Published online Jul 26, 2020. doi: 10.4252/wjsc.v12.i7.659
Peer-review started: February 26, 2020
First decision: April 22, 2020
Revised: May 3, 2020
Accepted: May 27, 2020
Article in press: May 27, 2020
Published online: July 26, 2020
Processing time: 151 Days and 3.2 Hours
The impairment of cutaneous wound healing results in chronic, non-healing wounds that are caused by altered wound environment oxygenation, tissue injury, and permissive microbial growth. Current modalities for the treatment of these wounds inadequately address the complex changes involved in chronic wound pathogenesis. Consequently, stem cell therapies have emerged as a potential therapeutic modality to promote cutaneous regeneration through trophic and paracrine activity.
To investigate current literature regarding use of stem cell therapies for the clinical treatment of chronic, non-healing wounds.
PubMed, EMBASE, Cochrane Library, Web of Science, and Scopus were queried with combinations of the search terms “mesenchymal stem cells,” “adult stem cells,” “embryonic stem cells,” “erythroid precursor cells,” “stem cell therapies,” and “chronic wounds” in order to find relevant articles published between the years of 2000 and 2019 to review a 20-year experience. Reference lists from the articles were reviewed to identify additional pertinent articles. Retrieved manuscripts (reviews, case reports/series, retrospective/prospective studies, and clinical trials) were evaluated by the authors for their depiction of clinical stem cell therapy use. Data were extracted from the articles using a standardized collection tool.
A total of 43 articles describing the use of stem cell therapies for the treatment of chronic wounds were included in this review. While stem cell therapies have been explored in in vitro and in vivo applications in the past, recent efforts are geared towards assessing their clinical role. A review of the literature revealed that adipose-derived stem cells, bone marrow-derived stem cells, bone marrow-derived mononuclear cells, epidermally-derived mesenchymal stem cells, fibroblast stem cells, keratinocyte stem cells, placental mesenchymal stem cells, and umbilical cord mesenchymal stem cells have all been employed in the treatment of chronic wounds of various etiologies. Most recently, embryonic stem cells have emerged as a novel stem cell therapy with the capacity for multifaceted germ cell layer differentiation. With the capacity for self-renewal and differentiation, stem cells can enrich existing cell populations in chronic wounds in order to overcome barriers impeding the progression of wound healing. Further, stem cell therapies can be utilized to augment cell engraftment, signaling and activity, and resultant patient outcomes.
Assessing observed clinical outcomes, potential for stem cell use, and relevant therapeutic challenges allows wound care stakeholders to make informed decisions regarding optimal treatment approaches for their patients’ chronic wounds.
Core tip: Chronic wounds impose a significant burden on patients and the healthcare system with poor outcomes noted with the use of standard wound care protocols alone. Using stem cell therapies to treat these wounds results in improved cell signaling, release of growth factors and cytokines, neo-vessel formation, and immunomodulatory properties. Accordingly, patients experience enhanced healing of their formerly recalcitrant wounds. The objective of this review is to systematically evaluate the use of a wide range of stem cell therapies for the treatment of chronic wounds in order to guide providers in selecting appropriate treatment options for improved patient wound healing.
- Citation: Raghuram AC, Yu RP, Lo AY, Sung CJ, Bircan M, Thompson HJ, Wong AK. Role of stem cell therapies in treating chronic wounds: A systematic review. World J Stem Cells 2020; 12(7): 659-675
- URL: https://www.wjgnet.com/1948-0210/full/v12/i7/659.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v12.i7.659
Cutaneous wound healing is a complex and intricate process contingent on a series of regulated factors that work in a concerted manner to repair skin injury and restore barrier function. Superficial wound healing tends to follow a predictable course, with aberrancies generally noted only in the case of underlying disease states, such as diabetes. However, impairment of the wound healing process can result in chronic, non-healing wounds. Chronic wounds that do not heal within an expected period of 3 mo[1] not only result in pain and disfigurement, but also impose a significant burden on patients and the healthcare system, with annual cost estimates approaching $30 billion in the United States alone[2]. Chronic wounds are the consequence of local tissue hypoxia, bacterial colonization, and repetitive ischemia-reperfusion injury[3]. Non-healing wounds can originate from a variety of etiologies, including arterial disease, diabetes, vasculitis, venous valve insufficiency, prior irradiation[4], and skin malignancies[5].
Overcoming the factors that cause delayed wound healing involves an assessment of underlying pathology and consideration of advanced therapeutic agents. While a variety of wound care treatment options are available, few have demonstrated efficacy in the healing of chronic wounds and restoration of tissue to its pre-injury state. In contrast with normally healing wounds, chronic wounds exhibit increased levels of proinflammatory cytokines, reactive oxygen species (ROS), proteases, and senescent cells[6]. Traditional chronic wound care protocols rely on the debridement of necrotic or infected tissue followed by the application of wound dressings and topical agents that serve to protect the wound from infection and accelerate healing. In the case of certain chronic wounds, such as diabetic foot ulcers, offloading with external compression is crucial in order to minimize pressure placed on the wound[7]. Additionally, diabetic foot ulcers, venous leg ulcers, and open amputation wounds can be treated with negative pressure wound therapy to promote granulation tissue formation, wound area contraction, primary healing, and improved skin graft retention upon application to the wound bed[8]. Management of the wound edge is further performed with electromagnetic therapy, laser therapy, ultrasound therapy, and systemic oxygen therapy[9,10]. Although these therapeutic options are varied and can be tailored to each patient, these techniques have limited success and do not consistently facilitate complete wound closure.
