Published online Feb 27, 2019. doi: 10.4254/wjh.v11.i2.173
Peer-review started: August 29, 2018
First decision: October 16, 2018
Revised: November 19, 2018
Accepted: January 28, 2019
Article in press: January 28, 2019
Published online: February 27, 2019
Processing time: 181 Days and 14.4 Hours
Hepatic encephalopathy (HE) is a major complication that is closely related to the progression of end-stage liver disease. Metabolic changes in advanced liver failure can promote cognition impairment, attention deficits and motor dysfunction that may result in coma and death. HE can be subdivided according to the type of hepatic injury, namely, type A, which results from acute liver failure, type B, which is associated with a portosystemic shunting without intrinsic liver disease, and type C, which is due to chronic liver disease. Several studies have investigated the pathogenesis of the disease, and most of the mechanisms have been explored using animal models. This article aimed to review the use of preclinical models to investigate HE. The most used animal species are rats and mice. Experimental models of type A HE include surgical procedures and the administration of hepatotoxic medications, whereas models of types B and C HE are generally surgically induced lesions in liver tissue, which evolve to hepatic cirrhosis. Preclinical models have allowed the comprehension of the pathways related to HE.
Core tip: Hepatic encephalopathy (HE) is a major complication closely related to the progression of end-stage liver disease. It can be subdivided according to the type of hepatic injury: type A, which results from acute liver failure, type B, which is associated with a portosystemic shunting without intrinsic liver disease, and type C, which is due to chronic liver disease. In this article, we have described the use of preclinical models to investigate HE. We have briefly described the applicability and the characteristics of these experimental models. In conclusion, preclinical models have allowed the comprehension of the pathways related to HE.
- Citation: Lima LCD, Miranda AS, Ferreira RN, Rachid MA, Simões e Silva AC. Hepatic encephalopathy: Lessons from preclinical studies. World J Hepatol 2019; 11(2): 173-185
- URL: https://www.wjgnet.com/1948-5182/full/v11/i2/173.htm
- DOI: https://dx.doi.org/10.4254/wjh.v11.i2.173
Hepatic encephalopathy (HE) is defined as a neuropsychiatric syndrome that occurs in patients with acute or chronic liver diseases[1]. Clinically, it is characterized by a spectrum of symptoms, including cognition impairment, altered levels of consciousness that may progress to coma and death[2]. The risk of mortality has usually been related to higher grades of HE, and the incidence of this disease is increased in patients with frequent infections[3,4]. Patients with HE are greatly impacted in terms of quality of life and the high costs caused by increased healthcare utilization[5,6].
The pathophysiology of HE is not completely understood, and several studies have shown biochemical disturbances, with elevation of serum ammonia levels and increased oxidative stress in blood, alterations of neurotransmission systems, development of brain edema, astrocyte swelling and inflammation[8]. A wide range of animals, including dogs, goats, pigs, rabbits, guinea pigs, rats and mice, have been used for better understanding the mechanisms underlying HE pathogenesis and for the development of novel therapeutic agents for HE, providing a basis for future clinical research. Therefore, the objective of this brief review is to discuss relevant aspects of the HE pathogenesis and the current animal models employed, which may closely resemble the disease in humans.
HE is a central nervous system (CNS) dysfunction resulting from acute or chronic liver failure that leads to a wide range of neuropsychiatric manifestations[7]. Clinically, patients with HE may present sleep-wake cycle disturbance, personality changes and cognitive, motor activity and coordination dysfunctions, which ultimately progress to stupor, coma and death. Importantly, HE often affects health-related quality of life, clinical management strategies, liver transplant priority, and patient survival[8].
According to the American Association for the Study of Liver Disease (AASLD) updated guidelines, HE should be classified as type A, B, or C based on the underlying disease. Type A (acute) results from an acute liver failure (ALF), while type B (bypass) is associated with a portosystemic shunting without intrinsic liver disease and type C (chronic) is the consequence of cirrhosis[7]. The incidence of ALF is low, affecting approximately 2000 people per year in the United States or Europe[8,9]. However, the mortality of ALF is high and is mostly attributed to fast progression to HE, leading to cerebral edema, increased intracranial pressure and cerebral herniation[8,9].
In contrast, chronic liver diseases are highly prevalent, affecting approximately 5.5 million individuals only in the United States. Approximately 80% of patients with cirrhosis will develop a less severe form of HE, known as minimal HE (MHE), which is characterized by mild cognitive impairment, attention deficits, psychomotor slowing and impaired visuomotor and bimanual coordination[10]. MHE is detected only by employing psychometric or neurophysiological tests and is considered an important predictive factor for the development of HE since 30%-40% of cirrhotic patients progress to this later condition[10,11]. Apart from its educational and social impact, HE also carries a significant economic burden. For instance, in 2009, in the United States, HE led to approximately 22931 hospitalizations, with an average cost of each stay ranging from 46663 to 63108 USD[8].
The broad spectrum of HE, especially regarding the type of underlying disease, severity of clinical manifestations, and precipitating factors (e.g., infections, gastrointestinal bleeding and drug toxicities) should be considered to understand the pathophysiological mechanisms of HE as well as to identify potential therapeutic targets[12].
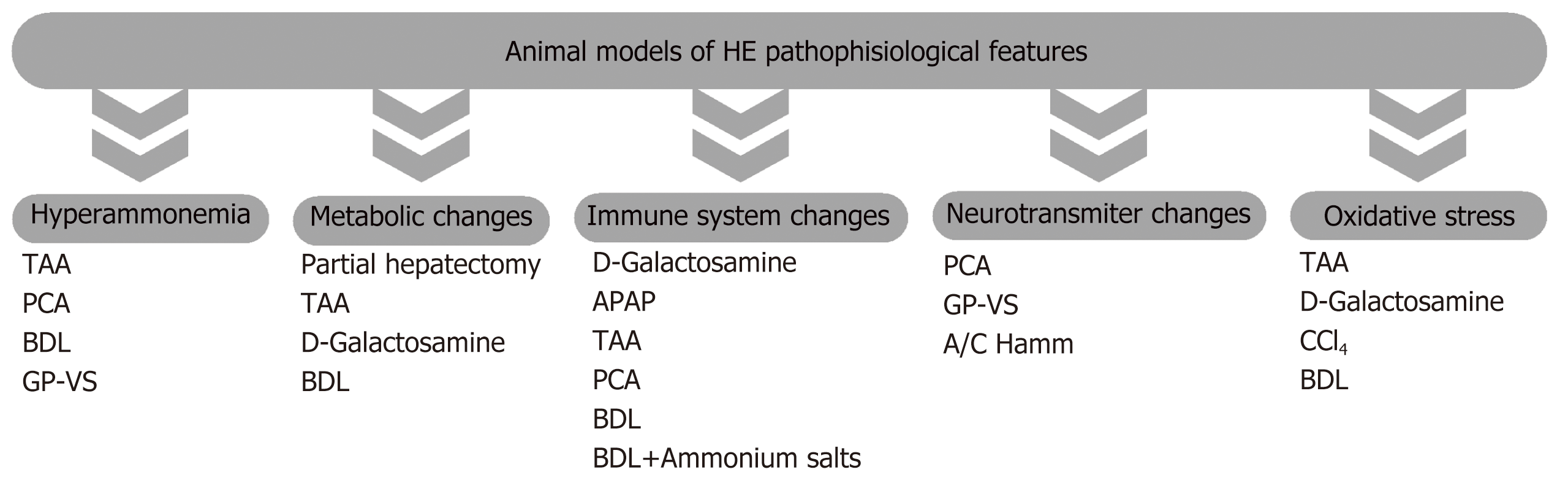
HE is a complex condition who’s cellular and molecular mechanisms remain to be fully elucidated. Over the past decades, some hypotheses have been proposed, including neurotransmitter system dysfunction, impaired energy and lactate metabolism and oxidative stress (for review see[1,13]). However, the hyperammonemia and the neuroinflammation hypotheses have been the mostly recognized ones and will be briefly revisited in the current review. Figure 1 shows the main pathophysiological features of the available models of HE.
