumours. Heide et al[66] noted that extracapsular extension in rectal resections was connected with adverse local control and a higher rate of distant metastases. In another study, the survival rates and disease-free survival rates for patients with metastatic lymph nodes showing an extracapsular invasion pattern were significantly worse than cases showing no evidence of extracapsular extension[67].
LNs negative for tumour may show reactive patterns such as follicular, parafollicular hyperplasia, as well as sinus histiocytosis. These have been regarded as indicators of active host immune response and are associated with an improved prognosis and 5-year survival rate[68]. A survival advantage has also been established in metastatic lymph nodes that also demonstrate a background of benign reactive inflammatory changes[69]. The host-response hypothesis may also explain why patients with lower lymph node yields are generally found to have a poorer prognosis, although reactive lymph nodes are more easily identified and may result in higher LNH and more accurate staging.
The use of the ratio of positive nodes to total LNH was first proposed by Berger et al[70] in 2005 as an additional prognostic factor. Several subsequent studies have corroborated the original findings[71-77], although what threshold to use is not clear. The results are not entirely consistent and there may also be differences between colonic and rectal tumour[78]. A minimum LNH is required to make the ratio valid. Conversely, large numbers of lymph nodes obtained through techniques such as fat clearing may increase the overall denominator, disproportionately reducing the ratio.
LNM from tumours showing prominent mucinous differentiation may manifest as pauci-cellular mucin pools. Following neoadjuvant treatment, these may be rendered acellular. Further step levels are helpful to exclude viable tumour cells, but if no cells are found, these are regarded by most pathologists as lymph node negative[79,80].
The role of the surgeon is to excise the primary tumour and an appropriate amount of mesenteric tissue with clear margins, to allow adequate staging, whilst minimising potential complications. This raises the question of how much tissue should be removed to achieve optimal oncological outcomes.
How much tissue to remove is guided by the interpretation of the fundamental purpose of lymphadenectomy. There are divergent views on whether it is directly therapeutic or whether it provides mainly staging and prognostication[81]. The model espoused by Halsted at the end of the 19th century assumes sequential and step-wise spread of tumour outwards from the primary site. Radical surgery to remove all tumour not only provides staging information, but also potentially cures the tumour. In contrast, the Cady et al[82] paradigm assumes systemic spread may occur early in tumour growth and that improved outcomes derive from delivery of the optimum adjuvant treatment as determined by accurate staging. The Halsted radical mastectomy has been consigned to surgical history, but it is unclear if principles gleaned from breast cancer can be extrapolated to colorectal cancer.
Indirect evidence for a therapeutic effect has been inferred from studies looking at lymph node counts. The Intergroup Trial INT-0089 showed 5-year overall survival increased from 51% to 71% for N2 disease if > 35 lymph nodes were harvested compared to < 35[83]. Given this was N2 disease, better staging and stage migration cannot entirely explain the results which showed superior survival to that of published trials using optimal adjuvant chemotherapy, implying a curative component. Other explanations are possible, e.g., high lymph node counts representing good host inflammatory response, but it is likely that lymphadenectomy is both prognostic and therapeutic, particularly in the rectum where total mesorectal excision (TME) achieves simultaneous local control and lymphadenectomy, with both components inherently inseparable. It is no surprise that when more mesenteric tissue is removed, LNH also increases. In theory, this leads to more accurate staging and potential therapeutic removal of involved lymph nodes. However, for many of the surgical techniques described below, the highest levels of evidence are lacking. It is therefore unclear whether the benefits of removing more tissue outweigh the increased operating time and potential morbidity associated with these procedures. A detailed review of surgical practice is beyond the scope of this review, but salient issues are considered below and readers are directed to other surgical guidelines[84-87].
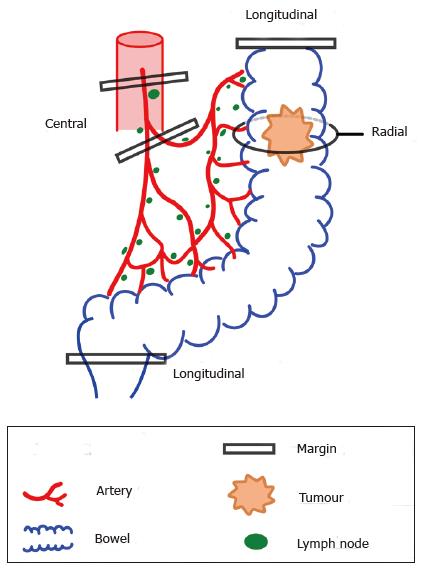
Margins can be thought of as extending to 3 anatomical boundaries (Figure 2). Firstly, the longitudinal margin as determined by the axial extent of the bowel excised. Secondly, the extent of mesenteric tissue excised, in a centripetal direction towards the root of the supplying artery. Thirdly, radial margins, in the broadest sense, which may include en bloc excision of advanced local spread, e.g., the abdominal wall or adjacent organs, but also encompasses the circumferential margin, or more accurately, the non-peritonealised margin. Frequently, these three margins cannot be manipulated independently of each other, but as a general rule, increasing the first two margins also increases lymph node yields.
Typically, the segment of bowel containing the tumour is excised along with the mesentery delineated by its arterial supply. For colonic tumours, at least 5 cm of longitudinal clearance is advised to minimise anastomotic recurrence[88,89]. In the rectum, 5 cm proximal and 2 cm distal appears sufficient[90]. The Japanese Society for Cancer of the Colon and Rectum guidelines recommend at least 5 cm in the direction of lymph flow and 10 cm in opposite direction[91]. In practice, it is the vascular supply that dictates the extent of surgery. If the tumour straddles two arterial branches, both segments should be excised. Anatomical and functional considerations may also extend resection beyond oncological requirements. For instance, in left-sided tumours, many surgeons avoid anastomoses with the sigmoid colon as it is regarded as a “high pressure” segment and also receives no contribution from the marginal artery.
The length of bowel resected may be extended in several scenarios on the basis that spread of tumour beyond normal segmental boundaries has been described, a finding partially borne out by intra-operative lymphatic mapping. Extended lymphadenectomy can be achieved by performing an extended right hemicolectomy for proximal right-sided tumours[92,93]. Extended right hemicolectomy is also common performed for transverse colon and splenic flexure tumours, although there are no randomised, controlled trials to support this. Similarly, for left-sided tumours, one of the few randomised controlled trials in this area showed no benefit of left hemicolectomy over segmental resection[94]. The type of surgery employed, particularly the length of specimen, has a clear influence on LNH, but without lymphatic mapping, is not clear when extended surgery should be performed.