Consequently, stem cell therapies have emerged as an exciting field of research because of their potential for the treatment of non-healing wounds. Notably, these wounds can be characterized by damaged or depleted stem cell populations[6,11]. Stem cells possess the unique capacity to self-renew and differentiate into various cell types. Moreover, stem cells can upregulate the secretion of cytokines and growth factors necessary for immunomodulation and regeneration[12], which are two critical features inadequately addressed in chronic wound healing pathogenesis. Ranging from immature pluripotent cells to more differentiated, multipotent cells, stem cells comprise a budding area of interest for improved healing of chronic wounds without the associated risks of major surgical procedures and added donor-site morbidity. The aim of the following review is to systematically review current literature that addresses the role of stem cell therapies in treating varied chronic, non-healing wounds in order to provide plastic surgeons, clinicians, and other chronic wound care stakeholders a comprehensive guide from which to select optimal therapies for improved patient outcomes.
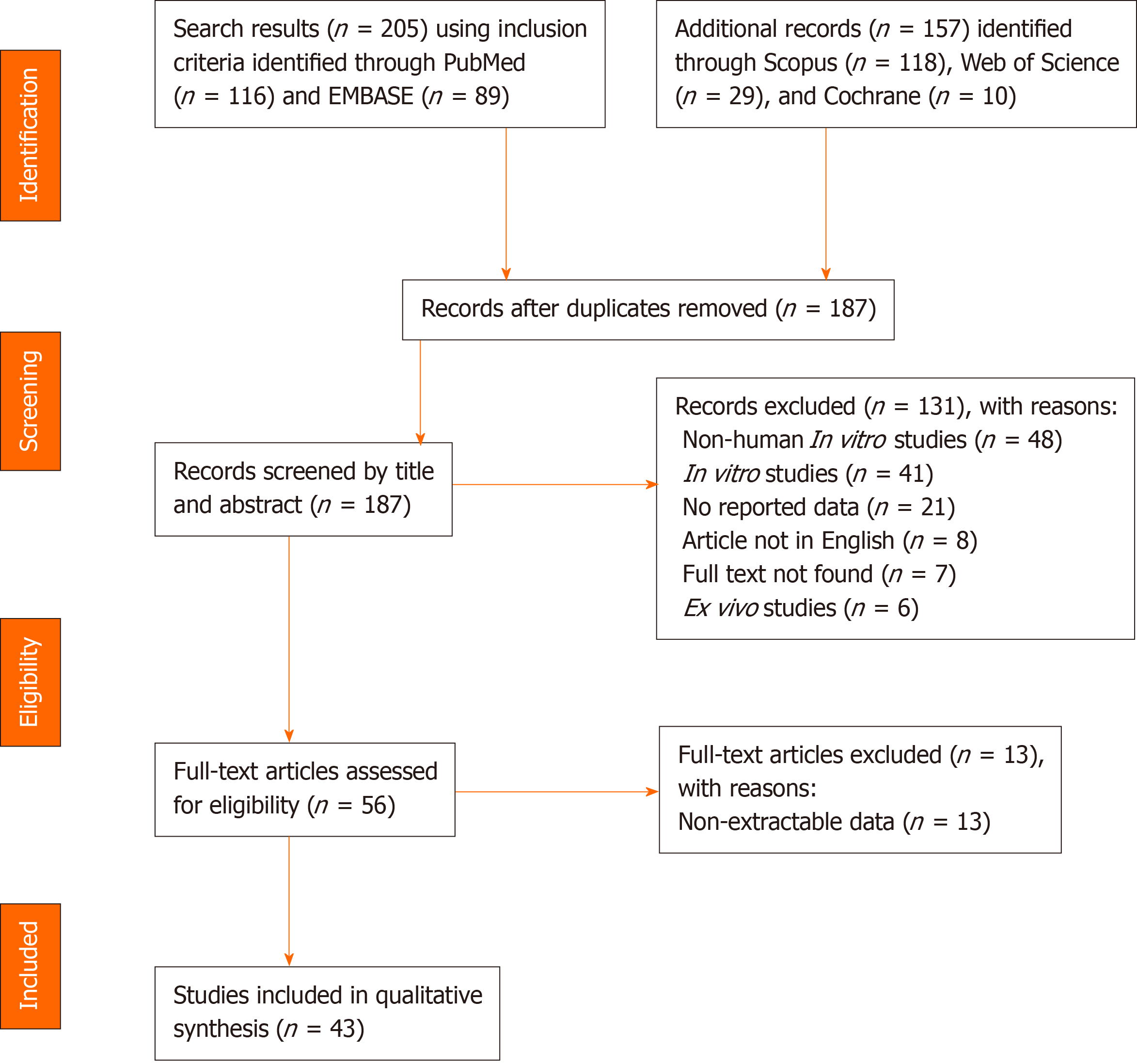
A systematic literature search was conducted according to the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) guidelines[13]. Figure 1 depicts the algorithm for article identification, screening, and review. Inclusion and exclusion criteria are presented in Table 1. PubMed, EMBASE, Cochrane Library, Web of Science, and Scopus were queried to identify relevant publications, articles, and abstracts that reported stem cell applications from the years 2000 to 2019. The queries employed a combination of search terms, including “mesenchymal stem cells,” “adult stem cells,” “embryonic stem cells,” “erythroid precursor cells,” “stem cell therapies,” and “chronic wounds.” The search parameters are described in detail in Supplementary Table 1. To eliminate bias, four authors independently screened all articles for inclusion or exclusion, and in the case of a conflict, a fifth author screened pertinent articles as a tiebreaker. Retrieved publications included systematic reviews, literature reviews, case reports and series, retrospective and prospective studies, and clinical trials. All studies that contained material applicable to the clinical use of stem cell therapies were reviewed, and data were extracted using a standardized collection tool. A total of 43 articles describing the use of stem cell therapies for the treatment of chronic wounds were included in this review[14-56].