Currently, increased systemic and brain levels of ammonia are the main factors implicated in HE pathogenesis. Under physiological conditions, the ammonia, resulting from nitrogenous compounds, such as proteins metabolized by gut microflora, is metabolized in the liver via the urea cycle, forming urea, which is mainly excreted by the kidneys. ALF, portosystemic shunting or chronic liver disease can impair liver function, leading to increased levels of ammonia in the blood[14].
Ammonia metabolism in the liver depends on phosphate-activated glutaminase (PAG), which catalyzes the hydrolysis of glutamine to produce glutamate, energy, nucleotide synthesis and ammonia. PAG has two isoforms, the hepatic type (L-PAG), restricted to the liver, and the kidney-type (K-PAG), found in the kidney, brain and enterocyte villi, especially in the small intestine. Interestingly, PAG activity in the intestine has been associated with increased systemic levels of ammonia during liver cirrhosis and seems to play a major role in the pathogenesis of HE[15,16].
As the levels of ammonia increase systemically, the molecule crosses the blood-brain barrier and starts to be metabolized in the CNS[14]. The ammonia detoxification in the brain requires its incorporation into glutamine by the action of the enzyme glutamine synthetase, which is present only in astrocytes. The glutamine accumulation in astrocytes as a result of ammonia detoxification results in increased water entry and osmotic forces, ultimately inducing astrocytes to swell and causing cytotoxic edema[17]. The impact of hyperammonemia on astrocyte function in response to HE remains to be fully elucidated. However, it has been reported that, apart from increasing oxidative stress and osmotic pressure, hyperammonemia may also influence inflammatory and signal transduction pathways[18,19], gene expression and neurotransmitter release[20] as well as posttranslational protein modifications[21].
Although a great deal of attention continues to be focused on ammonia as the main toxin related to HE pathogenesis, there is evidence that patients with elevated levels of systemic and local ammonia may not present HE symptoms; additionally, the ammonia concentration is not always consistent with the severity of HE in cirrhotic patients[22,23]. Moreover, ammonia-lowering agents, including L-ornithine, L-aspartate and lactulose, have so far been of limited value in preventing HE in ALF and in cirrhosis[24-26], supporting a role for other factors alone or in association with ammonia in the development of HE. For instance, the effect of the glycerol phenylbutyrate (GPB), approved by the Food and Drug Administration in 2013 for the treatment of urea cycle disorders, was investigated in a randomized, double-blind, placebo-controlled phase II trial with cirrhotic patients who experienced two or more HE events in the last 6 mo. The GPB acts as an ammonia-lowering agent by producing phenylacetyl glutamine, which is excreted in urine. GPB treatment in cirrhotic patients decreased plasma levels of ammonia, the proportion of patients who experienced HE and hospitalizations due to HE. These findings supported the involvement of ammonia in HE pathophysiology and the potential therapeutic role of GPB[27].
In addition to the ammonia hypothesis, brain inflammation, also known as neuroinflammation, is thought to be a major component in the development of HE. Clinical and experimental evidence of activation of microglia, the brain resident immune cells, in response to ALF and cirrhosis extensively supported the neuroinflammation hypothesis[28-32]. For instance, increased expression of the major histocompatibility complex class II antigen marker CD11b/c (also termed OX-42), an indicator of microglial activation, was found in an ALF model following liver ischemia in rats[30]. Importantly, the administration of minocycline, a potent inhibitor of microglial activation, attenuates the encephalopathy grade and prevents brain edema in the same ALF experimental model[32]. Corroborating these findings, increased expression levels of microglial activation markers, including human leukocyte antigen DR (CR3/43) and ionized calcium-binding adaptor molecule 1 (Iba-1), were found in the CNSs of patients with ALF associated with viral hepatitis[29] and in liver cirrhosis[31], respectively.
Microglial activation has often been associated with increased release of cytokines and chemokines, which are also implicated in the pathogenesis of neurodegenerative and neuropsychiatric diseases[33,34]. The first evidence of upregulation of inflammatory molecules in HE was obtained in a study of patients with ALF due to acetaminophen overdose. The levels of inflammatory cytokines (TNF, IL-1β and IL-6) were measured in blood samples from an artery and a reverse jugular catheter. Increased arterial levels of cytokines correlated with intracranial hypertension. Brain cytokine efflux was noted, indicating brain cytokine production in these patients[35]. Supporting data were also provided by several studies employing pharmacological and nonpharmacological liver failure models in rodents, which also showed increased levels of inflammatory cytokines (TNF, IL-1β and IL-6) and chemokines, such as CXCL-I, CCL2, CCL3, CCL5 and CX3CL1[30,32,36-38]. Importantly, anti-inflammatory-based strategies attenuated cognitive decline and motor activity impairment, supporting the involvement of neuroinflammation in HE pathophysiology[39-42]. It is worth mentioning that ammonia alone is capable of inducing microglial and astrocyte activation, leading to increased expression of inflammatory cytokines, such as IL-1β and IL-6. This finding suggests that hyperammonemia may trigger neuroin-flammation in HE in a synergistic manner. However, a direct link between both mechanisms is still missing[43].
In 2008, a commission formed by members of ISHEN (International Society of Hepatic Encephalopathy and Nitrogen Metabolism) gathered in Padua, Italy to establish guidelines for HE animal models. Due to the variety of etiologies of hepatic dysfunction and the many factors that may influence the development of neurological symptoms, HE is considered difficult to reproduce faithfully in animals. Table 1 summarizes the main advantages and disadvantages of the available HE models. Currently, there is no ideal animal model to resemble hepatic failure caused by ethanol, virus or acetaminophen, the most common etiologies in human beings[44].
