Published online Dec 27, 2012. doi: 10.4254/wjh.v4.i12.406
Revised: August 14, 2012
Accepted: November 14, 2012
Published online: December 27, 2012
Liver cysts are common, affecting 5%-10% of the population. Most are asymptomatic, however 5% of patients develop symptoms, sometimes due to complications and will require intervention. There is no consensus on their management because complications are so uncommon. The aim of this study was to perform a collected review of how a series of complications were managed at our institutions. Six different patients presenting with rare complications of liver cysts were obtained from Hepatobiliary Units in the United Kingdom and The Netherlands. History and radiological imaging were obtained from case notes and computerised radiology. As a result, 1 patient admitted with inferior vena cava obstruction was managed by cyst aspiration and lanreotide; 1 patient with common bile duct obstruction was first managed by endoscopic retrograde cholangiopancreatography and stenting, followed by open fenestration; 1 patient with ruptured cysts and significant medical co-morbidities was managed by percutaneous drainage; 1 patient with portal vein occlusion and varices was managed by open liver resection; 1 patient with infected cysts was treated with intravenous antibiotics and is awaiting liver transplantation. The final patient with a simple liver cyst mimicking a hydatid was managed by open liver resection. In conclusion, complications of cystic liver disease are rare, and we have demonstrated in this series that both operative and non-operative strategies have defined roles in management. The mainstays of treatment are either aspiration/sclerotherapy or, alternatively laparoscopic fenestration. Medical management with somatostatin analogues is a potentially new and exciting treatment option but requires further study.
- Citation: Macutkiewicz C, Plastow R, Chrispijn M, Filobbos R, Ammori BA, Sherlock DJ, Drenth JP, O'Reilly DA. Complications arising in simple and polycystic liver cysts. World J Hepatol 2012; 4(12): 406-411
- URL: https://www.wjgnet.com/1948-5182/full/v4/i12/406.htm
- DOI: https://dx.doi.org/10.4254/wjh.v4.i12.406
Liver cysts can be single, multiple or diffuse, as in polycystic liver disease[1,2]. Simple cysts have a prevalence of 5%-10% with a sharp rise in incidence with age[3]. Polycystic liver disease is an autosomal dominant condition caused by germline mutations of the PRKCSH and SEC63 genes, which encode the β-subunit of glucosidase II and Sec63 respectively[4]. Both proteins are components of the molecular machinery involved in the translocation, folding and quality control of newly synthesized glycoproteins in the endoplasmic reticulum[1]. Neoplastic cysts such as benign biliary cystadenomas and biliary cystadenocarcinomas are acquired, but the cause is unknown. Traumatic cysts are also acquired and result from bile leakage from an injured intrahepatic bile duct after trauma.
The majority of liver cysts are asymptomatic. They can occasionally be felt as a mass during physical examination. However most are found incidentally during abdominal imaging, especially on ultrasound. About 5% of patients are symptomatic and present with vague symptoms such as pain, nausea, early satiety, vomiting or heartburn[3]. Once found, there is a need to distinguish a simple cyst from other cystic lesions of the liver including hydatid cysts and cystic neoplasms. Previously reported complications of liver cysts include: intracystic haemorrhage[5], torsion[6], biliary fistula, rupture[7], infection[8], obstructive jaundice[9,10], malignancy[11], portal vein occlusion and varices[12], portal hypertension[13,14] and Budd-Chiari Syndrome[12].
Although complications of liver cysts are rare, they are often serious and sometimes life-threatening. There is no consensus on their management because complications are so uncommon. The aim of this study was to perform a collected review of how a series of complications were managed at our institutions.