| Inclusion criteria |
| Articles published in English |
| Stem cell therapies with demonstrated utility in the healing of chronic wounds either alone or as a complementary modality |
| Systematic reviews, literature reviews, case reports, case series, retrospective and prospective studies, and clinical trials between the years 2000-2019 |
| Exclusion criteria |
| Articles not published in English |
| No full text availability |
| No report of stem cell therapy application |
| Animal or non-human, ex vivo, or in vitro studies |
| Letters, comments, and editorials |
While stem cell therapies have been extensively explored in in vitro and in vivo settings, more recent investigative efforts have attempted to assess their clinical translatability. Transplanted cells deliver cytokines, chemokines, and growth factors, induce angiogenesis and innervation, and alter the wound inflammatory process[57]. To better assess the advantages of these stem cell properties in treating patients’ chronic wounds, we extracted data from studies that explore stem cell therapies in a clinical setting. Table 2 presents these treatment options, characteristic surface markers, indications for use, and mechanisms of action. Table 3 delineates the clinical outcomes observed, considerations for stem cell therapy optimization, and pertinent challenges associated with each therapy’s usage.
| Stem cell therapy | Cell markers | Indications | Mechanisms of action |
| Adipose-derived stem cells (ADSCs) | CD90+, CD105+, CD73+, CD44+, CD166+; CD34-, CD45- | Severe radiation injury, chronic ulcers, venous leg ulcers, chronic fistulae | Kim et al[61]: Promote angiogenesis, secrete growth factors and cytokines, and allow for human dermal fibroblast proliferation through direct cell contact and paracrine activation in the re-epithelialization phase; Trottier et al[62]: When combined with skin substitute with human extracellular matrix (ECM), ADSCs produce subcutaneous, dermal, and epidermal regenerated tissues |
| Bone marrow-derived stem cells (BMMSCs) | CD105+, CD73+, CD90+; CD13-, CD34-, CD45- | Severe radiation-associated wounds, chronic diabetic ulcers, advanced pressure ulcers in patients with spinal cord injury, and other intractable wounds | Han et al[64]: Synthesize high amounts of collagen, fibroblast growth factor (FGF), and vascular endothelial growth factor (VEGF); Ren et al[76]: Induce proliferation and potent differentiation of cells under low oxygen tension with morphologic and cell cycle changes towards bone and fat; Stoff et al[77]: Increase tensile strength of postoperative incisional wounds; Maxson et al[46]: Secrete antimicrobial factors and promote host immune response |
| Bone marrow-derived mononuclear cells (BMMNCs) | CD133+, CD117+, CD34+ | Chronic ulcers | Amato et al[15]: Secrete angiogenic growth factors to decrease local inflammation and promote vascularization |
| Epidermally-derived mesenchymal stem cells (EMSCs) | CD90+, CD73+, CD105+/-; CD34-, CD271- | Chronic ulcers, burns, generalized junctional epidermolysis bullosa (JEMB) | Yang et al[89]: Promote re-epithelialization in wound healing and regenerate functional epidermal layer |
| Fibroblast stem cells (FSCs) | CD34+, CD11b+, CD13+, MHC II+, CD86+, CD45+, collagen-1+, procollagen-1+; CD44- | Chronic ulcers | Amato et al[15]: Increase cell proliferation, ECM deposition, wound contraction, and vascularization with additional secretion of growth factors and cytokines |
| Keratinocyte stem cells (KSCs) | K5, K14, K15, integrins; CD34- | Chronic ulcers | Lampert et al[94]: Enable the formation of the stratified keratinizing epidermis; Domaszewska-Szostek et al[26]: Proliferate, migrate, and differentiate during re-epithelialization with mechanical, antibacterial, and nutritious roles. Keratinocytes also interact with fibroblasts during the wound healing process and tissue regeneration. They perform autocrine secretion of IL-6 and nitric oxide, release growth factors, and help restore the barrier function of skin |
| Placental mesenchymal stem cells (PMSCs) | CD105+, CD73+, CD90+, CD44+; CD34-, CD45- | Chronic venous ulcers | Farivar et al[30]: Stimulate tissue regeneration and repair for improved wound healing |
| Umbilical cord mesenchymal stem cells (UMSCs) | CD105+, CD73+, CD90+; CD34-, CD45- | Chronic diabetic ulcers | Hashemi et al[37]: Secrete growth factors for wound healing and can differentiate into fibroblast, epithelial, and endothelial cells for wound healing |
| Embryonic stem cells (ESCs) | Oct-4, Stage specific embryonic antigens (SSEAs) | Intractable wounds | Guenou et al[102]: Capable of differentiating into all three germ cell layers and can form functional human basal keratinocytes |
| Stem cell therapy | Clinical outcomes | Considerations for therapy optimization | Challenges associated with use |