| Model features | Advantages | Disadvantages | |
| Type A Model. Encephalopathy associated with acute liver failure | Type A models have been developed by exclusion (anhepatic models), partial removal of the liver or from the administration of a hepatotoxins | Anhepatic model: The course of HE is relatively rapid. Applied to studies of brain metabolism, neurotransmission abnormalities, gene expression and brain inflammation in ALF. The model responds to hypothermia, ammonia-lowering agents and anti-inflammatory drugs; Hepatotoxic model: Different hepatotoxins could be used to create Type A models. Generally, these models produce hypothermia, hypoglycemia and other systemic complications; Thioacetamide: Model with good repeatability, easy operation, and high similarity to human HE; Acetaminophen: Easy preparation, low price and dose-dependence; D-galactosamine: Good repeatability. Simulates the pathophysiological changes of acute liver failure. Shows manifestations of liver injury that are similar to viral hepatic failure | Anhepatic model: Neither procedure could lead to a potential recovery. The surgical procedure causes great trauma to the animal. Absence of injured or necrotic hepatic cells. The toxic substances and inflammatory mediators present in the injured liver are not perfused into the blood circulation; Partial hepatectomy: The surgery is difficult to control. Severe hypoglycemia leads to death; Hepatotoxic model: Each of these toxins could produce hepatitis with variable pathological nature. Animal-to-animal variations lead to a lack of reproducibility. Some hepatotoxins show extrahepatic toxicity; Acetaminophen: Poor reproducibility. Shows side effects in kidneys and other organs; D-galactosamine: High cost, short survival time and poor stability |
| Type B Model. Encephalopathy associated with portosystemic bypass without liver disease | Type B models have been developed by portosystemic shunting (portacaval anastomosis, congenital portacaval shunts, graded portal vein stenosis, and biliary duct ligation). Different HE aspects could be assessed using different shunt methods and species (pig, dog, rabbit, rat, and mouse) | Portacaval anastomosis: Can better simulate the clinical mild HE phenotypes in different animal models. (Dog) EEG changes and neurological status correlate with ammonia in the plasma; Congenital portacaval shunts: (Dogs) Naturally develop psychomotor dysfunction, reduced hepatic function, and hyperammonemia and are susceptible to high-protein diets; Graded portal vein stenosis: Easy to perform. The surgery may be reversed. (Rat) Provides a Minimal Hepatic Encephalopathy model. (Rat) Develop loss of activity, altered circadian rhythm, hyperammonemia, and altered ammonia/glutamine in the brain | Portacaval anastomosis: Cause severe coma due to hypersensitivity to ammonia. (Rabbit) Portacaval anastomosis may lead to death of animal in most cases. (Rat) Needs high surgical skills to perform; Congenital portacaval shunts: Access to animals with this congenital alteration |
| Type C Model. Encephalopathy associated with liver disease | HE associated with cirrhosis and portal hypertension (Type C) is the most common form of HE in patients. At present, there is no appropriate model to study HE that occurs in liver cirrhosis; nevertheless, some models have been developed | Biliary duct ligation: Animals develop liver failure, jaundice, portal hypertension, immune system dysfunction, and bacterial translocation. (Rat) Reproducible model of biliary cirrhosis with the development of hyperammonemia, low-grade encephalopathy, and decreased locomotor activity. (Rat) Bile duct-ligated animals fed with ammonium salts provide a model that reproduces human Alzheimer Type II astrocytosis | No satisfactory Type C animal model induced by alcoholic liver disease or viral hepatitis exists at the present time; Biliary duct ligation: Immune system dysfunction and other diseases may affect the final HE phenotypes. (Rat) Weight loss due to hunger suppression; CCl4: Animal-to-animal variations lead to a lack of reproducibility. Limited neurobehavioral assessment due to the presence of ascites. Possible health damage when a researcher uses this chemical |
| Models of acute/chronic hyperammonemia | These models are designed to study the effects of hyperammonemia on brain function without liver dysfunction | Inexpensive and simple to perform. The model shows alterations of multiple neurotransmitter systems in the brain. The model shows impairments in learning and memory | Limited to rats and mice. Time-consuming; not suitable for long-term studies. Lacks liver failure |
The availability of animal models is indispensable for studying the mechanisms of diseases and possible therapies[45]. Many animal species have been used for the investigation of HE, including dogs, goats, pigs, rabbits, guinea pigs, rats and mice. Large animals are advantageous for neurological examinations and the availability of biological samples, such as blood samples, body fluids and biopsies but are rarely used due to the cost of the animals, the maintenance involved and ethical concerns. The most common species currently used in HE models are rats and mice, mainly due to the accessibility of molecular and anatomical studies and the availability of literature on behavioral, pathological and biochemical methods and findings. Other advantages of these species are the complete characterization of the genome, easy availability of antibodies and molecular probes and low costs for obtaining and maintaining the animals[44].
According to the HE guidelines established by ISHEN, an animal model of HE in ALF has to essentially reproduce the clinical picture to facilitate staging of encephalopathy and to show the progression of symptoms, including brain edema and its complications (i.e., intracranial hypertension and brain herniation). The model should also be capable of being reversed, with high concentrations of ammonia and glutamine in the brain, classical hepatic and brain pathology and minimal hazards to personnel from toxins and infectious agents. All animal models of ALF produce hypothermia and hypoglycemia, making it essential to control the temperature and glucose levels and to provide supportive care to the animals. A brief description of ALF models is presented in Table 2.
| Species used | Main findings | |
| Surgical models | ||
| Hepatic devascularization | Rat, rabbit, pig | Increased AST, hypoglycemia, lethargy and coma |
| Hepatectomy | Rat, pig | Increased AST, TNF, PT, NH3, lactate, hypoglycemia, hepatic necrosis |
| Pharmacological models | ||
| Galactosamine (IP, IV, SC) | Rat, rabbit, guinea pig | Increased AST, PT, NH3, hepatic necrosis |
| Acetaminophen (IP, IV, SC, oral) | Rat, dog, pig | Increased AST, NH3, bilirubin, hypoglycemia, metabolic acidosis, centrilobular coagulative necrosis |
| Thioacetamide (IP, oral) | Rat | Increased AST, PT, NH3, metabolic acidosis, centrilobular necrosis |
| Azoxymethane (IP, SC) | Mouse | Increased AST, NH3 and bilirubin, hepatic necrosis |
Several hepatotoxins have been extensively used in the development of animal models of ALF[46]. The main hepatotoxic substances used to cause ALF in animals include galactosamine, acetaminophen, azoxymethane (AOM) and thioacetamide (TAA)[44]. Although the administration of carbon tetrachloride (CCL4) has been primarily described as a type A HE model, the ALF and behavioral changes in this model are not commonly observed. However, the ability of this model to induce liver cirrhosis makes it a valuable tool for studying type C HE[44]. Thus, in the current review, the CCL4 model will be discussed as a Type C HE model.
Thioacetamide: Until 1943, TAA was used as fungicide in orange crops. In 1948, it was discovered that chronic administration of the substance led to liver cirrhosis and hepatocellular carcinomas. TAA causes hepatocellular necrosis after biotransformation to an active metabolite via the flavin adenine dinucleotide monooxygenase pathway, resulting in the formation of TAA-S-oxide[47]. TAA reduces antioxidant activity and enhances lipid peroxidation in the liver, leading to oxidative stress and cellular necrosis[48]. The most common forms of administration of TAA are oral (in drinking water) and intraperitoneal injection[49]. TAA has been extensively used to induce ALF in rats and mice, producing encephalopathy, metabolic acidosis, high transaminases, abnormal coagulation and histological centrilobular necrosis. The TAA model of HE shows good reproducibility and well-described hepatic and cerebral changes[50-52]. Chronic administration can produce hepatic cirrhosis[49,53,54].
D-Galactosamine: D-Galactosamine (D-Gal) is an amino sugar that is metabolized in the liver, causing consumption of uridine nucleotides and blockade of transcriptional factors in the liver. The hepatic failure resulting from D-Gal administration has been, at least in part, associated with the production of uridine-containing compounds during hepatic biotransformation[55]. The administration of D-Gal in rats was described by Keppler et al[56] as an acute hepatic failure model that induced encephalopathy and increased aspartate transaminase, prothrombin time, ammonia and intracranial pressure. The same model was described by Blitzer et al[57] in rabbits and by Sielaff et al[58] and Diaz Buxo et al[59] in dogs.
There are differences in species susceptibility. Mice are resistant until high doses are administered, while rats are sensitive to D-Gal-induced hepatic failure[60]. D-Gal and acetaminophen models cause liver failure, although the development of HE is variable. Both models are difficult to reproduce and have poorly characterized cerebral pathology[44].