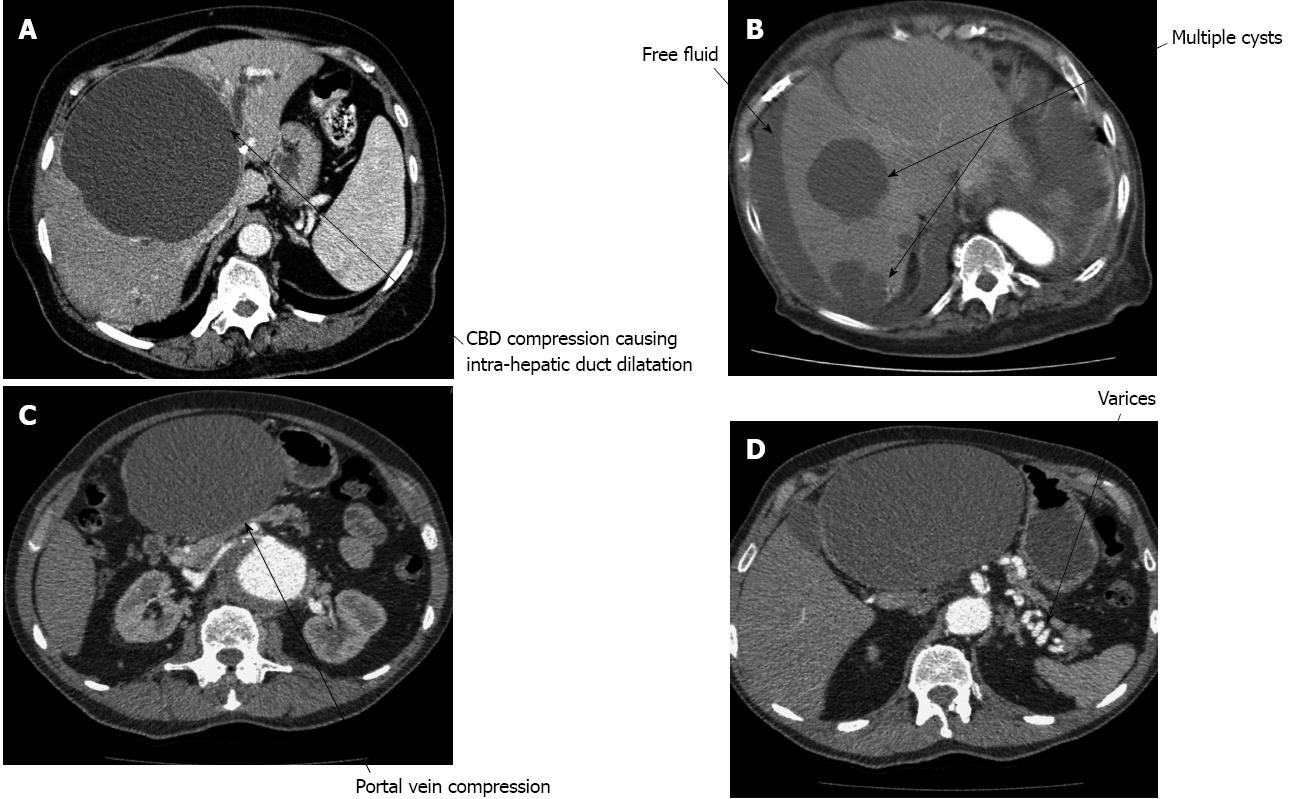
A 75-year-old female patient presented with jaundice and abdominal pain. Liver function tests revealed obstructive jaundice and an ultrasound scan of the abdomen showed a 19 cm unilocular liver cyst and dilated intrahepatic biliary ducts. An endoscopic retrograde cholangiopancreatography (ERCP) was performed which confirmed blockage of the ductal system, a long stricture in the common bile duct and a stent was inserted to temporarily relieve the jaundice.
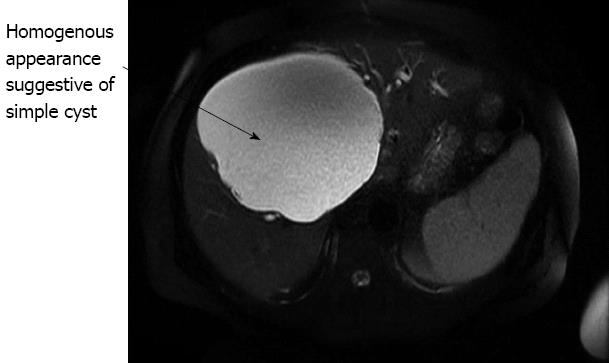
The computed tomography (CT) showed a large uncomplicated cyst occupying segments IV, V, VI and VIII. This was compressing the common bile duct causing obstruction with dilation of the intrahepatic bile ducts (Figure 1A). The magnetic resonance imaging (MRI) confirmed that the liver lesion was homogenous, suggestive of a simple liver cyst (Figure 2).