Classically, colonic tumour spreads along lymphatics in the distribution of the arterial supply[92,95,96]. Depending on their anatomical distribution, lymph nodes in the colon are described as pericolic, intermediate and apical/central/main, broadly corresponding to D1, D2 and D3 in the Japanese notation[87]. Lymphadenectomy can be performed up to and flush with the level of the origin of the artery[97], so-called complete vascular ligation, one of the key components of complete mesocolic excision (see below). This manoeuvre takes the apical node which is involved in about 3%-11% of tumours[93,97,98]. In tumours of the sigmoid colon and upper rectum, high ligation of the inferior mesenteric artery has been advocated as oncologically superior. This was first promulgated by Moynihan in 1908[99] and the debate on its value has continued for more than a century. Despite good results in several, mainly cohort studies, other studies have shown no benefit (see systematic reviews in[100-103]). No benefit was seen in sigmoid tumours in a multicentre randomised controlled trial[94]. The issue, however, has not entirely been laid to rest and a randomised controlled trial of high ligation in the context of laparoscopic surgery is on-going[104].
Routine excision or sampling of lymph nodes outside the typical lymph node basin has also been advocated. Tumours around the hepatic flexure may spread to infra-pyloric nodes[97]. In the rectosigmoid region, the arterial supply is variable and spread to lateral (extra-mesorectal) pelvic lymph nodes may occur[96]. One paper describes lateral pelvic node involvement in up to 18%, rising to 36% in the sub-group of Dukes’ C tumours[105]. Proponents of radical lymphadenectomy argue it is oncologically superior, both in achieving better staging but also therapeutically by removing all diseased lymph nodes. However, all of the above additions and modifications to “standard’’ lymphadenectomy may result in additional morbidity, particularly damage to neighbouring structures.
Depending on the anatomical site, this includes the duodenum, ureters and nerve plexuses[106]. Vascular compromise may occur from direct vascular damage or via reduction in collateral flow[107]. As extra-mesenteric and apical lymph node involvement is present only in a minority of cases, routine extended dissection represents unnecessary surgery for most patients. A selective approach has been advocated[98], but patients with the highest rates of aberrant lymph node involvement are those with high T-stage, the same group where lymphadenectomy is least likely to be curative due to the increased risk of systemic disease. The benefit of these procedures is unproven and potential morbidity may outweigh the benefits[108,109].
TME has been established as the optimal surgical technique for rectal tumours. Pioneered by Heald, introduction of the technique reduces local recurrence[110,111]. The same anatomical and oncological principles have been extrapolated to colonic tumours, so-called complete mesocolic excision (CME)[112]. Although a relatively new concept in the West, CME shares many features with D3 excisions that have been performed routinely in East Asia[113,114]. It is associated with better LNHs[115]. However, while it is supported by some compelling oncological and anatomical concepts, it encompasses many of the unproven surgical elements discussed above. The technique may prove itself in the fullness of time, but there is presently insufficient evidence to support it[116,117]. Furthermore, the unsuccessful attempts by European surgeons to adopt D3 lymphadenectomy for gastric cancer is a salutary reminder of how challenging it is to “import” purportedly superior surgical techniques from established centres[118].
Laparoscopic and laparoscopic-assisted surgery is increasingly the default surgical approach to colorectal cancer resection. Superior peri-operative recovery and oncological equivalence has been demonstrated by several randomised controlled trials, including no significant difference in lymph node counts[119]. Many of techniques described above can be achieved laparoscopically, e.g., CME[120-126], although randomised controlled trials are difficult to undertake. Laparoscopic CME therefore still lacks a convincing body of supportive evidence. The data on robotic surgery are promising[127], but at present only includes a single randomised-controlled trial.
A number of procedures can be performed intra-operatively to assist in lymph node staging. As previously discussed, lymphatic mapping entails injecting a tracer at the tumour site, which travels along lymphatics and facilitates identification of lymph nodes[24], including the sentinel lymph node. SLNs can be excised intra-operatively and for immediate results, can be subject to frozen section histological examination or OSNA[128]. Other technologies that provide immediate intra-operative results are the subject of on-going research, e.g., optical coherence tomography and real time elastography[129].
Outside these techniques, the default histological analysis is performed on sections cut after formalin-fixation and paraffin embedding of the SLN. The results are therefore not available to influence immediate operative management. The exception is where the lymphatic mapping process identifies tracer in “aberrant” lymph node territory. The surgeon can choose to sample the abnormal lymph nodes or perform more radical lymphadenectomy. In 2 studies, in vivo lymphatic mapping changed the procedure in 9% and 22% of cases respectively[129,130]. In the latter study, nodal positivity was higher in patients undergoing a change of procedure.
Several patient characteristics have been identified that influence LNHs[131]. However, factors identified in some studies are not corroborated by others. Fewer lymph nodes are generally obtained from specimens from older patients[132-134]. Gender seems to have no effect, while low counts have an inconsistent association with obesity, as measured by body mass index[135,136].
Several histological characteristics of the primary tumour have been shown to be associated with an increased risk of LNM. One meta-analysis identified 42 different factors. Only 15 were reported in 2 or more studies and not all are routinely analysed during standard reporting procedures[137]. Factors that are easily assessed during routine histological reporting include tumour site[133,134,138], stage[133] and differentiation. Higher counts are seen in tumours with microsatellite instability[139]. While not an exhaustive list, many of these features lack reproducibility, sensitivity and/or specificity. It is therefore uncertain whether any single feature in isolation is reliable enough to influence decisions on adjuvant treatment. Neoadjuvant chemotherapy and/or radiotherapy typically reduces the numbers of nodes sampled[133,140].
Predictive factors in submucosal (pT1) tumours are of particular interest as these are typically resected endoscopically and may require segmental resection for lymphadenectomy. Adverse factors in this group include poor tumour differentiation, depth of invasion and lymphovascular invasion[37-40]. If sentinel lymph node biopsy techniques can be refined, this would greatly aid decision-making in this group on whether additional surgery is appropriate.
Many of the surgical and histopathological techniques discussed are based on the presumption that increased lymph node yields invariably leads to more accurate stage. This assumption warrants critical appraisal. Many organizations such as the American Society of Clinical Oncology, the National Comprehensive Cancer Network and the United Kingdom Royal College of Pathologists have guidance stipulating a minimum lymph node yield of 12 lymph nodes per case. The choice of 12 was proposed in 1990 by the Working Party Report to the World Congress of Gastroenterology in Sydney[141], partly supported by subsequent studies, but has a poor evidence base.
Others have suggested alternative minimum numbers depending on the T-stage of tumour[142], with more numbers required for low T-stage disease. While it has been clearly demonstrated in numerous studies that prognosis improves with the number of lymph nodes sampled[143-146], this association does not prove causation. Furthermore, the association of lymph node counts and survival applies even in node-negative disease[147] which lends support to the alternative explanation that high lymph node yields are a surrogate marker of a vigorous host immune response to tumour. Conversely, low lymph node counts are associated with a worse survival and may, in itself, be an indication for adjuvant treatment. Much of the existing evidence is based on studies where LNs were harvested without recourse to special techniques and it is unclear if the same survival associations apply when additional or special techniques “inflate” the number of lymph nodes sampled. Not all studies have demonstrated a beneficial effect of higher lymph node yields[148]. Yet others have observed a trend of increased lymph node yields over several years, most likely reflecting better surgical and histopathological practice, but without a corresponding increase in the detection rate of LNM[149,150]. Similarly, the use of special techniques fares no better[151]. At the risk of repetition, we need to clearly distinguish the principle of association from causality. Increased lymph node yields show an association with survival, but do not cause it. Various techniques may increase the lymph node count, but may not change the underlying nature of the disease. It is established that lymph node yields are multifactorial, influenced by a combination of patient, tumour, surgical and pathological factors[131,152].