Acetaminophen: Acetaminophen is known as APAP (in the United States) and Paracetamol (in Europe and other areas of the world). Acetaminophen has been widely used as an antipyretic and/or analgesic since 1955, particularly because it is easily accessible in various formulations as an over-the-counter medication. However, acetaminophen is one of the most common causes of ALF, accounting for more than 60% of all cases in the United States[61].
The metabolism of APAP occurs in the liver. Under normal circumstances, the medication undergoes biotransformation by a combination of glucuronidation and sulfation and is then excreted by the kidneys. When excessive amounts of acetaminophen are administered, these metabolic pathways are saturated, and APAP is metabolized by the P450 cytochrome oxidase enzyme system, leading to the formation of a toxic electrophile, N-acetyl-p-benzoquinoneimine (NAPQI). NAPQI produces cell injury unless conjugated with endogenous glutathione. NAPQI is supposed to disrupt mitochondrial calcium flux, resulting in cell damage by the production of free radical oxygen species, hydroxyl radicals, nitrites and nitrates. The cascade is amplified by the activation of Kupffer cells and the release of cytotoxic mediators (e.g., cytokines and free radicals), leading to apoptosis and cell necrosis[62-65].
Miller et al[66] reported a model of APAP-induced liver failure in pigs. Animals exhibited metabolic acidosis, encephalopathy, coma, increased transaminase levels and histological evidence of severe centrilobular coagulative necrosis. However, changes produced by APAP administration were variable according to animal species used, as was described by Gazzard et al[67], who showed a variable clinical outcome in greyhound dogs.
Later, Francavilla et al[68] induced ALF by APAP administration for the first time in beagles. The authors compared the routes of administration: intravenous, intramuscular and subcutaneous. APAP toxicity was very variable in different animal models, and the incidence of sudden death due to methemoglobinemia was frequently observed. This drug exhibited cardiotoxicity and nephrotoxicity, which was associated with acute lung injury[69-71].
Azoxymethane: AOM is an active metabolite of the cycad palm nut found on the island of Guam. AOM is a potent hepatotoxin that induces ALF in mice in a dose-dependent manner. Liver toxicity has also been reported in humans, livestock and rats following the ingestion of Guam cycad palm nuts due to AOM toxicity. The AOM model was first described as a model of hepatotoxin-induced liver failure and HE by Matkowskyj et al[72]. This model leads to encephalopathy, cerebral edema, elevated brain ammonia and unbalanced amino acid levels. This model also shows characteristic pathologic aspects[28].
The AOM model generates microvesicular steatosis, dilation of hepatic sinusoids, and hepatocyte necrosis in addition to elevations in serum transaminases and bile acids, with the largest increases observed when mice progressed toward coma[73].
The surgical models can be divided into variations of partial and total hepatectomy and partial and complete devascularization of the liver[46].
Hepatic devascularization: Rappaport was the first to describe a devascularization model, in 1953[74]. The model is usually produced by a portocaval anastomosis (PCA) with subsequent hepatic artery ligation (HAL). ALF can be reversible by the occlusion of the hepatic artery for only a short period of time. There is no blood lost. The presence of necrotic hepatic tissue is comparable to ALF in humans[45]. Hypoxic insult results in dysfunction of the mitochondrial respiratory chain, which, in turn, reduces ATP levels due to impaired oxidative phosphorylation, interferes with the intracellular calcium homeostasis and activates enzymes responsible for protein, lipid and DNA damage. After hypoxic injury, the reperfusion process induces more damage in liver tissue[75-77].
Hepatectomy: Hepatectomy is considered a model of postoperative liver failure (POLF) that can accurately reproduce all neurological and metabolic changes as a consequence of extensive liver resections in humans[78]. Almost 100% survival and intense regeneration occur in partial hepatectomy that removes approximately 70% of the liver in rats and pigs[79-81]. In contrast, progressive necrosis follows partial resections of the liver, as reported by Panis et al[82]. Among other disadvantages of partial hepatectomy are the lack of consistency of injury degree and the increased intraoperative blood loss. Additionally, the surgery often hampers the animal’s capability to immediately restore drink and food consumption, which may lead to severe hypoglycemia and death[83].
The altered physiological state is attributed to increased total blood flow through the remnant liver tissue, leading to flow injury and damage to sinusoidal endothelial cells, activation of Kupffer cells and release of inflammatory cytokines[84]. This model is also associated with increased levels of aspartate transaminase (AST), tumor necrosis factor (TNF) and hypoglycemia[85].
The “anhepatic” model is made by the total removal of the liver. In clinical practice, total hepatectomy has been performed only in cases of very severe ALF, pending the arrival of a donor liver, which follows the removal of a “toxic” organ[86,87]. Overall, the anhepatic model seems not to be suitable for evaluating therapies as an artificial liver support for ALF. Moreover, due to the lack of the liver, this model has very poor reversibility[46].
An ideal model of HE classified as type B or C should have some essential features, including a precipitant factor and neuropathological findings of HE, such as symptoms of encephalopathy, ranging from MHE to coma. Additionally, increased brain ammonia/glutamine along with low-grade brain edema and Alzheimer Type II astrocytosis at advanced stages of HE might also be observed. These models should exhibit clinical responses to established treatments[29]. Table 3 displays the animal models of HE associated with portosystemic shunting (type B) and chronic liver disease (type C).
| Experimental model | Animal species | Biological findings | Clinical Signs |
| Portacaval anastomosis | Rats, dog, rabbit, pig | Increased brain ammonia/glutamine | Altered circadian cycle, hypokinesia, reduced memory and learning ability |
| Congenital portacaval shunts | Dogs, cats | Hyperammonemia | Hepatic dysfunction, psychomotor dysfunction, motor signs |
| Graded portal vein stenosis | Rats | Increased brain ammonia/glutamine | Minimal hepatic encephalopathy, loss of activity, altered circadian cycle |
| Carbon tetrachloride (CCl4) | Rats, mice | Generation of free radicals, lipoperoxidation, tissue fibrosis, increased hepatic membrane permeability | Hepatic failure, motor activity dysfunction |
| Bile duct ligation | Rats | Bacterial translocation, immune system dysfunction, hyperammonemia | Liver failure, portal hypertension, decreased locomotor activities due to low-grade encephalopathy |
As previously mentioned, animal models of type B HE are based on portal-systemic shunting and include the following approaches.
Portacaval anastomosis: PCA is the most common animal model used in the study of chronic HE. The basis of this model is the creation of a portal-systemic shunt (end-to-side PCA) that mimics the situation induced in cirrhosis by collateral circulation[45].
It is known that the deviation of portal blood decreases the total hepatic blood flow, depriving the liver of oxygen and hepatotrophic factors from the portal vein that are necessary for metabolic processes. These phenomena induce hepatic parenchyma atrophy. Among other factors, the high levels of ammonia, as a consequence of reduced hepatic metabolism, contribute to HE. The severity of neurological manifestations depends on the intensity of liver injury. Animals submitted to PCA have increased brain ammonia/glutamine, altered circadian cycle, hypokinesia and reduced memory and learning ability[88].
Congenital portacaval shunts: Dogs and cats with congenital portacaval shunts are considered naturally occurring models of type B HE. These animals develop psychomotor dysfunction, abnormal motor signs, altered day–night rhythms, hyperammonemia, and hepatic dysfunction[89].
Graded portal vein stenosis: Graded portal vein stenosis provides a MHE model in rats. This procedure is easier to perform than the end-to-side portacaval anastomosis. Alterations associated with this model are hypoactivity, altered circadian cycle and increased brain ammonia/glutamine[44].