The patient underwent fenestration (deroofing) of the cyst occupying segments IV, V, VI and VIII and cholecystectomy. Three litres of clear fluid was drained, the roof of the cyst was excised and the cyst lining ablated with Argon Plasma Coagulation diathermy. She was reviewed in the out-patient clinic six weeks later where she reported no further pain, her jaundice had settled and the biliary stent was then removed at ERCP 1 wk later.
A 91-year-old female was admitted with right upper quadrant abdominal pain radiating to the right shoulder tip. She had a past medical history of simple liver cysts, ischaemic heart disease and chronic obstructive pulmonary disease with a poor exercise tolerance. On examination she had tender hepatomegaly. A CT scan showed free fluid in the abdomen and a large residual cyst in segments V and VI (Figure 1B).
Due to her age, medical co-morbidity, and the fact that the cyst had already ruptured it was decided to opt for percutaneous drainage alone. Two drains were inserted; one into the cyst remnant, and another into the subphrenic collection. Apart from a right basal pneumonia, the patient made a full recovery and remains clinically well at 1 year.
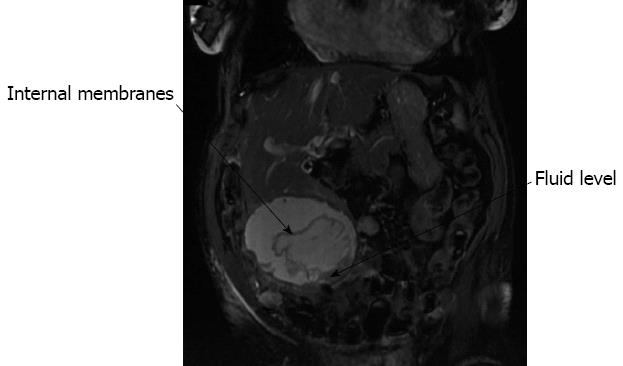
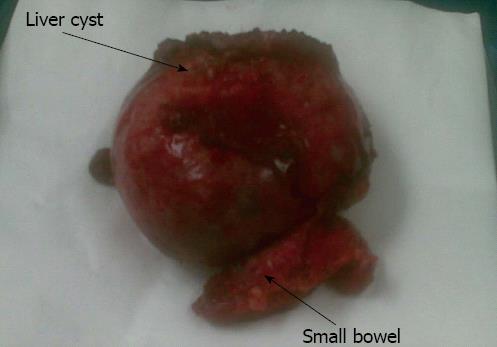
An 82-year-old female was admitted with a three week history of indigestion and epigastric abdominal pain. Her past medical history was of hypertension, left ventricular heart failure and diverticular disease. On examination there was a large mass in the right iliac fossa. An ultrasound scan of the abdomen showed an 8 cm cyst arising from the liver and extending to the right iliac fossa. A MRI of the liver showed a complex exophytic lesion arising from segments V and VI, with apparent internal membranes and fluid levels (Figure 3). Due to the indeterminate nature of the pathology and, despite negative hydatid serology, the patient underwent a liver resection, where a large cystic lesion, containing chocolate-like material was found, arising from segments V and VI. This extended to the right iliac fossa, was attached to the colon and eroding into the small bowel. There were also multiple smaller liver cysts noted. The liver cyst was resected along with a loop of small bowel (Figure 4). Histology confirmed a haemorrhagic simple cyst. The patient made an uncomplicated recovery.