| Adipose-derived stem cells (ADSCs) | Studies that demonstrated chronic wound healing: Akita et al[14]: No recurrences or wound abnormalities at 8 ± 2.2 wk (n = 5 patients); Lee et al: Wound healing rate of 66.7%, with improvement in pain and claudication walking distance (n = 15 patients); García-Olmo et al[72]: Epithelial covering of chronic Crohn's fistulas and healing by 8 wk (n = 4 patients); Rigotti et al[63]: Improved tissue ultrastructure, hydration, and neo-vessel formation in chronically radiated wounds over 31 mo (n = 20 patients); Studies that demonstrated a decrease in peripheral arterial disease ulcer size, number, and pain; Marino et al[71]: 6 patients had complete healing over 3 mo (n = 10 patients); Bura et al[106]: Improved transcutaneous saturation over 6 mo (n = 7 patients). Studies that showed higher chronic wound closure: Raposio et al[66]: Higher chronic wound closure than with control treatment (n = 16 patients); Carstens et al[67]: Complete chronic wound closure in 9 mo (n = 4 patients) with reduced pain; Han et al[64]: 100% chronic diabetic wound closure at 8 wk (n = 28 patients) vs 62% in the control group (n = 26 patients). Studies that demonstrated ulcer closure: Chopinaud et al[68]: Hypertensive leg ulcer closure of 93.1% at 6 mo with reduced fibrin, necrosis, and pain (n = 10 patients); Konstantinow et al[69]: 100% wound venous and arterial-venous ulcer closure over 6 mo (n = 13 patients) with reduced pain within days; Darinskas et al[70]: Complete ulcer healing (n = 15 patients) with less pain and walking improvement. | Stem cell delivery: Akita et al[14]: A 2-layered (atelocollagen + collagen) artificial dermis scaffold with injected ADSCs protects cells from infection and ambient dryness; Garg et al[73]: Capillary force ADSC seeding of hydrogels increases cell genetic expression and survival; Larsen et al[43]: ADSCs can be administered on an OASIS wound matrix for improved ulcer healing. Stem cell harvest: Akita et al[14]: ADSCs need to be cultured in very lean patients. When harvesting from subcutaneous adipose tissue, take care not to penetrate deeper viscera and vasculature. When injecting in chronic radiation injury sites, avoid surface rupture or laceration. | Gauglitz et al[31]: ADSCs are not immortal and display signs of "old age" when subject to culturing. Adipose tissue varies in metabolic activity and capacity for proliferation and differentiation, depending on the location of tissue harvest and other patient variables (age, gender); Marfia et al[45]: Autologous ADSCs have an altered genotype in diabetic patients, resulting in decreased potency, and decreased expression of vascular endothelial growth factor A and chemokine receptor CXCR4; Rezaie et al[50]: Short survival, poor transplantation, inferior homing, possibility of tumor formation, and loss of chemokine markers during ex vivo expansion. |
| Bone marrow-derived stem cells (BMMSCs) | Badiavas et al[78]: Demonstrated complete closure, dermal rebuilding, reduced scarring, and successful engraftment of cells over 1 year (n = 3 patients); Vojtassák et al[82]: Increase in dermal vascularity and dermal thickness of the wound bed after 29 d of treatment (n = 1 patient). Studies that demonstrated improved ulcer healing: Lu et al[79]: Improved healing in chronic diabetic ulcers at 6 wk with 100% healing 4 wk earlier than treatment with BMMNCs (n = 18 patients); Dash et al[80]: Improved Buerger disease (n = 9 patients) and diabetic (n = 3 patients) ulcer healing at 12 wk; Sarasúa et al[33]: Full healing of longstanding stage IV pressure ulcers in patients with spinal cord injury (n = 19 patients). Studies that showed decrease in chronic wound size: Gupta et al[35]: 70% reduction in chronic wound size over 3 wk followed by complete closure at 1 mo with application of cultured BMMSCs, rather than application of bone marrow aspirate alone (n = 19 patients); Humpert et al[75]: Reduction in chronic venous and neuroischemic wound size, increased vascularization, and infiltration of mononuclear cells after 7 d of treatment (n = 1 patient); Rogers et al[81]: Reduction in size in chronic wounds of different etiologies (n = 3 patients); Wettstein et al[54]: Reduction in chronic wound size of 50% over 3 wk of therapy (n = 3 patients). | Stem cell delivery: Yoshikawa et al[83]: Improved skin generation when BMMSCs are cultured in an artificial collagen dermis (n = 18 patients); Falanga et al[29]: 40% reduction in chronic wound size when BMMSCs are cultured in fibrin spray (n = 5 patients); Ravari et al[84]: Demonstrated significant reduction in chronic diabetic wounds after 4 wk of treatment when BMMSCs are co-administered with platelets, fibrin glue, and bone marrow-impregnated collagen matrix (n = 8 patients). | Rezaie et al[50]: Short survival, poor transplantation, inferior homing, possibility of tumor formation, and loss of chemokine markers during ex vivo expansion. |
| Bone marrow-derived mononuclear cells (BMMNCs) | Studies that demonstrated a higher wound healing rate: Yamaguchi et al[87]: Epidermal grafting significantly accelerated chronic diabetic foot ulcer healing (n = 10 patients); Jain et al[86]: Decreased wound area by 17.4% (n = 25 patients) compared to 4.84% with control (n = 23 patients) at 2 wk with average decrease of 36.4% vs 27.24% in wound area at 12 wk; Deng et al[24]: Demonstrated wound healing rate of 34.55% ± 11.18%, compared to control wound healing of 10.16% ± 2.67% (n = 10 patients); Deng et al[25]: Demonstrated wound healing of 26.5% ± 9.51% when administered as high density nanofat combined with negative pressure wound therapy (NPWT), compared to control healing of 12.02+4.2% with NPWT alone (n = 8 patients). | Not reported in reviewed articles. | Not reported in reviewed articles. |
| Epidermally-derived mesenchymal stem cells (EMSCs) | Bauer et al[90]: Regeneration of functional epidermis with gene-corrected EMSCs in previously infected, non-healing chronic ulcers due to junctional epidermolysis bullosa (n = 1 patient). | Teng et al[52]: Cultured epidermal autografts enriched with EMSCs on an ECM-compatible substrate can overcome EMSC deficiency in chronic wounds and provide ECM materials to stabilize the wound site. | Not reported in reviewed articles. |