Animal models of type C HE should lead to decompensated liver cirrhosis. It is worth noting that there is currently no ideal animal model of type C HE[44]. Some models that resemble human hepatic cirrhosis have been employed, as described below.
CCl4: CCl4 was extensively used in the 1970s as a model of acute hepatic failure, but the species variation was significant, and it was difficult to reproduce. It has been used more recently in the study of liver cirrhosis[90,91]. CCl4 is metabolized by cytochrome P-450, generating free radicals, causing lipoperoxidation, increased hepatic membrane permeability, tissue fibrosis and hepatic failure. This model does not seem to produce many behavioral changes, except in cases of advanced cirrhosis[92]. It is used in the study of astrocytic response at the level of RNA synthesis. Inconsistent lesions animal to animal may represent a major disadvantage. Moreover, the presence of ascites may limit neurobehavioral assessments[44].
Bile duct ligation: The ligation of the common bile duct ligation (BDL) induces a reproducible model of biliary cirrhosis in rats, leading to liver failure, portal hypertension, translocation of bacteria and immune system dysfunction. BDL rats show hyperammonemia and decreased motor activities, but only low-grade encephalopathy[45,93-95]. It is possible to reproduce the human neuropathology of type C HE, Alzheimer type II astrocytosis, altered brain osmolytes, low-grade brain edema, inflammation and motor activity deficits by feeding BDL rats with ammonium salts. Of note, by giving ammonium salts to BDL rats, it is possible to obtain a model of acute-on-chronic HE[96].
These models are usually limited to rats and mice and are used to study the effects of hyperammonemia on brain function, in the absence of liver damage or portacaval shunting. They are produced by means of the administration of high-ammonia diets, parenteral infusion of ammonia or urease treatment. These models are inexpensive and easily reproducible and result in animals with impaired memory and learning skills[44]. However, the disadvantages of these models include the time involved to produce alterations and their unsuitability for long-term studies[45].
HE is a very severe complication in the context of liver failure. HE can be subdivided according to the type of hepatic injury, namely, type A, which results from ALF, type B, which is associated with a portosystemic shunting without intrinsic liver disease, and type C, which is due to chronic liver disease. The pathophysiology of HE has two major factors, namely, increased ammonia levels and neuroinflammation.
Preclinical models have been very useful in investigating the mechanisms of HE and in evaluating novel therapeutic approaches. The most used animal species are rats and mice. Experimental models of ALF (type A) include surgical procedures (hepatectomy and hepatic devascularization) and the administration of hepatotoxic medications. Surgical models resemble postoperative liver failure in humans, whereas pharmacological models are similar to adverse drug reactions due to hepatotoxicity.
Ideally, models of HE associated with portosystemic shunting (type B) and due to chronic liver disease (type C) must exhibit liver cirrhosis, a precipitant factor, neuropathological and neurochemical alterations. These models are generally surgically induced lesions in liver tissue, which evolve to hepatic cirrhosis.
In conclusion, preclinical models have allowed the comprehension of the pathways related to neurological damage as a consequence of acute and chronic liver injury, resulting in the identification of potential therapeutic targets.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Brazil
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): D, D
Grade E (Poor): 0
P- Reviewer: Dourakis S, Weiss N, Silva M S- Editor: Dou Y L- Editor: A E- Editor: Zhang YL
| 1. | Butterworth RF. The concept of "the inflamed brain" in acute liver failure: mechanisms and new therapeutic opportunities. Metab Brain Dis. 2016;31:1283-1287. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 2. | Wang AJ, Peng AP, Li BM, Gan N, Pei L, Zheng XL, Hong JB, Xiao HY, Zhong JW, Zhu X. Natural history of covert hepatic encephalopathy: An observational study of 366 cirrhotic patients. World J Gastroenterol. 2017;23:6321-6329. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 23] [Cited by in F6Publishing: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 3. | Wong RJ, Gish RG, Ahmed A. Hepatic encephalopathy is associated with significantly increased mortality among patients awaiting liver transplantation. Liver Transpl. 2014;20:1454-1461. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 23] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 4. | Yuan LT, Chuah SK, Yang SC, Liang CM, Wu CK, Tai WC, Hung TH, Nguang SH, Wang JW, Tseng KL, Ku MK, Hsu PI, Wu DC, Hsu CN. Multiple bacterial infections increase the risk of hepatic encephalopathy in patients with cirrhosis. PLoS One. 2018;13:e0197127. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 5. | Flamm SL. Considerations for the cost-effective management of hepatic encephalopathy. Am J Manag Care. 2018;24:S51-S61. [PubMed] [Cited in This Article: ] |
| 6. | Roggeri DP, Roggeri A, Rossi E, Cinconze E, Gasbarrini A, Monici Preti P, De Rosa M. Overt hepatic encephalopathy in Italy: clinical outcomes and healthcare costs. Hepat Med. 2015;7:37-42. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 7. | Vilstrup H, Amodio P, Bajaj J, Cordoba J, Ferenci P, Mullen KD, Weissenborn K, Wong P. Hepatic encephalopathy in chronic liver disease: 2014 Practice Guideline by the American Association for the Study of Liver Diseases and the European Association for the Study of the Liver. Hepatology. 2014;60:715-735. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1158] [Cited by in F6Publishing: 1270] [Article Influence: 127.0] [Reference Citation Analysis (1)] |
| 8. | Stepanova M, Mishra A, Venkatesan C, Younossi ZM. In-hospital mortality and economic burden associated with hepatic encephalopathy in the United States from 2005 to 2009. Clin Gastroenterol Hepatol. 2012;10:1034-41.e1. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 168] [Cited by in F6Publishing: 174] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 9. | Bernal W, Wendon J. Acute Liver Failure. New Engl J Med. 2013;2525. [DOI] [Cited in This Article: ] [Cited by in Crossref: 736] [Cited by in F6Publishing: 764] [Article Influence: 69.5] [Reference Citation Analysis (2)] |
| 10. | Ortiz M, Jacas C, Córdoba J. Minimal hepatic encephalopathy: diagnosis, clinical significance and recommendations. J Hepatol. 2005;42 Suppl:S45-S53. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 182] [Cited by in F6Publishing: 195] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 11. | Basu PP, Shah NJ. Clinical and Neurologic Manifestation of Minimal Hepatic Encephalopathy and Overt Hepatic Encephalopathy. Clin Liver Dis. 2015;19:461-472. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 12. | Felipo V. Hepatic encephalopathy: effects of liver failure on brain function. Nat Rev Neurosci. 2013;14:851-858. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 229] [Cited by in F6Publishing: 252] [Article Influence: 22.9] [Reference Citation Analysis (0)] |
