Copyright
©The Author(s) 2020.
World J Gastrointest Oncol. Apr 15, 2020; 12(4): 405-423
Published online Apr 15, 2020. doi: 10.4251/wjgo.v12.i4.405
Published online Apr 15, 2020. doi: 10.4251/wjgo.v12.i4.405
Figure 1 Study profile.
1Patients who received both 5-fluorouracil/leucovorin/oxaliplatin and 5-fluorouracil/leucovorin/irinotecan (i.e. 5-fluorouracil/leucovorin/ oxaliplatin/irinotecan) in the first-line were excluded. In addition, patients who received any other drugs as part of first-line therapy were excluded. 2Biomarker status for KRAS wild-type before or within 28 d of starting first-line therapy. If a patient had more than one successful test with a conflicting result during this window, the test result closest to the index date was used. In cases with conflicting test results on the same day, patients were excluded. CRC: Colorectal cancer; ICD: International Statistical Classification of Diseases and Related Health Problems; FOLFIRI: 5-fluorouracil/leucovorin/irinotecan; FOLFOX: 5-fluorouracil/ leucovorin/oxaliplatin; mCRC: Metastatic colorectal cancer; WT: Wild-type.
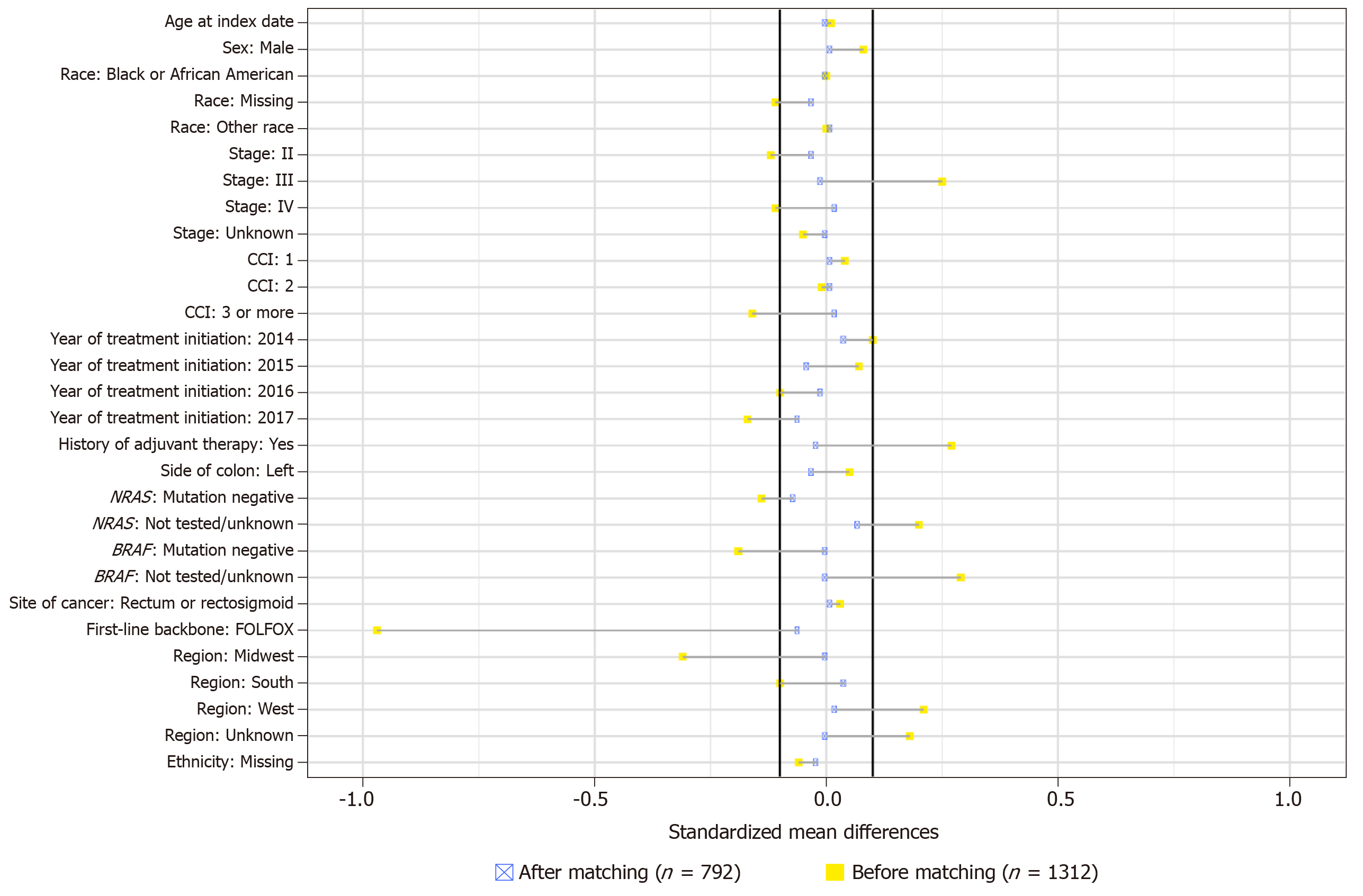
Figure 2 Standardized mean differences between groups across covariates before and after propensity score matching.