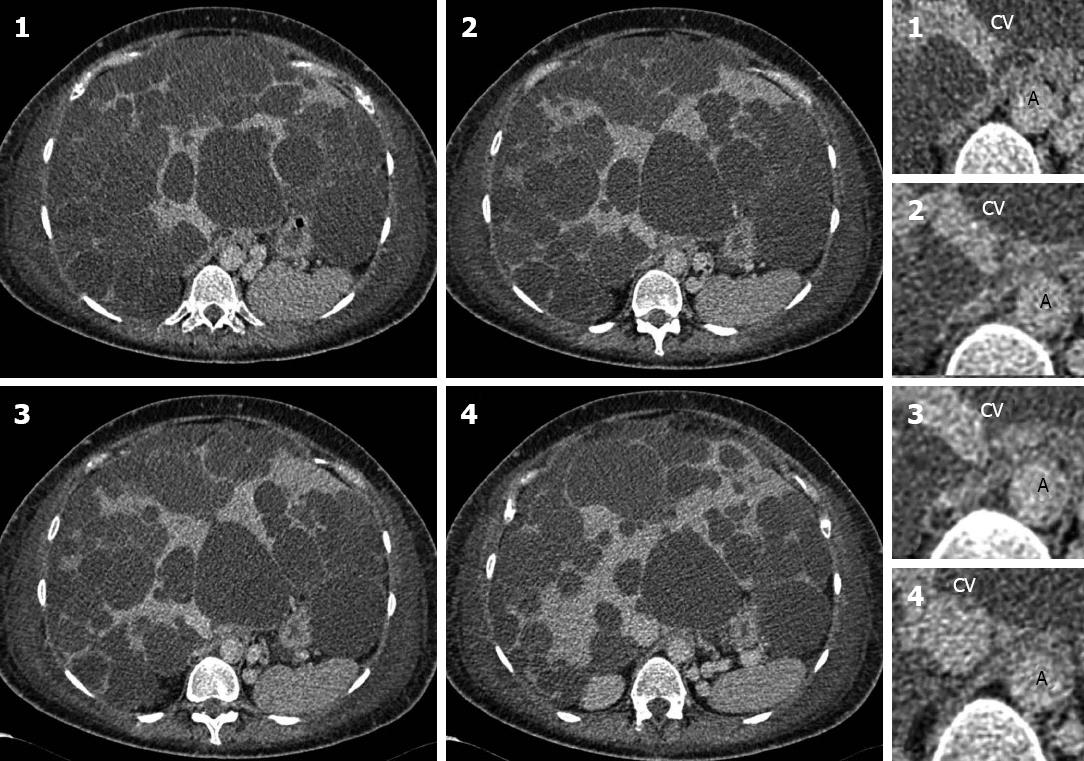
An 80-year-old patient presented with a two week history of abdominal pain and deranged liver function tests. An ultrasound of the abdomen showed a large liver cyst. A CT scan of the abdomen confirmed a large liver cyst occupying segments II and III compressing the portal vein leading to portal hypertension and splenic varices (Figure 1C and D).
The patient had a left hepatectomy. A large simple cyst was found causing portal vein obstruction with splenic varices. He had no post-operative complications.
A 44-year-old female with autosomal dominant polycystic liver disease was admitted because of massive oedema of her lower extremities extending to her abdominal wall. Her weight had increased by 15 kg. These symptoms were precipitated by laparoscopic fenestration of multiple liver cysts. CT scanning revealed the presence of ascites in the lower abdomen, while the upper abdomen was completely occupied by the polycystic liver (Figure 5). CT showed compression of the inferior vena cava. In order to relieve the caval pressure, 3 strategically located cysts were aspirated, the ascites was drained and diuretics and somatostatin analogues were started. She subsequently made a good recovery.
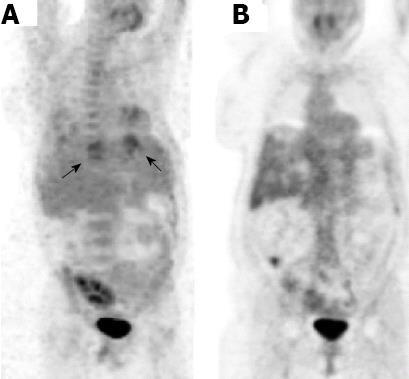
A 47-year-old female with autosomal dominant polycystic liver and kidney disease, was admitted because of pneumonia and multiple liver cyst infection with Klebsiella pneumoniae, diagnosed by blood culture. The liver was grossly enlarged because of numerous cysts. The liver volume was 4.5 L. The infection was successfully treated with ciprofloxacin. Two years later, the liver cysts recurred and blood culture grew Klebsiella pneumoniae, which was treated with oral ciprofloxacin 1 g/d for one year. One year later, she was again admitted because of recurrent infectious liver cysts, this time with an Escherichia coli (E. coli) infection. This was treated with cefuroxime 3 × 1.5 g iv and a single dose of gentamicin. The positron emission tomography CT (PET-CT) scan showed fluorodeoxyglucose (FDG) accumulation at the rim of the cyst. After antibiotic treatment, these FDG-accumulations disappeared (Figure 6). She subsequently developed multiple recurrences of infected liver cysts, most recently with an extended spectrum beta-lactamase-positive E. coli.