| Fibroblast stem cells (FSCs) | Yamada et al[107]: Chronic wound size reduction of 33.3% (n = 5 patients); You et al[105]: Complete diabetic foot ulcer healing in 84% of patients (n = 26 patients) over 36.4+17.6 d. | Stem cell delivery: Brower et al[20]: Enhanced cell adherence, proliferation, and migration when delivered in a 2-chamber fibrin sealant; Yonezawa et al[92]: Improved wound healing of 92% (n = 13 patients) when fibroblasts were cultured on a hyaluronic acid and atelo-collagen matrix; Marcelo et al[108]: More complete wound healing achieved when administered with autologous fibrin glue over the ulcer site. | Not reported in reviewed articles. |
| Keratinocyte stem cells (KSCs) | Studies that demonstrated improved wound healing: Vanscheidt et al[95]: Complete and faster wound healing in 38.3% of patients receiving autologous keratinocytes, compared to 22.4% in the control group (n = 44 patients); Moustafa et al[96]: improved wound healing over 6 wk when administered as cultured autologous keratinocytes on cell-free discs (n = 12 patients); Bayram et al[99]: Reduction in diabetic foot wound size of 92% (n = 20 patients), compared to control wound size reduction of 30%. Studies that demonstrated complete ulcer healing: Hartmann et al[97]: Complete ulcer healing at a mean of 14.5 wk when administered as keratinocytes transplanted in a fibrin carrier (n = 4 patients); De Luca et al[98]: Complete ulcer healing in 20 wk (n = 20 patients) with a 30%-84.4% reduction in size after 3 wk (n = 4 patients); Beele et al[100]: Demonstrated complete venous leg ulcer closure within 1 wk (n = 11 patients) over a period of 4.1-24.9 wk with decreased pain. | Stem cell delivery: Hartmann et al[97]: Better keratinocyte graft fixation and epithelial monolayer formation with low-density fibrin; Bayram et al[99]: More effective delivery when cultured keratinocytes are attached to microcarriers made of polyethylene and silica; Teepe et al[109]: Radial progression toward wound closure when administered as cryopreserved cultured allografts (n = 43 patients); Shukla et al[101]: Complete chronic wound healing over 12-48 wk when administered as keratinocytes along with epidermal cell suspension (n = 12 patients). | Rezaie et al[50]: Difficult to isolate, short lifespan during serial cultivation, and possible tumor formation. |
| Placental mesenchymal stem cells (PMSCs) | Farivar et al[30]: Complete chronic venous ulcer healing in 53% (n = 21 patients with 30 total ulcers) with 79% mean reduction in wound surface area over an average of 10.9 wk. | Farivar et al[30]: Improved cell survival when MSCs are administered in cryopreserved, aseptic placental tissue (hVWM). | Duscher et al[27]: Appropriate donor selection is necessary to avoid immune-mediated rejection or transmission of genetic diseases. More efficient cell isolation, culture, and expansion techniques are needed, along with close surveillance for malignant transformation. |
| Umbilical cord mesenchymal stem cells (UMSCs) | Hashemi et al[37]: Significant decrease in chronic diabetic ulcer size over 9 d with decreased wound healing time (n = 5 patients). | Hashemi et al[37]: Improved tissue regeneration and wound healing when cells are seeded on an acellular amniotic membrane scaffold. | Duscher et al[27]: Appropriate donor selection is necessary to avoid immune-mediated rejection or transmission of genetic diseases. More efficient cell isolation, culture, and expansion techniques are needed, along with close surveillance for malignant transformation. |
| Embryonic stem cells (ESCs) | Not reported in reviewed articles. | Not reported in reviewed articles. | Okano et al[104]: Patients will need to be monitored for tumorigenesis and teratoma formation. |
Adipose-derived stem cells (ADSCs) are mesenchymal stem cells that comprise a pluripotent, heterogenous cell population within human adipose tissue. The popularity of ADSCs can be attributed to their ease of harvest and limited donor-site morbidity; they can be isolated via liposuction aspiration or excision of fat samples[27]. The most common approach for isolating ADSCs involves collagenase digestion followed by centrifugal density gradient separation[58]. Isolated ADSCs can then be expanded in monolayer cultures on standard tissue dishes with a basal medium of 10% fetal bovine serum[59]. Clinical use of ADSCs requires in vitro expansion that complies with good manufacturing practice (GMP) guidelines to ensure that no xenogeneic components are cultivated. As ADSCs are mesenchymal stem cells, their surface markers include CD90, CD105, CD73, CD44, and CD166 without the expression of hematopoietic cell surface markers CD34 and CD45[60].
ADSCs have demonstrated clinical efficacy in the treatment of chronic wounds secondary to severe radiation injury, chronic fistulae, and ulceration, which includes venous leg ulcers. Mechanistically, this stem cell population facilitates angiogenesis, augments the secretion of growth factors and cytokines, and allows for human dermal fibroblast proliferation through direct cell contact and paracrine activation during the re-epithelialization phase of wound healing[61]. Notably, when combined with skin substitute containing human extracellular matrix (ECM), ADSCs permit regeneration of subcutaneous, dermal, and epidermal tissues[62].