CCI: Charlson Comorbidity Index; FOLFOX: 5-fluorouracil/leucovorin/oxaliplatin.
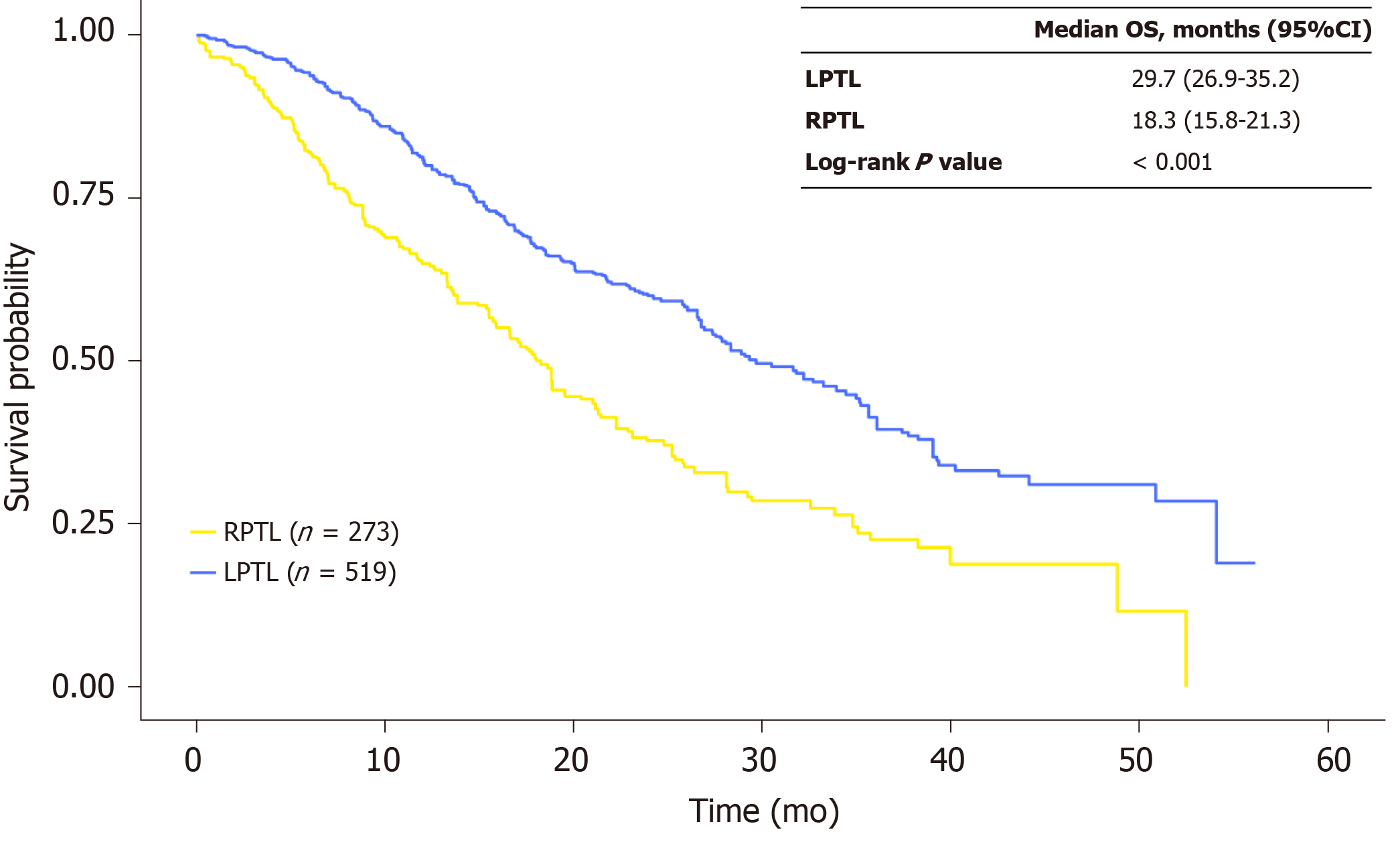
Figure 3 Kaplan-Meier overall survival curves for patients with left-sided primary tumor location vs right-sided primary tumor location in the propensity score–matched sample.