Because of the multiple recurrences, the resistant organisms and the fact that the infected liver cysts were spread throughout the liver, the patient is currently waiting for a liver transplantation.
Therapeutic intervention is warranted in the management of symptomatic liver cysts or when complications occur. For symptomatic cases, procedures available include: percutaneous aspiration with ethanol sclerotherapy, laparoscopic or open cyst fenestration (deroofing), hepatic resection and liver transplantation. The somatostatin analogue, lanreotide, reduces the volume of polycystic livers but has a modest clinical effect[15]. Complications of cystic liver disease are rare, and we have demonstrated in this series that both operative and non-operative strategies have defined roles in management.
Biliary obstruction can initially be relieved with ERCP and stent insertion but definitive treatment is required. In this series, definitive treatment was with liver cyst fenestration. Laparoscopic fenestration has largely superseded the open approach for symptomatic liver cysts. This treatment is particularly suited to solitary liver cysts, particularly if more anteriorly located. With careful selection, good operative technique and, in a high-volume centre, good results can be obtained. A recent series of 51 patients reported complete relief of symptoms at a median follow-up of 13 mo, nine minor perioperative complications and a median hospital stay of 2 d[16]. Symptomatic relief and a median volume reduction of 12.5% has been achieved in patients with polycystic liver disease[17]. Percutaneous aspiration with ethanol sclerotherapy would be an acceptable alternative strategy[18], as although the cysts will not disappear, they will reduce in size and this may be sufficient to relieve the extrinsic compression by the cyst on the bile duct.
Liver cyst rupture was managed conservatively with percutaneous drainage in the second patient in this series, due to the patient’s multiple medical co-morbidities and age. Although simple percutaneous drainage of liver cysts is almost inevitably associated with recurrence[19], it was used in this case because the cyst had already ruptured and surgery was not an appropriate option for this patient. Sclerotherapy was not performed because the cyst had already ruptured.
Diagnostic uncertainty often results in surgical resection, in order not to miss a neoplastic condition, such as cystadenoma or cystadenocarcinoma or where the cyst has features of hydatid disease. The latter consideration, coupled with symptoms, led to the decision to undertake open resection of the cyst, with an adjacent segment of small bowel in patient 3. The bowel was intimately adhered to the cyst wall, presumably as a result of a cyst rupture, which the bowel had contained. Hydatid serology was negative in this case but a negative result is not completely reliable and immunodiagnosis plays a minor role in the definitive diagnosis of hydatid disease[20].
Portal vein occlusion, associated with splenic varices occurred as a complication in patient 4. Although segment II and III liver resection is now commonly performed laparoscopically, the presence of portal hypertension and varices indicated that the patient was at high risk of haemorrhage so open resection of the cyst was performed. Experience with liver resection for cystic liver disease is limited; the largest series published to date reported on 124 patients with polycystic liver disease undergoing partial hepatectomy with cyst fenestration, over a 21-year period[21]. Although good symptom relief were obtained, this was at a cost of a 63% in-hospital complication rate and a 3% mortality rate. This operation is technically demanding, as the polycystic liver is large and rigid with decreased mobility and reduced access to vascular inflow and outflow. Hepatic transection is complicated by displacement of hepatic veins and bile ducts from their normal anatomical positions. Liver resection is not, therefore, a first-line treatment for liver cysts but has a particular role when doubt as to the nature of the cyst exists[22].
The fifth patient presented with ascites and obstruction of the inferior vena cava. This was treated by a combination of cyst aspiration, diuretics and somatostatin analogues. The rationale for somatostatin analogue therapy is its inhibitory effect on cholangiocyte proliferation and cyst fluid secretion[23]. The clinical effect of this has recently been evaluated in a randomised double-blind, placebo-controlled trial with the somatostatin analogue, lanreotide in patients with polycystic liver disease[15]. Although an effect on the natural course of the disease was confirmed, this only amounted to a 2.9% reduction of liver volume. Further clinical trials are required before this treatment can be recommended for adoption into practice.