Clearly, there must be minimum standards in both surgical and histopathological practice. Surgery that fails to remove enough mesentery for staging and a cursory, hurried dissection by a pathologist, sampling only a handful of lymph nodes are likely, in combination, to lead to under-staging. However, for the majority of practitioners, the message about the importance of achieving accurate lymph node staging has been heard and implemented. Audit of LNHs is good practice, but the unthinking pursuit of ever higher lymph node yields should be resisted. In particular, it is unreasonable to link lymph node yields with quality payments, particularly when it is established that many factors influencing lymph node yields are outside the control of both surgeon and pathologist.
The importance of colorectal cancer lymph node staging cannot be over-emphasised. We have discussed many of the controversies associated with this challenging area and provided guidance about the rational application of additional techniques. TNM7 has not been universally adopted internationally[22], but publication of TNM8 is anticipated in this year. The authors anticipate that this will address some of the issues and lead to a consensus approach. The variable contribution of surgical, pathological, patient and tumour related factors means that this remains a contentious subject. This complex area continues to evolve with new developments, surgically and pathologically, providing novel methods to evaluate nodal disease.
P- Reviewer: Maurel J S- Editor: Qiu S L- Editor: A E- Editor: Wu HL
| 1. | Fang SH, Efron JE, Berho ME, Wexner SD. Dilemma of stage II colon cancer and decision making for adjuvant chemotherapy. J Am Coll Surg. 2014;219:1056-1069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 2. | Dienstmann R, Salazar R, Tabernero J. Personalizing colon cancer adjuvant therapy: selecting optimal treatments for individual patients. J Clin Oncol. 2015;33:1787-1796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 271] [Article Influence: 27.1] [Reference Citation Analysis (1)] |
| 3. | Nagtegaal ID, Quirke P. Colorectal tumour deposits in the mesorectum and pericolon; a critical review. Histopathology. 2007;51:141-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 128] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 4. | Quirke P, Williams GT, Ectors N, Ensari A, Piard F, Nagtegaal I. The future of the TNM staging system in colorectal cancer: time for a debate? Lancet Oncol. 2007;8:651-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 119] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 5. | Nagtegaal ID, Quirke P, Schmoll HJ. Has the new TNM classification for colorectal cancer improved care? Nat Rev Clin Oncol. 2012;9:119-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 154] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 6. | Sobin L, Wittekind C. TNM classification of malignant tumours. 6th ed. Hoboken: Wiley-Blackwell 2002; . |
| 7. | Sobin L, Wittekind C. TNM classification of malignant tumours. 5th ed. Hoboken: Wiley-Blackwell 1997; . |
| 8. | Wünsch K, Müller J, Jähnig H, Herrmann RA, Arnholdt HM, Märkl B. Shape is not associated with the origin of pericolonic tumor deposits. Am J Clin Pathol. 2010;133:388-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 46] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 9. | Nagtegaal ID, Tot T, Jayne DG, McShane P, Nihlberg A, Marshall HC, Påhlman L, Brown JM, Guillou PJ, Quirke P. Lymph nodes, tumor deposits, and TNM: are we getting better? J Clin Oncol. 2011;29:2487-2492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 111] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 10. | Howarth SM, Morgan MJ, Williams GT. The new (6th edition) TNM classification of colorectal cancer a stage too far (Abstract 081). Proceedings of the British Society of Gastroenterology Annual Meeting;. Hoboken: Wiley-Blackwell 2004; Glasgow, 2004. |
| 11. | Rock JB, Washington MK, Adsay NV, Greenson JK, Montgomery EA, Robert ME, Yantiss RK, Lehman AM, Frankel WL. Debating deposits: an interobserver variability study of lymph nodes and pericolonic tumor deposits in colonic adenocarcinoma. Arch Pathol Lab Med. 2014;138:636-642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 52] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 12. | Chen VW, Hsieh MC, Charlton ME, Ruiz BA, Karlitz J, Altekruse SF, Ries LA, Jessup JM. Analysis of stage and clinical/prognostic factors for colon and rectal cancer from SEER registries: AJCC and collaborative stage data collection system. Cancer. 2014;120 Suppl 23:3793-3806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 13. | Jin M, Roth R, Rock JB, Washington MK, Lehman A, Frankel WL. The impact of tumor deposits on colonic adenocarcinoma AJCC TNM staging and outcome. Am J Surg Pathol. 2015;39:109-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 63] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 14. | Blenkinsopp WK, Stewart-Brown S, Blesovsky L, Kearney G, Fielding LP. Histopathology reporting in large bowel cancer. J Clin Pathol. 1981;34:509-513. [PubMed] |
| 15. | Evans MD, Barton K, Rees A, Stamatakis JD, Karandikar SS. The impact of surgeon and pathologist on lymph node retrieval in colorectal cancer and its impact on survival for patients with Dukes’ stage B disease. Colorectal Dis. 2008;10:157-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 40] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 16. | Johnson PM, Malatjalian D, Porter GA. Adequacy of nodal harvest in colorectal cancer: a consecutive cohort study. J Gastrointest Surg. 2002;6:883-888; discussion 889-890. [PubMed] |
| 17. | Shaw A, Collins EE, Fakis A, Patel P, Semeraro D, Lund JN. Colorectal surgeons and biomedical scientists improve lymph node harvest in colorectal cancer. Tech Coloproctol. 2008;12:295-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 18. | Reese JA, Hall C, Bowles K, Moesinger RC. Colorectal surgical specimen lymph node harvest: improvement of lymph node yield with a pathology assistant. J Gastrointest Surg. 2009;13:1459-1463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 19. | Kuijpers CC, van Slooten HJ, Schreurs WH, Moormann GR, Abtahi MA, Slappendel A, Cliteur V, van Diest PJ, Jiwa NM. Better retrieval of lymph nodes in colorectal resection specimens by pathologists’ assistants. J Clin Pathol. 2013;66:18-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 27] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 20. | de Burlet KJ, van den Hout MF, Putter H, Smit VT, Hartgrink HH. Total number of lymph nodes in oncologic resections, is there more to be found? J Gastrointest Surg. 2015;19:943-948. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 21. | Thorpe A, Al-Jafari M, Allen D, Carr R, Helliwell T, Sanders S. Guidelines on staffing and workload for Histopathology and cytopathology departments. Royal College of Pathologists;. Hoboken: Wiley-Blackwell 2012; . |