ADSCs are among the more robustly explored stem cell therapy type in the clinical treatment of chronic wounds. They have demonstrated results of improved wound healing and closure, tissue ultrastructure and hydration, neo-vessel formation, and patient symptomatology of pain and claudication[14,63-70]. In the treatment of chronic ulcers secondary to peripheral arterial disease, Marino et al[71] observed decreased ulcer size, depth, pain, and improved transcutaneous saturation in all patients over the course of treatment. The versatility of ADSCs is further highlighted in treating chronic Crohn’s fistulas; treated patients experienced improved wound healing seen over 8 wk without the incidence of adverse events[72]. Akita et al[14] found that the use of an artificial dermis (Terudermis, Japan) scaffold protected ADSCs from infection and ambient dryness, while Larsen et al[43] found improved ulcer re-epithelialization and healing when ADSCs were administered on an OASIS wound matrix in conjunction with compression therapy. In addition, seeding ADSCs in hydrogel delivery vehicles by capillary force and then administering these hydrogel systems in vivo effectively enhances stem cell genetic expression and survival for improved wound healing outcomes[73]. When harvesting ADSCs from subcutaneous adipose tissue, it is important to avoid penetration of the deeper visceral cavity or underlying major muscles, vessels, and nerves. Upon injection of ADSCs to chronically radiated wound beds, care must also be taken to avoid surface rupture or laceration[14].
Though their clinical potential has been favorably evidenced, there are some challenges to consider with the use of ADSCs. This cell population is not immortal and displays signs of aging and loss of chemokine markers with repeat culturing[50]. Further, adipose tissue can widely vary in its metabolic activity and capacity for proliferation and differentiation depending on the site of tissue harvest and patient characteristics, such as age and sex[31]. Marfia et al[45] identified that autologous ADSCs have an altered genotype in diabetic patients, which is characterized by decreased potency and expression of vascular endothelial growth factor-A (VEGF-A) and chemokine receptor CXCR4. Judicious use of ADSC therapy requires close patient surveillance for tumor formation as well as consideration of strategies to improve cell homing and transplantation[50].
Bone marrow-derived stem cells (BMMSCs) were originally isolated by Friedenstein et al[74] in 1966 and presently constitute a mesenchymal stem cell population that is typically harvested via iliac crest aspiration. This aspirate is then subjected to in vitro selection, cell expansion in culture, and topical application to wounds for tissue regeneration. BMMSCs display the typical mesenchymal stem cell surface markers, including CD105, CD73, and CD90. Their clinical utility encompasses the treatment of severe radiation-associated wounds, chronic diabetic ulcers, advanced pressure ulcers, as seen in patients who have undergone spinal cord injury, and other forms of intractable wounds.
Consistent with other mesenchymal stem cell types, BMMSCs have immunomodulatory properties[46] and promote angiogenesis[75]. These stem cells synthesize high amounts of collagen, fibroblast growth factor (FGF), and VEGF[64], allow for cell proliferation and differentiation under low oxygen tension conditions[76], and demonstrate increased tensile strength when applied to postoperative incisional wounds[77]. When clinically administered, BMMSCs have shown complete closure, dermal rebuilding, reduced scarring, and successful cell engraftment in non-healing wounds[78]. Particularly in the case of chronic ulcers and wounds resultant from traumatic, thermal, electric, or infectious etiologies, this stem cell therapy is shown to significantly reduce wound size[29,35,54,79-82]. For chronic diabetic ulcers, BMMSC therapy results in improved limb perfusion, ankle-brachial indices, transcutaneous oxygen pressure, and magnetic resonance angiography analysis[79]. Sarasúa et al[33] found that BMMSC treatment of longstanding stage IV pressure ulcers in patients with spinal cord injury decreased the mean hospital stay from 85.16 to 43.06 d, when compared with standard treatment modalities alone. In addition, patients given the BMMSC treatment did not evidence any ulcer recurrence over a follow-up period of 19 mo.
In order to improve the delivery and outcomes associated with BMMSC therapy, these cells can be cultured and placed in an artificial collagen dermis as a composite graft, which can improve skin regeneration processes[83]. Additionally, when BMMSCs are cultured and administered via a fibrin spray, chronic wound or ulcer size is reduced significantly by 40% over a period of 20 wk, with no adverse events reported[29]. To enhance their favorable therapeutic profile, BMMSCs can be administered along with platelets, fibrin glue, and bone marrow-impregnated collagen matrix; this strategy has resulted in significant diabetic wound closure in patients with formerly recalcitrant wounds[84]. Similar to ADSCs, BMMSCs necessitate careful monitoring of patients for possible tumor formation as well as appropriate but not excessive culturing in order to avoid transplantation of aged cells with inferior homing abilities and loss of chemokine markers[50].
Bone marrow-derived mononuclear cells (BMMNCs) are a heterogeneous group of cells that include mature B cells, T cells, monocytes, and a smaller proportion of progenitor cells, including hematopoietic stem cells, mesenchymal stem cells, endothelial progenitor cells, and embryonic-like cells[85]. BMMNCs offer ease of harvest, processing, and administration, making them a favorable option for clinical testing. Cell surface markers include CD133, CD117, and CD34, and BMMNCs have been clinically applied most frequently in the treatment of chronic ulcers. Their most notable property is the ability to secrete angiogenic growth factors that decrease local inflammation and promote vascularization[15].
BMMNCs simultaneously accelerate the rate of wound healing and decrease wound area compared to non-stem cell therapy treatment[24,86]. When applied with epidermal grafting for the treatment of chronic diabetic foot ulcers, Yamaguchi et al[87] observed significant ulcer healing without incidence of osteomyelitis or the need for patient amputation. Moreover, when administered in conjunction with high density nanofat and negative pressure wound therapy, Deng et al[25] reported improved patient wound healing outcomes as well as decreased lymphocyte recruitment, higher collagen deposition, and increased vessel growth.