CI: Confidence interval; LPTL: Left-sided primary tumor location; OS: Overall survival; RPTL: Right-sided primary tumor location.
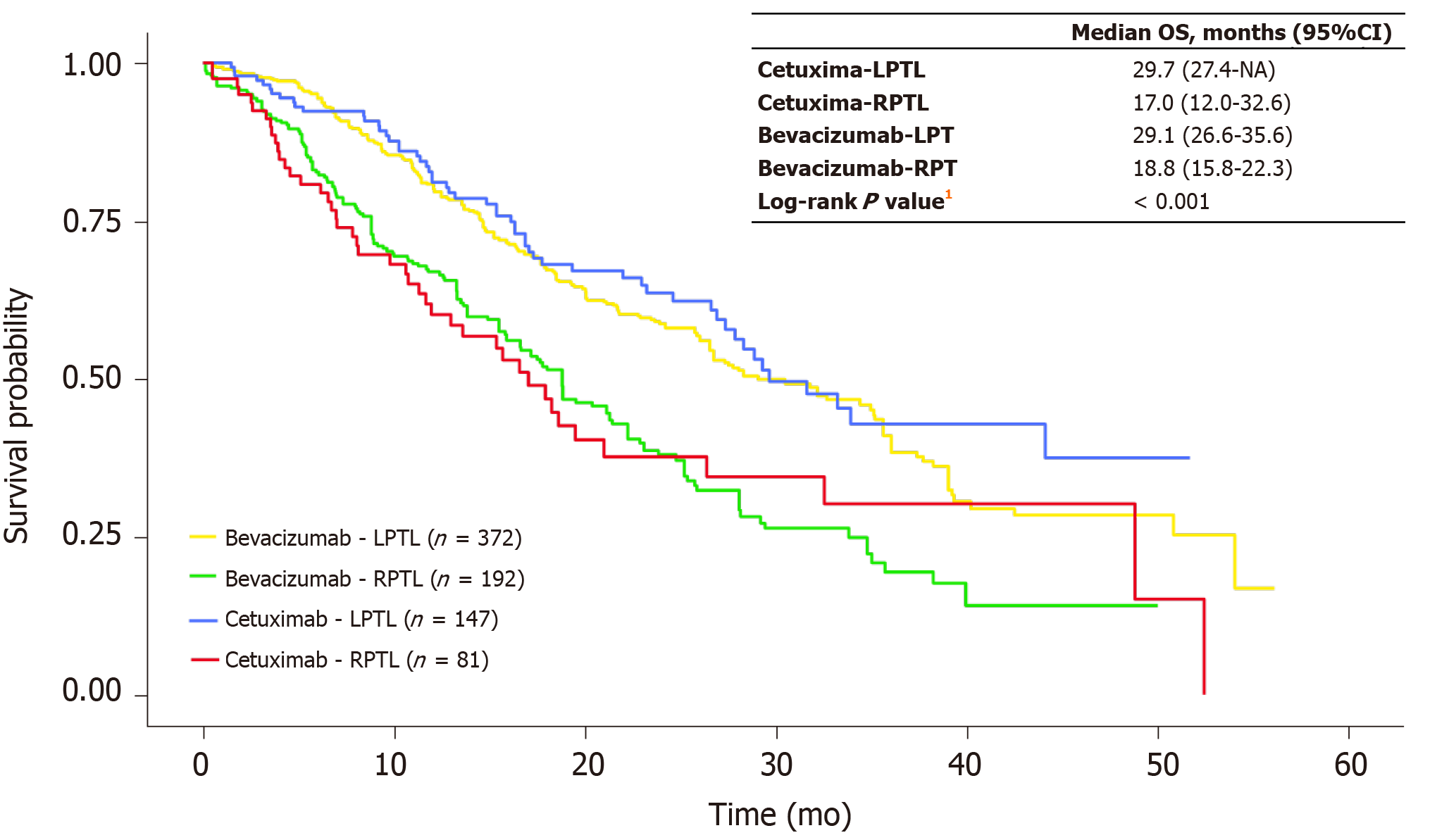
Figure 4 Kaplan-Meier overall survival curves for patients treated with first-line cetuximab versus first-line bevacizumab by primary tumor location in the propensity score–matched sample.
1P value indicates that a significant difference exists across the 4 categories. CI: Confidence interval; LPTL: Left-sided primary tumor location; NA: Not available; OS: Overall survival; RPTL: Right-sided primary tumor location.
- Citation: Aggarwal H, Sheffield KM, Li L, Lenis D, Sorg R, Barzi A, Miksad R. Primary tumor location and survival in colorectal cancer: A retrospective cohort study. World J Gastrointest Oncol 2020; 12(4): 405-423
- URL: https://www.wjgnet.com/1948-5204/full/v12/i4/405.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v12.i4.405