| 22. | Loughrey MB, Quirke P, Shepherd NA. Dataset for colorectal cancer histopathology reports. The Royal College of Pathologists;. Hoboken: Wiley-Blackwell 2014; . |
| 23. | Verrill C, Carr NJ, Wilkinson-Smith E, Seel EH. Histopathological assessment of lymph nodes in colorectal carcinoma: does triple levelling detect significantly more metastases? J Clin Pathol. 2004;57:1165-1167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 24. | Hirche C, Mohr Z, Kneif S, Doniga S, Murawa D, Strik M, Hünerbein M. Ultrastaging of colon cancer by sentinel node biopsy using fluorescence navigation with indocyanine green. Int J Colorectal Dis. 2012;27:319-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 91] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 25. | Esser S, Reilly WT, Riley LB, Eyvazzadeh C, Arcona S. The role of sentinel lymph node mapping in staging of colon and rectal cancer. Dis Colon Rectum. 2001;44:850-854; discussion 854-856. [PubMed] |
| 26. | Cahill RA, Lindsey I, Cunningham C. Sentinel node mapping by colonic tattooing. Surg Endosc. 2010;24:2365-2366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 27. | Kang J, Park HS, Kim IK, Song Y, Baik SH, Sohn SK, Lee KY. Effect of preoperative colonoscopic tattooing on lymph node harvest in T1 colorectal cancer. Int J Colorectal Dis. 2015;30:1349-1355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 28. | Märkl B, Arnholdt HM, Jähnig H, Spatz H, Anthuber M, Oruzio DV, Kerwel TG. A new concept for the role of ex vivo sentinel lymph nodes in node-negative colorectal cancer. Ann Surg Oncol. 2010;17:2647-2655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 29. | Borowski DW, Banky B, Banerjee AK, Agarwal AK, Tabaqchali MA, Garg DK, Hobday C, Hegab M, Gill TS. Intra-arterial methylene blue injection into ex vivo colorectal cancer specimens improves lymph node staging accuracy: a randomized controlled trial. Colorectal Dis. 2014;16:681-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 30. | Brown HG, Luckasevic TM, Medich DS, Celebrezze JP, Jones SM. Efficacy of manual dissection of lymph nodes in colon cancer resections. Mod Pathol. 2004;17:402-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 73] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 31. | Abbassi-Ghadi N, Boshier PR, Goldin R, Hanna GB. Techniques to increase lymph node harvest from gastrointestinal cancer specimens: a systematic review and meta-analysis. Histopathology. 2012;61:531-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 32. | Chen G, McIver CM, Texler M, Lloyd JM, Rieger N, Hewett PJ, Sen Wan D, Hardingham JE. Detection of occult metastasis in lymph nodes from colorectal cancer patients: a multiple-marker reverse transcriptase-polymerase chain reaction study. Dis Colon Rectum. 2004;47:679-686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 33. | Güller U, Zettl A, Worni M, Langer I, Cabalzar-Wondberg D, Viehl CT, Demartines N, Zuber M. Molecular investigation of lymph nodes in colon cancer patients using one-step nucleic acid amplification (OSNA): a new road to better staging? Cancer. 2012;118:6039-6045. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 57] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 34. | Croner RS, Geppert CI, Bader FG, Nitsche U, Späth C, Rosenberg R, Zettl A, Matias-Guiu X, Tarragona J, Güller U. Molecular staging of lymph node-negative colon carcinomas by one-step nucleic acid amplification (OSNA) results in upstaging of a quarter of patients in a prospective, European, multicentre study. Br J Cancer. 2014;110:2544-2550. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 35. | Vogelaar FJ, Reimers MS, van der Linden RL, van der Linden JC, Smit VT, Lips DJ, van de Velde CJ, Bosscha K. The diagnostic value of one-step nucleic acid amplification (OSNA) for sentinel lymph nodes in colon cancer patients. Ann Surg Oncol. 2014;21:3924-3930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 36. | Cahill RA. What’s wrong with sentinel node mapping in colon cancer? World J Gastroenterol. 2007;13:6291-6294. [PubMed] |
| 37. | Tateishi Y, Nakanishi Y, Taniguchi H, Shimoda T, Umemura S. Pathological prognostic factors predicting lymph node metastasis in submucosal invasive (T1) colorectal carcinoma. Mod Pathol. 2010;23:1068-1072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 143] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 38. | Mou S, Soetikno R, Shimoda T, Rouse R, Kaltenbach T. Pathologic predictive factors for lymph node metastasis in submucosal invasive (T1) colorectal cancer: a systematic review and meta-analysis. Surg Endosc. 2013;27:2692-2703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 82] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 39. | Toh EW, Brown P, Morris E, Botterill I, Quirke P. Area of submucosal invasion and width of invasion predicts lymph node metastasis in pT1 colorectal cancers. Dis Colon Rectum. 2015;58:393-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 55] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 40. | Wada H, Shiozawa M, Katayama K, Okamoto N, Miyagi Y, Rino Y, Masuda M, Akaike M. Systematic review and meta-analysis of histopathological predictive factors for lymph node metastasis in T1 colorectal cancer. J Gastroenterol. 2015;50:727-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 70] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 41. | Kitagawa Y, Ohgami M, Fujii H, Mukai M, Kubota T, Ando N, Watanabe M, Otani Y, Ozawa S, Hasegawa H. Laparoscopic detection of sentinel lymph nodes in gastrointestinal cancer: a novel and minimally invasive approach. Ann Surg Oncol. 2001;8:86S-89S. [PubMed] |
| 42. | Currie A, Brigic A, Thomas-Gibson S, Suzuki N, Faiz O, Kennedy RH. Technical considerations in laparoscopic near-infrared sentinel lymph node mapping in early colonic neoplasia--a video vignette. Colorectal Dis. 2015;17:454-455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 43. | Cahill RA. Regional nodal staging for early stage colon cancer in the era of endoscopic resection and N.O.T.E.S. Surg Oncol. 2009;18:169-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 44. | Cahill RA, Leroy J, Marescaux J. Could lymphatic mapping and sentinel node biopsy provide oncological providence for local resectional techniques for colon cancer? A review of the literature. BMC Surg. 2008;8:17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 36] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 45. | Merrie AE, van Rij AM, Phillips LV, Rossaak JI, Yun K, Mccall JL. Diagnostic use of the sentinel node in colon cancer. Dis Colon Rectum. 2001;44:410-417. [PubMed] |