| 13. | Liere V, Sandhu G, DeMorrow S. Recent advances in hepatic encephalopathy. F1000Res. 2017;6:1637. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 14. | Parekh PJ, Balart LA. Ammonia and Its Role in the Pathogenesis of Hepatic Encephalopathy. Clin Liver Dis. 2015;19:529-537. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 15. | Romero-Gómez M, Jover M, Galán JJ, Ruiz A. Gut ammonia production and its modulation. Metab Brain Dis. 2009;24:147-157. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 66] [Cited by in F6Publishing: 59] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 16. | Romero-Gómez M, Ramos-Guerrero R, Grande L, de Terán LC, Corpas R, Camacho I, Bautista JD. Intestinal glutaminase activity is increased in liver cirrhosis and correlates with minimal hepatic encephalopathy. J Hepatol. 2004;41:49-54. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 76] [Cited by in F6Publishing: 80] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 17. | Romero-Gómez M, Montagnese S, Jalan R. Hepatic encephalopathy in patients with acute decompensation of cirrhosis and acute-on-chronic liver failure. J Hepatol. 2015;62:437-447. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 150] [Cited by in F6Publishing: 152] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 18. | Liang C, Du T, Zhou J, Verkhratsky A, Peng L. Ammonium increases Ca(2+) signalling and up-regulates expression of TRPC1 gene in astrocytes in primary cultures and in the in vivo brain. Neurochem Res. 2014;39:2127-2135. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 19] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 19. | Wang F, Du T, Liang C, Verkhratsky A, Peng L. Ammonium increases Ca(2+) signalling and upregulates expression of Cav1.2 gene in astrocytes in primary cultures and in the in vivo brain. Acta Physiol (Oxf). 2015;214:261-274. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 20. | Sobczyk K, Jördens MS, Karababa A, Görg B, Häussinger D. Ephrin/Ephrin receptor expression in ammonia-treated rat astrocytes and in human cerebral cortex in hepatic encephalopathy. Neurochem Res. 2015;40:274-283. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 19] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 21. | Karababa A, Görg B, Schliess F, Häussinger D. O-GlcNAcylation as a novel ammonia-induced posttranslational protein modification in cultured rat astrocytes. Metab Brain Dis. 2014;29:975-982. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 22. | Ong JP, Aggarwal A, Krieger D, Easley KA, Karafa MT, Van Lente F, Arroliga AC, Mullen KD. Correlation between ammonia levels and the severity of hepatic encephalopathy. Am J Med. 2003;114:188-193. [PubMed] [Cited in This Article: ] |
| 23. | Shawcross DL, Wright G, Olde Damink SW, Jalan R. Role of ammonia and inflammation in minimal hepatic encephalopathy. Metab Brain Dis. 2007;22:125-138. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 186] [Cited by in F6Publishing: 188] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 24. | Acharya SK, Bhatia V, Sreenivas V, Khanal S, Panda SK. Efficacy of L-ornithine L-aspartate in acute liver failure: a double-blind, randomized, placebo-controlled study. Gastroenterology. 2009;136:2159-2168. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 148] [Cited by in F6Publishing: 116] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 25. | Jalan R. Acute liver failure: current management and future prospects. J Hepatol. 2005;42 Suppl:S115-S123. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 70] [Cited by in F6Publishing: 72] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 26. | Sharma P, Sharma BC, Sarin SK. Predictors of nonresponse to lactulose in patients with cirrhosis and hepatic encephalopathy. Eur J Gastroenterol Hepatol. 2010;22:526-531. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 27. | Rockey DC, Vierling JM, Mantry P, Ghabril M, Brown RS, Alexeeva O, Zupanets IA, Grinevich V, Baranovsky A, Dudar L, Fadieienko G, Kharchenko N, Klaryts'ka I, Morozov V, Grewal P, McCashland T, Reddy KG, Reddy KR, Syplyviy V, Bass NM, Dickinson K, Norris C, Coakley D, Mokhtarani M, Scharschmidt BF; HALT-HE Study Group. Randomized, double-blind, controlled study of glycerol phenylbutyrate in hepatic encephalopathy. Hepatology. 2014;59:1073-1083. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 112] [Cited by in F6Publishing: 110] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 28. | Bélanger M, Côté J, Butterworth RF. Neurobiological characterization of an azoxymethane mouse model of acute liver failure. Neurochem Int. 2006;48:434-440. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 52] [Cited by in F6Publishing: 58] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 29. | Butterworth RF. Hepatic encephalopathy: a central neuroinflammatory disorder? Hepatology. 2011;53:1372-1376. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 153] [Cited by in F6Publishing: 164] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 30. | Jiang W, Desjardins P, Butterworth RF. Direct evidence for central proinflammatory mechanisms in rats with experimental acute liver failure: protective effect of hypothermia. J Cereb Blood Flow Metab. 2009;29:944-952. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 95] [Cited by in F6Publishing: 96] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 31. | Zemtsova I, Görg B, Keitel V, Bidmon HJ, Schrör K, Häussinger D. Microglia activation in hepatic encephalopathy in rats and humans. Hepatology. 2011;54:204-215. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 114] [Cited by in F6Publishing: 120] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 32. | Jiang W, Desjardins P, Butterworth RF. Cerebral inflammation contributes to encephalopathy and brain edema in acute liver failure: protective effect of minocycline. J Neurochem. 2009;109:485-493. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 101] [Cited by in F6Publishing: 104] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 33. | Blank T, Prinz M. Microglia as modulators of cognition and neuropsychiatric disorders. Glia. 2013;61:62-70. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 122] [Cited by in F6Publishing: 131] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 34. | Rodríguez JJ, Verkhratsky A. Neuroglial roots of neurodegenerative diseases? Mol Neurobiol. 2011;43:87-96. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 55] [Cited by in F6Publishing: 59] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 35. | Wright G, Shawcross D, Olde Damink SW, Jalan R. Brain cytokine flux in acute liver failure and its relationship with intracranial hypertension. Metab Brain Dis. 2007;22:375-388. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 93] [Cited by in F6Publishing: 99] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 36. | Bémeur C, Qu H, Desjardins P, Butterworth RF. IL-1 or TNF receptor gene deletion delays onset of encephalopathy and attenuates brain edema in experimental acute liver failure. Neurochem Int. 2010;56:213-215. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 83] [Cited by in F6Publishing: 85] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 37. | Faleiros BE, Miranda AS, Campos AC, Gomides LF, Kangussu LM, Guatimosim C, Camargos ER, Menezes GB, Rachid MA, Teixeira AL. Up-regulation of brain cytokines and chemokines mediates neurotoxicity in early acute liver failure by a mechanism independent of microglial activation. Brain Res. 2014;1578:49-59. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 38. | McMillin M, Grant S, Frampton G, Andry S, Brown A, DeMorrow S. Fractalkine suppression during hepatic encephalopathy promotes neuroinflammation in mice. J Neuroinflammation. 2016;13:198. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 39. | Agusti A, Hernández-Rabaza V, Balzano T, Taoro-Gonzalez L, Ibañez-Grau A, Cabrera-Pastor A, Fustero S, Llansola M, Montoliu C, Felipo V. Sildenafil reduces neuroinflammation in cerebellum, restores GABAergic tone, and improves motor in-coordination in rats with hepatic encephalopathy. CNS Neurosci Ther. 2017;23:386-394. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 40. | Hernandez-Rabaza V, Agusti A, Cabrera-Pastor A, Fustero S, Delgado O, Taoro-Gonzalez L, Montoliu C, Llansola M, Felipo V. Sildenafil reduces neuroinflammation and restores spatial learning in rats with hepatic encephalopathy: underlying mechanisms. J Neuroinflammation. 2015;12:195. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 52] [Cited by in F6Publishing: 54] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 41. | Hernandez-Rabaza V, Cabrera-Pastor A, Taoro-Gonzalez L, Gonzalez-Usano A, Agusti A, Balzano T, Llansola M, Felipo V. Neuroinflammation increases GABAergic tone and impairs cognitive and motor function in hyperammonemia by increasing GAT-3 membrane expression. Reversal by sulforaphane by promoting M2 polarization of microglia. J Neuroinflammation. 