Finally, we demonstrate the utility of PET-CT imaging in demonstrating complete resolution of infected liver cysts with appropriate antibiotic therapy. Multiple proven recurrences have led to this patient with polycystic liver disease being referred for liver transplantation. Liver transplantation remains the only curative option for the minority of patients with severe polycystic liver disease who have disabling symptoms and poor quality of life. Recent results of liver transplantation are encouraging. In a large cohort of 58 patients with isolated polycystic liver disease the 5-year patient survival was 92% and in the cohort with 121 patients with polycystic kidney and liver disease this survival rate was 89%[24]. The collective experience on 218 patients from the European Liver Transplant Registry indicated a 5-year survival of 80%[25].
The current series has demonstrated several rare complications of simple liver cysts and polycystic liver disease. As these are rare, there are no consensus treatment strategies, but we highlight the role of tailored individual treatment, taking into account, cyst site and size, possibility of malignancy, medical co-morbidities and technical feasibility. We have shown that a role for liver resection remains, especially when there is diagnostic uncertainty. The mainstays of treatment are either aspiration/sclerotherapy or, alternatively laparoscopic fenestration. Medical management with somatostatin analogues is a potentially new and exciting treatment option but requires further study.
Peer reviewer: Marcelo AF Ribeiro JR, MD, PhD, Professor of Surgery, Al. Gregorio Bogossian Sobrinho, 80 casa 155, Santana de Parnaiba, SP 06543-385, Brazil
S- Editor Li JY L- Editor A E- Editor Li JY
| 1. | Drenth JP, Martina JA, van de Kerkhof R, Bonifacino JS, Jansen JB. Polycystic liver disease is a disorder of cotranslational protein processing. Trends Mol Med. 2005;11:37-42. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 68] [Cited by in F6Publishing: 57] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 2. | Hoevenaren IA, Wester R, Schrier RW, McFann K, Doctor RB, Drenth JP, Everson GT. Polycystic liver: clinical characteristics of patients with isolated polycystic liver disease compared with patients with polycystic liver and autosomal dominant polycystic kidney disease. Liver Int. 2008;28:264-270. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 82] [Cited by in F6Publishing: 73] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 3. | Gall TM, Oniscu GC, Madhavan K, Parks RW, Garden OJ. Surgical management and longterm follow-up of non-parasitic hepatic cysts. HPB (Oxford). 2009;11:235-241. [PubMed] [Cited in This Article: ] |
| 4. | Drenth JP, te Morsche RH, Smink R, Bonifacino JS, Jansen JB. Germline mutations in PRKCSH are associated with autosomal dominant polycystic liver disease. Nat Genet. 2003;33:345-347. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 5. | Chan CY, Tan CH, Chew SP, Teh CH. Laparoscopic fenestration of a simple hepatic cyst. Singapore Med J. 2001;42:268-270. [PubMed] [Cited in This Article: ] |
| 6. | Davis CK, Schoffstall RO, Glass TF. Fatal complication of hepatic cystic disease. South Med J. 1981;74:1409-1411. [PubMed] [Cited in This Article: ] |
| 7. | Akriviadis EA, Steindel H, Ralls P, Redeker AG. Spontaneous rupture of nonparasitic cyst of the liver. Gastroenterology. 1989;97:213-215. [PubMed] [Cited in This Article: ] |
| 8. | Gesundheit N, Kent DL, Fawcett HD, Effron MK, Maffly RH. Infected liver cyst in a patient with polycystic kidney disease. West J Med. 1982;136:246-249. [PubMed] [Cited in This Article: ] |
| 9. | Lai EC, Wong J. Symptomatic nonparasitic cysts of the liver. World J Surg. 1990;14:452-456. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 47] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 10. | Cappell MS. Obstructive jaundice from benign, nonparasitic hepatic cysts: identification of risk factors and percutaneous aspiration for diagnosis and treatment. Am J Gastroenterol. 1988;83:93-96. [PubMed] [Cited in This Article: ] |