| 46. | Merrie AE, Phillips LV, Yun K, McCall JL. Skip metastases in colon cancer: assessment by lymph node mapping using molecular detection. Surgery. 2001;129:684-691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 72] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 47. | Bembenek AE, Rosenberg R, Wagler E, Gretschel S, Sendler A, Siewert JR, Nährig J, Witzigmann H, Hauss J, Knorr C. Sentinel lymph node biopsy in colon cancer: a prospective multicenter trial. Ann Surg. 2007;245:858-863. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 102] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 48. | Stojadinovic A, Allen PJ, Protic M, Potter JF, Shriver CD, Nelson JM, Peoples GE. Colon sentinel lymph node mapping: practical surgical applications. J Am Coll Surg. 2005;201:297-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 49. | Chirieac L, Suehiro Y, Niemisto A, Shmulevich I, Lunagomez S, Morris J, Hamilton S. Size and number of negative lymph nodes impact outcome in patients with node-negative stage II colorectal cancer. Mod Pathol. 2005;1 Suppl:100A (abstract 453). [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 50. | Mönig SP, Baldus SE, Zirbes TK, Schröder W, Lindemann DG, Dienes HP, Hölscher AH. Lymph node size and metastatic infiltration in colon cancer. Ann Surg Oncol. 1999;6:579-581. [PubMed] |
| 51. | Cserni G. The influence of nodal size on the staging of colorectal carcinomas. J Clin Pathol. 2002;55:386-390. [PubMed] |
| 52. | Rodriguez-Bigas MA, Maamoun S, Weber TK, Penetrante RB, Blumenson LE, Petrelli NJ. Clinical significance of colorectal cancer: metastases in lymph nodes & lt; 5 mm in size. Ann Surg Oncol. 1996;3:124-130. [PubMed] |
| 53. | Bjelovic M, Kalezic V, Petrovic M, Pesko P, Usaj SK, Marinkovic J, Radovanovic N. Correlation of macroscopic and histological characteristics in the regional lymph nodes of patients with rectal and sigmoidal adenocarcinoma. Hepatogastroenterology. 1998;45:433-438. [PubMed] |
| 54. | Kotanagi H, Fukuoka T, Shibata Y, Yoshioka T, Aizawa O, Saito Y, Tur GE, Koyama K. The size of regional lymph nodes does not correlate with the presence or absence of metastasis in lymph nodes in rectal cancer. J Surg Oncol. 1993;54:252-254. [PubMed] |
| 55. | Dhar DK, Yoshimura H, Kinukawa N, Maruyama R, Tachibana M, Kohno H, Kubota H, Nagasue N. Metastatic lymph node size and colorectal cancer prognosis. J Am Coll Surg. 2005;200:20-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 56. | Wong JH, Steinemann S, Tom P, Morita S, Tauchi-Nishi P. Volume of lymphatic metastases does not independently influence prognosis in colorectal cancer. J Clin Oncol. 2002;20:1506-1511. [PubMed] |
| 57. | Greenson JK, Isenhart CE, Rice R, Mojzisik C, Houchens D, Martin EW. Identification of occult micrometastases in pericolic lymph nodes of Duke’s B colorectal cancer patients using monoclonal antibodies against cytokeratin and CC49. Correlation with long-term survival. Cancer. 1994;73:563-569. [PubMed] |
| 58. | Sasaki M, Watanabe H, Jass JR, Ajioka Y, Kobayashi M, Matsuda K, Hatakeyama K. Occult lymph node metastases detected by cytokeratin immunohistochemistry predict recurrence in “node-negative” colorectal cancer. J Gastroenterol. 1997;32:758-764. [PubMed] |
| 59. | Clarke G, Ryan E, O’Keane JC, Crowe J, MacMathuna P. The detection of cytokeratins in lymph nodes of Duke’s B colorectal cancer subjects predicts a poor outcome. Eur J Gastroenterol Hepatol. 2000;12:549-552. [PubMed] |
| 60. | Mescoli C, Albertoni L, Pucciarelli S, Giacomelli L, Russo VM, Fassan M, Nitti D, Rugge M. Isolated tumor cells in regional lymph nodes as relapse predictors in stage I and II colorectal cancer. J Clin Oncol. 2012;30:965-971. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 42] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 61. | Cutait R, Alves VA, Lopes LC, Cutait DE, Borges JL, Singer J, da Silva JH, Goffi FS. Restaging of colorectal cancer based on the identification of lymph node micrometastases through immunoperoxidase staining of CEA and cytokeratins. Dis Colon Rectum. 1991;34:917-920. [PubMed] |
| 62. | Oberg A, Stenling R, Tavelin B, Lindmark G. Are lymph node micrometastases of any clinical significance in Dukes Stages A and B colorectal cancer? Dis Colon Rectum. 1998;41:1244-1249. [PubMed] |
| 63. | Lee MR, Hong CW, Yoon SN, Lim SB, Park KJ, Lee MJ, Kim WH, Park JG. Isolated tumor cells in lymph nodes are not a prognostic marker for patients with stage I and stage II colorectal cancer. J Surg Oncol. 2006;93:13-18; discussion 18-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 64. | Davies M, Arumugam PJ, Shah VI, Watkins A, Roger Morgan A, Carr ND, Beynon J. The clinical significance of lymph node micrometastasis in stage I and stage II colorectal cancer. Clin Transl Oncol. 2008;10:175-179. [PubMed] |
| 65. | Sloothaak DA, Sahami S, van der Zaag-Loonen HJ, van der Zaag ES, Tanis PJ, Bemelman WA, Buskens CJ. The prognostic value of micrometastases and isolated tumour cells in histologically negative lymph nodes of patients with colorectal cancer: a systematic review and meta-analysis. Eur J Surg Oncol. 2014;40:263-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 66. | Heide J, Krüll A, Berger J. Extracapsular spread of nodal metastasis as a prognostic factor in rectal cancer. Int J Radiat Oncol Biol Phys. 2004;58:773-778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 33] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 67. | Yano H, Saito Y, Kirihara Y, Takashima J. Tumor invasion of lymph node capsules in patients with Dukes C colorectal adenocarcinoma. Dis Colon Rectum. 2006;49:1867-1877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 68. | Brynes RK, Hunter RL, Vellios F. Immunomorphologic changes in regional lymph nodes associated with cancer. Arch Pathol Lab Med. 1983;107:217-221. [PubMed] |
| 69. | Pihl E, Nairn RC, Milne BJ, Cuthbertson AM, Hughes ES, Rollo A. Lymphoid hyperplasia: a major prognostic feature in 519 cases of colorectal carcinoma. Am J Pathol. 1980;100:469-480. [PubMed] |
| 70. | Berger AC, Sigurdson ER, LeVoyer T, Hanlon A, Mayer RJ, Macdonald JS, Catalano PJ, Haller DG. Colon cancer survival is associated with decreasing ratio of metastatic to examined lymph nodes. J Clin Oncol. 2005;23:8706-8712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 368] [Cited by in RCA: 408] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 71. | Kobayashi H, Enomoto M, Higuchi T, Uetake H, Iida S, Ishikawa T, Ishiguro M, Kato S, Sugihara K. Clinical significance of lymph node ratio and location of nodal involvement in patients with right colon cancer. Dig Surg. 2011;28:190-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 37] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 72. | Storli KE, Søndenaa K, Bukholm IR, Nesvik I, Bru T, Furnes B, Hjelmeland B, Iversen KB, Eide GE. Overall survival after resection for colon cancer in a national cohort study was adversely affected by TNM stage, lymph node ratio, gender, and old age. Int J Colorectal Dis. 2011;26:1299-1307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 37] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 73. | Sjo OH, Merok MA, Svindland A, Nesbakken A. Prognostic impact of lymph node harvest and lymph node ratio in patients with colon cancer. Dis Colon Rectum. 