2016;13:83. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 93] [Cited by in F6Publishing: 89] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 42. | McMillin M, Frampton G, Thompson M, Galindo C, Standeford H, Whittington E, Alpini G, DeMorrow S. Neuronal CCL2 is upregulated during hepatic encephalopathy and contributes to microglia activation and neurological decline. J Neuroinflammation. 2014;11:121. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 58] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 43. | Hernández-Rabaza V, Cabrera-Pastor A, Taoro-González L, Malaguarnera M, Agustí A, Llansola M, Felipo V. Hyperammonemia induces glial activation, neuroinflammation and alters neurotransmitter receptors in hippocampus, impairing spatial learning: reversal by sulforaphane. J Neuroinflammation. 2016;13:41. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 69] [Cited by in F6Publishing: 93] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 44. | Butterworth RF, Norenberg MD, Felipo V, Ferenci P, Albrecht J, Blei AT, Members of the ISHEN Commission on Experimental Models of HE. Experimental models of hepatic encephalopathy: ISHEN guidelines. Liver Int. 2009;29:783-788. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 225] [Cited by in F6Publishing: 228] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 45. | Jover R, Madaria E, Felipo V, Rodrigo R, Candela A, Compañ A. Animal models in the study of episodic hepatic encephalopathy in cirrhosis. Metab Brain Dis. 2005;20:399-408. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 46. | Rahman TM, Hodgson HJ. Animal models of acute hepatic failure. Int J Exp Pathol. 2000;81:145-157. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 129] [Cited by in F6Publishing: 130] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 47. | Chieli E, Malvaldi G. Role of the microsomal FAD-containing monooxygenase in the liver toxicity of thioacetamide S-oxide. Toxicology. 1984;31:41-52. [PubMed] [Cited in This Article: ] |
| 48. | Hajovsky H, Hu G, Koen Y, Sarma D, Cui W, Moore DS, Staudinger JL, Hanzlik RP. Metabolism and toxicity of thioacetamide and thioacetamide S-oxide in rat hepatocytes. Chem Res Toxicol. 2012;25:1955-1963. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 130] [Cited by in F6Publishing: 133] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 49. | Fontana L, Moreira E, Torres MI, Fernández MI, Ríos A, Sánchez de Medina F, Gil A. Serum amino acid changes in rats with thioacetamide-induced liver cirrhosis. Toxicology. 1996;106:197-206. [PubMed] [Cited in This Article: ] |
| 50. | Bruck R, Oren R, Shirin H, Aeed H, Papa M, Matas Z, Zaidel L, Avni Y, Halpern Z. Hypothyroidism minimizes liver damage and improves survival in rats with thioacetamide induced fulminant hepatic failure. Hepatology. 1998;27:1013-1020. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 39] [Cited by in F6Publishing: 38] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 51. | Peeling J, Shoemaker L, Gauthier T, Benarroch A, Sutherland GR, Minuk GY. Cerebral metabolic and histological effects of thioacetamide-induced liver failure. Am J Physiol. 1993;265:G572-G578. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 19] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 52. | Zimmermann C, Ferenci P, Pifl C, Yurdaydin C, Ebner J, Lassmann H, Roth E, Hörtnagl H. Hepatic encephalopathy in thioacetamide-induced acute liver failure in rats: characterization of an improved model and study of amino acid-ergic neurotransmission. Hepatology. 1989;9:594-601. [PubMed] [Cited in This Article: ] |
| 53. | Nakano A, Kanda T, Abe H. Liver and Bone. J Gastroen Hepatol. 1996;1143. [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 18] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 54. | Petermann H, Heymann S, Vogl S, Dargel R. Phagocytic function and metabolite production in thioacetamide-induced liver cirrhosis: a comparative study in perfused livers and cultured Kupffer cells. J Hepatol. 1996;24:468-477. [PubMed] [Cited in This Article: ] |
| 55. | Takahashi N, Ishizuya T. Modification of D-galactosamine-induced liver damage in rats by intravenous injection of newly isolated intact splenic cells. Int J Tissue React. 1990;12:39-45. [PubMed] [Cited in This Article: ] |
| 56. | Keppler D, Lesch R, Reutter W, Decker K. Experimental hepatitis induced by D-galactosamine. Exp Mol Pathol. 1968;9:279-290. [PubMed] [Cited in This Article: ] |
| 57. | Blitzer BL, Waggoner JG, Jones EA, Gralnick HR, Towne D, Butler J, Weise V, Kopin IJ, Walters I, Teychenne PF, Goodman DG, Berk PD. A model of fulminant hepatic failure in the rabbit. Gastroenterology. 1978;74:664-671. [PubMed] [Cited in This Article: ] |
| 58. | Sielaff TD, Hu MY, Rollins MD, Bloomer JR, Amiot B, Hu WS, Cerra FB. An anesthetized model of lethal canine galactosamine fulminant hepatic failure. Hepatology. 1995;21:796-804. [PubMed] [Cited in This Article: ] |
| 59. | Diaz-Buxo JA, Blumenthal S, Hayes D, Gores P, Gordon B. Galactosamine-induced fulminant hepatic necrosis in unanesthetized canines. Hepatology. 1997;25:950-957. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 37] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 60. | Leist M, Gantner F, Künstle G, Bohlinger I, Tiegs G, Bluethmann H, Wendel A. The 55-kD tumor necrosis factor receptor and CD95 independently signal murine hepatocyte apoptosis and subsequent liver failure. Mol Med. 1996;2:109-124. [PubMed] [Cited in This Article: ] |
| 61. | Lee KK, Wu DB, Chow PY, Lee VW, Li H. Economic analysis between entecavir and lamivudine for the treatment of chronic hepatitis B in Hong Kong. J Gastroenterol Hepatol. 2012;27:1167-1174. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 11] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 62. | Black M. Acetaminophen hepatotoxicity. Gastroenterology. 1980;78:382-392. [PubMed] [Cited in This Article: ] |
| 63. | Boyd EM, Bereczky GM. Liver necrosis from paracetamol. Br J Pharmacol Chemother. 1966;26:606-614. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 80] [Cited by in F6Publishing: 105] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 64. | Boyd EM, Hogan SE. The chronic oral toxicity of paracetamol at the range of the LD50 (100 days) in albino rats. Can J Physiol Pharmacol. 1968;46:239-245. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 20] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 65. | Davidson DG, Eastham WN. Acute liver necrosis following overdose of paracetamol. Br Med J. 1966;2:497-499. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 359] [Cited by in F6Publishing: 354] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 66. | Miller DJ, Hickman R, Fratter R, Terblanche J, Saunders SJ. An animal model of fulminant hepatic failure: a feasibility study. Gastroenterology. 1976;71:109-113. [PubMed] [Cited in This Article: ] |
| 67. | Gazzard BG, Hughes RD, Mellon PJ, Portmann B, Williams R. A dog model of fulminant hepatic failure produced by paracetamol administration. Br J Exp Pathol. 1975;56:408-411. [PubMed] [Cited in This Article: ] |
| 68. | Francavilla A, Makowka L, Polimeno L, Barone M, Demetris J, Prelich J, Van Thiel DH, Starzl TE. A dog model for acetaminophen-induced fulminant hepatic failure. Gastroenterology. 1989;96:470-478. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 43] [Cited by in F6Publishing: 36] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 69. | Baudouin SV, Howdle P, O'Grady JG, Webster NR. Acute lung injury in fulminant hepatic failure following paracetamol poisoning. Thorax. 1995;50:399-402. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 77] [Cited by in F6Publishing: 76] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 70. | Eguia L, Materson BJ. Acetaminophen-related acute renal failure without fulminant liver failure. Pharmacotherapy. 1997;17:363-370. [PubMed] [Cited in This Article: ] |