| 11. | Bloustein PA, Silverberg SG. Squamous cell carcinoma originating in an hepatic cyst. Case report with a review of the hepatic cyst-carcinoma association. Cancer. 1976;38:2002-2005. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 2] [Reference Citation Analysis (0)] |
| 12. | Johnstone AJ, Turnbull LW, Allan PL, Garden OJ. Cholangitis and Budd Chiari syndrome as complications of simple cystic liver disease. HPB Surg. 1993;6:223-227; discussion 227-228. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 10] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 13. | Ratcliffe PJ, Reeders S, Theaker JM. Bleeding oesophageal varices and hepatic dysfunction in adult polycystic kidney disease. Br Med J (Clin Res Ed). 1984;288:1330-1331. [PubMed] [Cited in This Article: ] |
| 14. | Ribeiro Jr MAF, De Molla Neto OL, Goncalves A, Saad WA, Chaib E. Portal hypertension due to a non-parasitic hepatic cyst. Einstein. 2003;1:110-112. [Cited in This Article: ] |
| 15. | van Keimpema L, Nevens F, Vanslembrouck R, van Oijen MG, Hoffmann AL, Dekker HM, de Man RA, Drenth JP. Lanreotide reduces the volume of polycystic liver: a randomized, double-blind, placebo-controlled trial. Gastroenterology. 2009;137:1661-1668. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 189] [Cited by in F6Publishing: 174] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 16. | Gamblin TC, Holloway SE, Heckman JT, Geller DA. Laparoscopic resection of benign hepatic cysts: a new standard. J Am Coll Surg. 2008;207:731-736. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 47] [Cited by in F6Publishing: 51] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 17. | van Keimpema L, Ruurda JP, Ernst MF, van Geffen HJ, Drenth JP. Laparoscopic fenestration of liver cysts in polycystic liver disease results in a median volume reduction of 12.5%. J Gastrointest Surg. 2008;12:477-482. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 47] [Cited by in F6Publishing: 48] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 18. | van Keimpema L, de Koning DB, Strijk SP, Drenth JP. Aspiration-sclerotherapy results in effective control of liver volume in patients with liver cysts. Dig Dis Sci. 2008;53:2251-2257. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 34] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 19. | Saini S, Mueller PR, Ferrucci JT, Simeone JF, Wittenberg J, Butch RJ. Percutaneous aspiration of hepatic cysts does not provide definitive therapy. AJR Am J Roentgenol. 1983;141:559-560. [PubMed] [Cited in This Article: ] |
| 20. | Zhang W, Li J, McManus DP. Concepts in immunology and diagnosis of hydatid disease. Clin Microbiol Rev. 2003;16:18-36. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 245] [Cited by in F6Publishing: 244] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 21. | Schnelldorfer T, Torres VE, Zakaria S, Rosen CB, Nagorney DM. Polycystic liver disease: a critical appraisal of hepatic resection, cyst fenestration, and liver transplantation. Ann Surg. 2009;250:112-118. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 118] [Cited by in F6Publishing: 105] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 22. | Ammori BJ, Jenkins BL, Lim PC, Prasad KR, Pollard SG, Lodge JP. Surgical strategy for cystic diseases of the liver in a western hepatobiliary center. World J Surg. 2002;26:462-469. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 73] [Cited by in F6Publishing: 62] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 23. | Alvaro D, Gigliozzi A, Attili AF. Regulation and deregulation of cholangiocyte proliferation. J Hepatol. 2000;33:333-340. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 82] [Cited by in F6Publishing: 86] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 24. | van Keimpema L, Nevens F, Adam R, Porte RJ, Fikatas P, Becker T, Kirkegaard P, Metselaar HJ, Drenth JP. Excellent survival after liver transplantation for isolated polycystic liver disease: an European Liver Transplant Registry study. Transpl Int. 2011;24:1239-1245. [PubMed] [Cited in This Article: ] |
| 25. | Adam R, McMaster P, O'Grady JG, Castaing D, Klempnauer JL, Jamieson N, Neuhaus P, Lerut J, Salizzoni M, Pollard S. Evolution of liver transplantation in Europe: report of the European Liver Transplant Registry. Liver Transpl. 2003;9:1231-1243. [PubMed] [Cited in This Article: ] |