2012;55:307-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 83] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 74. | Schiffmann L, Eiken AK, Gock M, Klar E. Is the lymph node ratio superior to the Union for International Cancer Control (UICC) TNM system in prognosis of colon cancer? World J Surg Oncol. 2013;11:79. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 75. | Gleisner AL, Mogal H, Dodson R, Efron J, Gearhart S, Wick E, Lidor A, Herman JM, Pawlik TM. Nodal status, number of lymph nodes examined, and lymph node ratio: what defines prognosis after resection of colon adenocarcinoma? J Am Coll Surg. 2013;217:1090-1100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 55] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 76. | Zhang J, Lv L, Ye Y, Jiang K, Shen Z, Wang S. Comparison of metastatic lymph node ratio staging system with the 7th AJCC system for colorectal cancer. J Cancer Res Clin Oncol. 2013;139:1947-1953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 77. | Moug SJ, Oliphant R, Balsitis M, Molloy RG, Morrison DS. The lymph node ratio optimises staging in patients with node positive colon cancer with implications for adjuvant chemotherapy. Int J Colorectal Dis. 2014;29:599-604. [PubMed] |
| 78. | Medani M, Kelly N, Samaha G, Duff G, Healy V, Mulcahy E, Condon E, Waldron D, Saunders J, Coffey JC. An appraisal of lymph node ratio in colon and rectal cancer: not one size fits all. Int J Colorectal Dis. 2013;28:1377-1384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 79. | Shia J, McManus M, Guillem JG, Leibold T, Zhou Q, Tang LH, Riedel ER, Weiser MR, Paty PB, Temple LK. Significance of acellular mucin pools in rectal carcinoma after neoadjuvant chemoradiotherapy. Am J Surg Pathol. 2011;35:127-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 84] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 80. | Frankel WL, Jin M. Serosal surfaces, mucin pools, and deposits, oh my: challenges in staging colorectal carcinoma. Mod Pathol. 2015;28 Suppl 1:S95-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 81. | Sigurdson ER. Lymph node dissection: is it diagnostic or therapeutic? J Clin Oncol. 2003;21:965-967. [PubMed] |
| 82. | Fisher B. From Halsted to prevention and beyond: advances in the management of breast cancer during the twentieth century. Eur J Cancer. 1999;35:1963-1973. [PubMed] |
| 83. | Le Voyer TE, Sigurdson ER, Hanlon AL, Mayer RJ, Macdonald JS, Catalano PJ, Haller DG. Colon cancer survival is associated with increasing number of lymph nodes analyzed: a secondary survey of intergroup trial INT-0089. J Clin Oncol. 2003;21:2912-2919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 821] [Cited by in RCA: 845] [Article Influence: 38.4] [Reference Citation Analysis (0)] |
| 84. | Nelson H, Petrelli N, Carlin A, Couture J, Fleshman J, Guillem J, Miedema B, Ota D, Sargent D. Guidelines 2000 for colon and rectal cancer surgery. J Natl Cancer Inst. 2001;93:583-596. [PubMed] |
| 85. | Guidelines for the management of colorectal cancer, 3rd ed. Association of Coloproctology of Great Britain & Ireland; 2007. Available from: http://www.acpgbi.org.uk/resources/guidelines/guidelines-for-the-management-of-colorectal-cancer/. |
| 86. | Smith AJ, Driman DK, Spithoff K, Hunter A, McLeod RS, Simunovic M, Langer B. Guideline for optimization of colorectal cancer surgery and pathology. J Surg Oncol. 2010;101:5-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 48] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 87. | Watanabe T, Itabashi M, Shimada Y, Tanaka S, Ito Y, Ajioka Y, Hamaguchi T, Hyodo I, Igarashi M, Ishida H. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2010 for the treatment of colorectal cancer. Int J Clin Oncol. 2012;17:1-29. [PubMed] |
| 88. | Grinnell RS. Distal intramural spread of carcinoma of the rectum and rectosigmoid. Surg Gynecol Obstet. 1954;99:421-430. [PubMed] |
| 89. | Devereux DF, Deckers PJ. Contributions of pathologic margins and Dukes’ stage to local recurrence in colorectal carcinoma. Am J Surg. 1985;149:323-326. [PubMed] |
| 90. | Williams NS, Dixon MF, Johnston D. Reappraisal of the 5 centimetre rule of distal excision for carcinoma of the rectum: a study of distal intramural spread and of patients’ survival. Br J Surg. 1983;70:150-154. [PubMed] |
| 91. | Japanese classification of colorectal carcinoma. Japanese Society for Cancer of the Colon. 2nd ed. Kanehara: Kanehara & Co. Ltd 2009; . |
| 92. | Yada H, Sawai K, Taniguchi H, Hoshima M, Katoh M, Takahashi T. Analysis of vascular anatomy and lymph node metastases warrants radical segmental bowel resection for colon cancer. World J Surg. 1997;21:109-115. [PubMed] |
| 93. | Park IJ, Choi GS, Kang BM, Lim KH, Jun SH. Lymph node metastasis patterns in right-sided colon cancers: is segmental resection of these tumors oncologically safe? Ann Surg Oncol. 2009;16:1501-1506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 70] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 94. | Rouffet F, Hay JM, Vacher B, Fingerhut A, Elhadad A, Flamant Y, Mathon C, Gainant A. Curative resection for left colonic carcinoma: hemicolectomy vs. segmental colectomy. A prospective, controlled, multicenter trial. French Association for Surgical Research. Dis Colon Rectum. 1994;37:651-659. [PubMed] |
| 96. | Morikawa E, Yasutomi M, Shindou K, Matsuda T, Mori N, Hida J, Kubo R, Kitaoka M, Nakamura M, Fujimoto K. Distribution of metastatic lymph nodes in colorectal cancer by the modified clearing method. Dis Colon Rectum. 1994;37:219-223. [PubMed] |
| 97. | Toyota S, Ohta H, Anazawa S. Rationale for extent of lymph node dissection for right colon cancer. Dis Colon Rectum. 1995;38:705-711. [PubMed] |
| 98. | Chin CC, Yeh CY, Tang R, Changchien CR, Huang WS, Wang JY. The oncologic benefit of high ligation of the inferior mesenteric artery in the surgical treatment of rectal or sigmoid colon cancer. Int J Colorectal Dis. 2008;23:783-788. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 74] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 99. | Moynihan B. The surgical treatment of cancer of the sigmoid flexure and rectum. Surg Gynecol Obstet. 1908;6:463-466. |
| 100. | Titu LV, Tweedle E, Rooney PS. High tie of the inferior mesenteric artery in curative surgery for left colonic and rectal cancers: a systematic review. Dig Surg. 2008;25:148-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 106] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 101. | Lange MM, Buunen M, van de Velde CJ, Lange JF. Level of arterial ligation in rectal cancer surgery: low tie preferred over high tie. A review. Dis Colon Rectum. 2008;51:1139-1145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 160] [Cited by in RCA: 157] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 102. | Cirocchi R, Trastulli S, Farinella E, Desiderio J, Vettoretto N, Parisi A, Boselli C, Noya G. High tie versus low tie of the inferior mesenteric artery in colorectal cancer: a RCT is needed. Surg Oncol. 2012;21:e111-e123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 99] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 103. | Hida J, Okuno K. High ligation of the inferior mesenteric artery in rectal cancer surgery. Surg Today. 2013;43:8-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 63] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 104. | Mari G, Maggioni D, Costanzi A, Miranda A, Rigamonti L, Crippa J, Magistro C, Di Lernia S, Forgione A, Carnevali P. “High or low Inferior Mesenteric Artery ligation in Laparoscopic low Anterior Resection: study protocol for a randomized controlled trial” (HIGHLOW trial). Trials. 2015;16:21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 105. | Moriya Y, Hojo K, Sawada T, Koyama Y. Significance of lateral node dissection for advanced rectal carcinoma at or below the peritoneal reflection. Dis Colon Rectum. 1989;32:307-315. [PubMed] |