| 71. | Fujimura H, Kawasaki N, Tanimoto T, Sasaki H, Suzuki T. Effects of acetaminophen on the ultrastructure of isolated rat hepatocytes. Exp Toxicol Pathol. 1995;47:345-351. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 13] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 72. | Matkowskyj KA, Marrero JA, Carroll RE, Danilkovich AV, Green RM, Benya RV. Azoxymethane-induced fulminant hepatic failure in C57BL/6J mice: characterization of a new animal model. Am J Physiol. 1999;277:G455-G462. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 59] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 73. | Grant S, McMillin M, Frampton G, Petrescu AD, Williams E, Jaeger V, Kain J, DeMorrow S. Direct Comparison of the Thioacetamide and Azoxymethane Models of Type A Hepatic Encephalopathy in Mice. Gene Expr. 2018;18:171-185. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 74. | Rappaport AM, Macdonald MH, Borowy ZJ. Hepatic coma following ischemia of the liver. Surg Gynecol Obstet. 1953;97:748-762. [PubMed] [Cited in This Article: ] |
| 75. | Borghi-Scoazec G, Scoazec JY, Durand F, Bernuau J, Belghiti J, Feldmann G, Henin D, Degott C. Apoptosis after ischemia-reperfusion in human liver allografts. Liver Transpl Surg. 1997;3:407-415. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 97] [Cited by in F6Publishing: 97] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 76. | Gasbarrini A, Colantoni A, Di Campli C, De Notariis S, Masetti M, Iovine E, Mazziotti A, Massari I, Gasbarrini G, Pola P, Bernardi M. Intermittent anoxia reduces oxygen free radicals formation during reoxygenation in rat hepatocytes. Free Radic Biol Med. 1997;23:1067-1072. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 12] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 77. | Shirasugi N, Wakabayashi G, Shimazu M, Oshima A, Shito M, Kawachi S, Karahashi T, Kumamoto Y, Yoshida M, Kitajima M. Up-regulation of oxygen-derived free radicals by interleukin-1 in hepatic ischemia/reperfusion injury. Transplantation. 1997;64:1398-1403. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 59] [Cited by in F6Publishing: 62] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 78. | Arkadopoulos N, Defterevos G, Nastos C, Papalois A, Kalimeris K, Papoutsidakis N, Kampouroglou G, Kypriotis D, Pafiti A, Kostopanagiotou G, Smyrniotis V. Development of a porcine model of post-hepatectomy liver failure. J Surg Res. 2011;170:e233-e242. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 31] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 79. | Emond JC, Mayes JT, Rouch DA, Thistlethwaite JR, Broelsch CE. Experience with radical resection in the management of proximal bile duct cancer. HPB Surg. 1989;1:297-305; discussion 305-7. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 6] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 80. | Kahn D, Hickman R, Terblanche J, von Sommoggy S. Partial hepatectomy and liver regeneration in pigs--the response to different resection sizes. J Surg Res. 1988;45:176-180. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 47] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 81. | Wang X, Andersson R, Ding J, Norgren L, Bengmark S. Reticuloendothelial system function following acute liver failure induced by 90% hepatectomy in the rat. HPB Surg. 1993;6:151-162. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 16] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 82. | Panis Y, McMullan DM, Emond JC. Progressive necrosis after hepatectomy and the pathophysiology of liver failure after massive resection. Surgery. 1997;121:142-149. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 145] [Cited by in F6Publishing: 154] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 83. | Li J, Xiao J, So KF. Research progress on hepatic encephalopathy: animal models and disease mechanisms. Abdomen. 2015;2. [DOI] [Cited in This Article: ] [Cited by in Crossref: 2] [Cited by in F6Publishing: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 84. | Kahn D, van Hoorn-Hickman R, Terblanche J. Liver blood flow after partial hepatectomy in the pig. J Surg Res. 1984;37:290-294. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 41] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 85. | Roger V, Balladur P, Honiger J, Delelo R, Baudrimont M, Robert A, Calmus Y, Capeau J, Nordlinger B. [A good model of acute hepatic failure: 95% hepatectomy. Treatment by transplantation of hepatocytes]. Chirurgie. 1996;121:470-473. [PubMed] [Cited in This Article: ] |
| 86. | Ejlersen E, Larsen FS, Pott F, Gyrtrup HJ, Kirkegaard P, Secher NH. Hepatectomy corrects cerebral hyperperfusion in fulminant hepatic failure. Transplant Proc. 1994;26:1794-1795. [PubMed] [Cited in This Article: ] |
| 87. | Rozga J, Podesta L, LePage E, Hoffman A, Morsiani E, Sher L, Woolf GM, Makowka L, Demetriou AA. Control of cerebral oedema by total hepatectomy and extracorporeal liver support in fulminant hepatic failure. Lancet. 1993;342:898-899. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 122] [Cited by in F6Publishing: 118] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 88. | Hindfelt B, Plum F, Duffy TE. Effect of acute ammonia intoxication on cerebral metabolism in rats with portacaval shunts. J Clin Invest. 1977;59:386-396. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 345] [Cited by in F6Publishing: 359] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 89. | Maddison JE, Dodd PR, Morrison M, Johnston GA, Farrell GC. Plasma GABA, GABA-like activity and the brain GABA-benzodiazepine receptor complex in rats with chronic hepatic encephalopathy. Hepatology. 1987;7:621-628. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 71] [Cited by in F6Publishing: 72] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 90. | Das PK, Chopra P, Nayak NC. Hepatocellular tolerance to carbon tetrachloride induced injury in the rat: a study of its nature and possible mode of evolution. Exp Mol Pathol. 1974;21:218-236. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7] [Cited by in F6Publishing: 7] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 91. | Shi Z, Wakil AE, Rockey DC. Strain-specific differences in mouse hepatic wound healing are mediated by divergent T helper cytokine responses. Proc Natl Acad Sci U S A. 1997;94:10663-10668. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 240] [Cited by in F6Publishing: 244] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 92. | Mullen KD, Birgisson S, Gacad RC, Conjeevaram H. Animal models of hepatic encephalopathy and hyperammonemia. Adv Exp Med Biol. 1994;368:1-10. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 10] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 93. | Chan CY, Huang SW, Wang TF, Lu RH, Lee FY, Chang FY, Chu CJ, Chen YC, Chan CC, Huang HC, Lee SD. Lack of detrimental effects of nitric oxide inhibition in bile duct-ligated rats with hepatic encephalopathy. Eur J Clin Invest. 2004;34:122-128. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 19] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 94. | Greve JW, Gouma DJ, Soeters PB, Buurman WA. Suppression of cellular immunity in obstructive jaundice is caused by endotoxins: a study with germ-free rats. Gastroenterology. 1990;98:478-485. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 77] [Cited by in F6Publishing: 82] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 95. | Kountouras J, Billing BH, Scheuer PJ. Prolonged bile duct obstruction: a new experimental model for cirrhosis in the rat. Br J Exp Pathol. 1984;65:305-311. [PubMed] [Cited in This Article: ] |
| 96. | Jover R, Rodrigo R, Felipo V, Insausti R, Sáez-Valero J, García-Ayllón MS, Suárez I, Candela A, Compañ A, Esteban A, Cauli O, Ausó E, Rodríguez E, Gutiérrez A, Girona E, Erceg S, Berbel P, Pérez-Mateo M. Brain edema and inflammatory activation in bile duct ligated rats with diet-induced hyperammonemia: A model of hepatic encephalopathy in cirrhosis. Hepatology. 2006;43:1257-1266. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 123] [Cited by in F6Publishing: 119] [Article Influence: 6.6] [Reference Citation Analysis (0)] |