Epidermally-derived mesenchymal stem cells (EMSCs) categorize the mesenchymal stem cell population present within the epidermis that is responsible for homeostasis of the superficial skin layers[88]. This population includes interfollicular, sebaceous gland, and bulge area stem cells. EMSCs are characterized by the cell surface markers CD90 and CD73, with variable expression of CD105. Clinically, this stem cell therapy has been employed to treat chronic ulcers, burns, and non-healing wounds secondary to junctional epidermolysis bullosa (JEMB). EMSCs promote both re-epithelialization in wound healing and regeneration of a functional epidermal skin layer[89]. Of note, if EMSCs undergo gene correction, they promote enhanced functional epidermal regeneration in JEMB ulcers[90]. Teng et al[52] found that cultured epidermal autografts enriched with EMSCs on an ECM-compatible substrate not only replenished EMSCs in chronic wounds, but also stabilized the ECM substrate within the wound site. As a stem cell category, EMSCs can be manipulated and stimulated via biomaterial-based approaches to modify their spatial and temporal cues for precise niche conditions that are beneficial in wound healing and skin regeneration[27].
Fibroblast stem cells (FSCs) are part of a relatively novel focus in regenerative medicine and can be generated by reprogramming adult fibroblasts into an immature, pluripotent state[91]. These autologous induced pluripotent stem cells (iPSCs) are furthermore nonimmunogenic. The addition of fibroblasts to the chronic wound healing environment addresses the deficiency of appropriately functioning fibroblasts in the setting of this chronic inflammatory state. In non-healing wounds, fibroblasts exhibit premature, stress-induced cellular senescence with decreased proliferative potential, impaired reactivity to growth factors, and abnormal protein production[26]. Fibroblasts and their stem cell equivalent states have been employed in the treatment of chronic ulcers, and their characteristic cell surface markers include CD34, CD11b, CD13, MHC II, CD86, CD45, collagen-1, and procollagen-1. This cell population functions to promote proliferation, ECM deposition, wound contraction, vascularization, and secretion of growth factors and cytokines[15].
Clinically, FSCs have been shown to be more effective when co-delivered or co-cultured with additives, such as fibrin glue[57]. Brower et al[20] found that commercially available 2-chamber fibrin sealants, containing fibrinogen and thrombin, enhanced the adherence, proliferation, and migration of fibrocytes. Furthermore, fibroblasts have also demonstrated improved wound healing when cultured on hyaluronic acid and atelo-collagen matrices[92] prior to application to chronic wound sites.
Keratinocytes comprise the majority of cells within the human epidermis and promote re-epithelialization via proliferation, migration, and differentiation[93]. Keratinocyte stem cells (KSCs) reside in the basal epidermis, hair follicles, and sebaceous glands and can serve to replenish depleted keratinocyte populations found in chronic wounds. Characteristic cell surface markers of KSCs include K5, K14, K15, and integrins, and KSCs have been most widely applied in the treatment of chronic ulcers. These stem cells enable the formation of a stratified, keratinizing epidermis[94], contribute to the process of re-epithelialization, and offer antibacterial and nutritious roles[26]. When differentiated into keratinocytes, these cells interact with fibroblasts and perform autocrine secretion of interleukin-6 (IL-6) and nitric oxide, release growth factors, and facilitate the restoration of the skin’s barrier function.
When administered to patients with chronic wounds, keratinocytes promote complete wound healing over a shorter period of time[95-99] compared to standard wound healing therapy, with additionally decreased local wound pain[100]. Moreover, keratinocytes can be cultured on cell-free discs[96], administered as cryopreserved cultured allografts, or suspended with epidermal cells in order to improve chronic wound healing outcomes[101]. Graft fixation can be performed with low-density fibrin to ensure better cell survival and epithelial monolayer formation, thereby optimizing keratinocyte therapy[97]. Bayram et al[99] found that cultured keratinocytes attached to microcarriers of polyethylene and silica resulted in better cell delivery than keratinocytes administered alone. When considering KSC or keratinocyte therapy, it is important to recognize that these cells can be difficult to isolate, display a short lifespan throughout serial cultivation, and necessitate patient monitoring for tumor formation[50].
Placental mesenchymal stem cells (PMSCs) are another member of the mesenchymal stem cell category with clinical benefits. PMSCs stimulate the wound healing process through the release of trophic mediators, promotion of new vessel formation, recruitment of endogenous progenitor cells, and facilitation of cell differentiation, proliferation, and ECM formation[27]. As with other mesenchymal stem cells, PMSC surface markers include CD105, CD73, CD90, and CD44. PMSCs have been clinically investigated in the treatment of chronic venous ulcers with resultant higher quality tissue regeneration and repair[30].
When administered as cryopreserved placental tissue containing PMSCs, Farivar et al[30] demonstrated improved chronic venous ulcer healing, compared to standard therapy, with a significant reduction in size from baseline. PMSC therapy can be optimized when the cells are cryopreserved in aseptic placental tissue to protect placental tissue components, including growth factors and collagen-rich extracellular membranes, from degradation[30]. These stem cells require careful donor selection to avoid immune rejection or genetic disease transmission. Additionally, patient surveillance is imperative to monitor for malignant transformation[27].
Umbilical cord mesenchymal stem cells (UMSCs) represent another mesenchymal stem cell population aptly isolated from umbilical cord-lining tissue. Notably, umbilical cord epithelial cells have stem-cell like properties and can form stratified epithelium[27]. As with placental-derived mesenchymal stem cells, UMSCs are extracted from an extra-fetal source, and donors must be appropriately selected to avoid immune-mediated rejection or transmission of genetic diseases. Characteristic cell surface markers include CD105, CD73, and CD90. UMSCs secrete growth factors for wound healing and are capable of differentiating into fibroblast, epithelial, and endothelial cell subtypes for improved wound healing[37].