| 106. | Moszkowicz D, Alsaid B, Bessede T, Penna C, Nordlinger B, Benoît G, Peschaud F. Where does pelvic nerve injury occur during rectal surgery for cancer? Colorectal Dis. 2011;13:1326-1334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 74] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 107. | Tsujinaka S, Kawamura YJ, Tan KY, Mizokami K, Sasaki J, Maeda T, Kuwahara Y, Konishi F, Lefor A. Proximal bowel necrosis after high ligation of the inferior mesenteric artery in colorectal surgery. Scand J Surg. 2012;101:21-25. [PubMed] |
| 108. | Hashiguchi Y, Hase K, Ueno H, Mochizuki H, Shinto E, Yamamoto J. Optimal margins and lymphadenectomy in colonic cancer surgery. Br J Surg. 2011;98:1171-1178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 67] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 109. | Georgiou P, Tan E, Gouvas N, Antoniou A, Brown G, Nicholls RJ, Tekkis P. Extended lymphadenectomy versus conventional surgery for rectal cancer: a meta-analysis. Lancet Oncol. 2009;10:1053-1062. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 224] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 110. | Heald RJ, Husband EM, Ryall RD. The mesorectum in rectal cancer surgery--the clue to pelvic recurrence? Br J Surg. 1982;69:613-616. [PubMed] |
| 111. | Heald RJ, Ryall RD. Recurrence and survival after total mesorectal excision for rectal cancer. Lancet. 1986;1:1479-1482. [PubMed] |
| 112. | Hohenberger W, Weber K, Matzel K, Papadopoulos T, Merkel S. Standardized surgery for colonic cancer: complete mesocolic excision and central ligation--technical notes and outcome. Colorectal Dis. 2009;11:354-364; discussion 364-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 990] [Cited by in RCA: 1090] [Article Influence: 68.1] [Reference Citation Analysis (0)] |
| 113. | Søndenaa K, Quirke P, Hohenberger W, Sugihara K, Kobayashi H, Kessler H, Brown G, Tudyka V, D’Hoore A, Kennedy RH. The rationale behind complete mesocolic excision (CME) and a central vascular ligation for colon cancer in open and laparoscopic surgery : proceedings of a consensus conference. Int J Colorectal Dis. 2014;29:419-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 163] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 114. | Chow CF, Kim SH. Laparoscopic complete mesocolic excision: West meets East. World J Gastroenterol. 2014;20:14301-14307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 40] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 115. | West NP, Hohenberger W, Weber K, Perrakis A, Finan PJ, Quirke P. Complete mesocolic excision with central vascular ligation produces an oncologically superior specimen compared with standard surgery for carcinoma of the colon. J Clin Oncol. 2010;28:272-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 496] [Cited by in RCA: 531] [Article Influence: 33.2] [Reference Citation Analysis (0)] |
| 116. | Killeen S, Mannion M, Devaney A, Winter DC. Complete mesocolic resection and extended lymphadenectomy for colon cancer: a systematic review. Colorectal Dis. 2014;16:577-594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 89] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 117. | Willaert W, Ceelen W. Extent of surgery in cancer of the colon: is more better? World J Gastroenterol. 2015;21:132-138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 38] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 118. | Griffin SM. Gastric cancer in the East: same disease, different patient. Br J Surg. 2005;92:1055-1056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 119. | Vennix S, Pelzers L, Bouvy N, Beets GL, Pierie JP, Wiggers T, Breukink S. Laparoscopic versus open total mesorectal excision for rectal cancer. Cochrane Database Syst Rev. 2014;4:CD005200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 101] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 120. | Gouvas N, Pechlivanides G, Zervakis N, Kafousi M, Xynos E. Complete mesocolic excision in colon cancer surgery: a comparison between open and laparoscopic approach. Colorectal Dis. 2012;14:1357-1364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 86] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 121. | Uematsu D, Akiyama G, Magishi A. Multimedia article. Radical lymphadenectomy for advanced colon cancer via separation of the mesocolon into two layers as in filleting fish. Surg Endosc. 2011;25:1659-1660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 122. | Adamina M, Manwaring ML, Park KJ, Delaney CP. Laparoscopic complete mesocolic excision for right colon cancer. Surg Endosc. 2012;26:2976-2980. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 88] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 123. | Feng B, Sun J, Ling TL, Lu AG, Wang ML, Chen XY, Ma JJ, Li JW, Zang L, Han DP. Laparoscopic complete mesocolic excision (CME) with medial access for right-hemi colon cancer: feasibility and technical strategies. Surg Endosc. 2012;26:3669-3675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 80] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 124. | Han DP, Lu AG, Feng H, Wang PX, Cao QF, Zong YP, Feng B, Zheng MH. Long-term results of laparoscopy-assisted radical right hemicolectomy with D3 lymphadenectomy: clinical analysis with 177 cases. Int J Colorectal Dis. 2013;28:623-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 125. | Shin JW, Amar AH, Kim SH, Kwak JM, Baek SJ, Cho JS, Kim J. Complete mesocolic excision with D3 lymph node dissection in laparoscopic colectomy for stages II and III colon cancer: long-term oncologic outcomes in 168 patients. Tech Coloproctol. 2014;18:795-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 126. | West NP, Kennedy RH, Magro T, Luglio G, Sala S, Jenkins JT, Quirke P. Morphometric analysis and lymph node yield in laparoscopic complete mesocolic excision performed by supervised trainees. Br J Surg. 2014;101:1460-1467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 127. | Trinh BB, Jackson NR, Hauch AT, Hu T, Kandil E. Robotic versus laparoscopic colorectal surgery. JSLS. 2014;18:pii: e2014.00187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 41] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 128. | Nordgård O, Oltedal S, Kørner H, Aasprong OG, Tjensvoll K, Gilje B, Heikkilä R. Quantitative RT-PCR detection of tumor cells in sentinel lymph nodes isolated from colon cancer patients with an ex vivo approach. Ann Surg. 2009;249:602-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 34] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 129. | Tiernan JP, Ansari I, Hirst NA, Millner PA, Hughes TA, Jayne DG. Intra-operative tumour detection and staging in colorectal cancer surgery. Colorectal Dis. 2012;14:e510-e520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 130. | Bianchi PP, Ceriani C, Rottoli M, Torzilli G, Roncalli M, Spinelli A, Montorsi M. Laparoscopic lymphatic mapping and sentinel lymph node detection in colon cancer: technical aspects and preliminary results. Surg Endosc. 2007;21:1567-1571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 131. | Nash GM, Row D, Weiss A, Shia J, Guillem JG, Paty PB, Gonen M, Weiser MR, Temple LK, Fitzmaurice G. A predictive model for lymph node yield in colon cancer resection specimens. Ann Surg. 2011;253:318-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 132. | Wright FC, Law CH, Last L, Khalifa M, Arnaout A, Naseer Z, Klar N, Gallinger S, Smith AJ. Lymph node retrieval and assessment in stage II colorectal cancer: a population-based study. Ann Surg Oncol. 2003;10:903-909. [PubMed] |
| 133. | Tekkis PP, Smith JJ, Heriot AG, Darzi AW, Thompson MR, Stamatakis JD. A national study on lymph node retrieval in resectional surgery for colorectal cancer. Dis Colon Rectum. 2006;49:1673-1683. [PubMed] |
| 134. | Shen SS, Haupt BX, Ro JY, Zhu J, Bailey HR, Schwartz MR. Number of lymph nodes examined and associated clinicopathologic factors in colorectal carcinoma. Arch Pathol Lab Med. 2009;133:781-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 135. | Görög D, Nagy P, Péter A, Perner F. Influence of obesity on lymph node recovery from rectal resection specimens. Pathol Oncol Res. 2003;9:180-183. [PubMed] |
| 136. | Linebarger JH, Mathiason MA, Kallies KJ, Shapiro SB. Does obesity impact lymph node retrieval in colon cancer surgery? Am J Surg. 2010;200:478-482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 137. | Glasgow SC, Bleier JI, Burgart LJ, Finne CO, Lowry AC. Meta-analysis of histopathological features of primary colorectal cancers that predict lymph node metastases. J Gastrointest Surg. 2012;16:1019-1028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 80] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 138. | El-Gazzaz G, Hull T, Hammel J, Geisler D. Does a laparoscopic approach affect the number of lymph nodes harvested during curative surgery for colorectal cancer? Surg Endosc. 2010;24:113-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 139. | Berg M, Guriby M, Nordgård O, Nedrebø BS, Ahlquist TC, Smaaland R, Oltedal S, Søreide JA, Kørner H, Lothe RA. Influence of microsatellite instability and KRAS and BRAF mutations on lymph node harvest in stage I-III colon cancers. Mol Med. 2013;19:286-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 140. | Miller ED, Robb BW, Cummings OW, Johnstone PA. The effects of preoperative chemoradiotherapy on lymph node sampling in rectal cancer. Dis Colon Rectum. 2012;55:1002-1007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 64] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 141. | Fielding LP, Arsenault PA, Chapuis PH, Dent O, Gathright B, Hardcastle JD, Hermanek P, Jass JR, Newland RC. Clinicopathological staging for colorectal cancer: an International Documentation System (IDS) and an International Comprehensive Anatomical Terminology (ICAT). J Gastroenterol Hepatol. 1991;6:325-344. [PubMed] |
| 142. | Joseph NE, Sigurdson ER, Hanlon AL, Wang H, Mayer RJ, MacDonald JS, Catalano PJ, Haller DG. Accuracy of determining nodal negativity in colorectal cancer on the basis of the number of nodes retrieved on resection. Ann Surg Oncol. 2003;10:213-218. [PubMed] |
| 143. | Sarli L, Bader G, Iusco D, Salvemini C, Mauro DD, Mazzeo A, Regina G, Roncoroni L. Number of lymph nodes examined and prognosis of TNM stage II colorectal cancer. Eur J Cancer. 2005;41:272-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 235] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 144. | Swanson RS, Compton CC, Stewart AK, Bland KI. The prognosis of T3N0 colon cancer is dependent on the number of lymph nodes examined. Ann Surg Oncol. 2003;10:65-71. [PubMed] |
| 145. | Chang GJ, Rodriguez-Bigas MA, Skibber JM, Moyer VA. Lymph node evaluation and survival after curative resection of colon cancer: systematic review. J Natl Cancer Inst. 2007;99:433-441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 708] [Cited by in RCA: 778] [Article Influence: 43.2] [Reference Citation Analysis (0)] |
| 146. | Kotake K, Honjo S, Sugihara K, Hashiguchi Y, Kato T, Kodaira S, Muto T, Koyama Y. Number of lymph nodes retrieved is an important determinant of survival of patients with stage II and stage III colorectal cancer. Jpn J Clin Oncol. 2012;42:29-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 66] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 147. | Bui L, Rempel E, Reeson D, Simunovic M. Lymph node counts, rates of positive lymph nodes, and patient survival for colon cancer surgery in Ontario, Canada: a population-based study. J Surg Oncol. 2006;93:439-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 109] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 148. | Wong SL, Ji H, Hollenbeck BK, Morris AM, Baser O, Birkmeyer JD. Hospital lymph node examination rates and survival after resection for colon cancer. JAMA. 2007;298:2149-2154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 235] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 149. | van Erning FN, Crolla RM, Rutten HJ, Beerepoot LV, van Krieken JH, Lemmens VE. No change in lymph node positivity rate despite increased lymph node yield and improved survival in colon cancer. Eur J Cancer. 2014;50:3221-3229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 32] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 150. | Parsons HM, Tuttle TM, Kuntz KM, Begun JW, McGovern PM, Virnig BA. Association between lymph node evaluation for colon cancer and node positivity over the past 20 years. JAMA. 2011;306:1089-1097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 144] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 151. | Märkl B, Schaller T, Krammer I, Cacchi C, Arnholdt HM, Schenkirsch G, Kretsinger H, Anthuber M, Spatz H. Methylene blue-assisted lymph node dissection technique is not associated with an increased detection of lymph node metastases in colorectal cancer. Mod Pathol. 2013;26:1246-1254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 152. | Mekenkamp LJ, van Krieken JH, Marijnen CA, van de Velde CJ, Nagtegaal ID. Lymph node retrieval in rectal cancer is dependent on many factors--the role of the tumor, the patient, the surgeon, the radiotherapist, and the pathologist. Am J Surg Pathol. 2009;33:1547-1553. [PubMed] |