UMSCs have been employed in the treatment of chronic diabetic ulcers and significantly decrease both ulcer size and time required for wound healing to occur[37]. When seeded on an acellular amniotic membrane scaffold, UMSCs promote tissue regeneration and improve wound healing outcomes. This scaffold not only confers anti-adhesive, bacteriostatic, and epithelialization properties, but also attenuates the wound pain reported by patients[37]. In refining the applications for UMSCs, it is necessary to develop more efficient techniques for cell isolation, culture, and expansion. Lastly, it is essential to monitor patients for the possibility of tumor formation[27].
Embryonic stem cells (ESCs) constitute the latest and perhaps most up-and-coming category in stem cell therapy development. ESCs are pluripotent stem cells derived from the blastocyst stage of embryos and, given the correct conditions, are capable of differentiating into any cell in all three germ cell layers[42]. These ESC-derived cell lineages include hematopoietic stem cells, differentiated T cells, and epidermal cells. Characteristic cell surface markers include octamer-binding transcription factor 4 (Oct-4) and stage specific embryonic antigens (SSEAs). Because of their pluripotent nature, ESCs have the unique potential to serve as a therapeutic modality in the treatment of multiple disease processes, including chronic wounds. Preclinical studies have demonstrated that ESCs can differentiate into fully functioning human basal keratinocytes, which can further develop into stratified epidermis[102]. In vivo studies in mice have also shown that when compared to endothelial progenitor cells derived from cord blood, human ESCs exhibit improved dermal regeneration and re-epithelialization in chronic wounds.
The clinical use of ESCs is, however, limited by the ethical controversy surrounding their procurement. ESC therapy has been widely debated because currently, human ESCs cannot be obtained with causing significant damage to the human embryo. Proponents of ESC therapy maintain that this treatment will advance medical science and that the politicization of scientific research stymies this progress[103]. On the other hand, induced pluripotent stem cells (iPSCs) offer an effective alternative to ESC therapy that demonstrate in vitro and in vivo potential in wound healing while circumventing the ethical concerns of ESCs. These iPSCs can be generated via programming of differentiated adult keratinocytes with embryonic-like stem cell properties. The process of reprogramming keratinocytes involves retroviral transduction of essential transcription factors, such as c-Myc, Klf4, Oct-3/4, and Sox2[48]. Nonetheless, the use of either iPSC or ESC therapy will require the medical community to address their respective safety concerns. Despite their associated risk for tumorigenesis and teratoma formation, these cells offer a promising option for the treatment of chronic wounds because of their ability to differentiate into all three germ cell layers[104].
The role of stem cell therapies in the treatment of chronic, non-healing wounds is continuously being refined within the scope of tissue engineering. Stem cells promote restoration of impaired signaling pathways for growth factors, delivery of important cytokines and chemokines, induction of vascularization and innervation, and more precise control of the inflammatory processes underlying chronic wounds[105]. Considerations with therapy usage involve donor variables, such as site and cell availability, patient age, and patient sex, as well as risks for diminished stem cell efficacy associated with repetitive culturing and possible malignant transformation. Nonetheless, stem cell therapy can be optimized extensively via a wide range of co-delivery techniques in the form of scaffolds, hydrogels, and other carriers with or without wound healing additives.
As with any systematic evaluation of the literature, there are some important limitations to consider with this review. Compared to preliminary, preclinical work, there are fewer clinical studies exploring the full scope of stem cell therapies for the treatment of chronic wounds. Given the lack of clear guidelines regarding therapy use for certain patient demographics, underlying health statuses, and presence of comorbid diseases, it is difficult to generalize the applicability of one therapy to another chronic wound etiology that has not been previously tested. Therapeutic success is determined on a case-by-case basis.
However, there are ongoing investigative efforts to improve understanding of stem cell therapies. Recently, the use of electrospun fiber scaffolds for stem cell therapy culture has been explored with the goal of enhancing cell proliferation and differentiation. This technique additionally opens up new possibilities for controlled fiber morphology and structure to generate layered skin substitute dressings[32]. Though clinical studies and trials have supplemented our knowledge of stem cell therapy usage for chronic, non-healing wounds, further studies are needed in order to more comprehensively examine the breadth of this therapeutic modality and more closely personalize wound care regimens for each individual patient. As these treatments become more advanced by optimizing wound healing outcomes while minimizing donor morbidity, stem cell-based therapy is likely to establish itself as a mainstay of chronic wound care and management.
Chronic wounds are defined as those that do not heal within a period of 3 mo, resulting in significant patient morbidity and healthcare burden. Due to local tissue hypoxia, bacterial colonization, ischemia-reperfusion injury, and diminished stem cell populations, these wounds do not progress through the normal wound healing phases. Further, non-healing wounds are attributable to a host of etiologies, including arterial disease, diabetes, vasculitis, venous valve insufficiency, irradiation, and malignancy. Their complex pathophysiology poses a formidable treatment challenge, and presently, there are ineffective techniques to facilitate wound closure and improved patient symptomatology. Stem cell therapies have therefore emerged as a unique therapeutic approach to modulate the chronic wound environment in favor of healing. In this systematic review, we evaluate literature over the past two decades to ascertain clinical findings associated with stem cell therapies for treating chronic wounds.
While adipose-derived stem cells (ADSCs) and bone marrow-derived stem cells (BMMSCs) have been tested the most frequently in clinical settings, it is unclear how other emerging stem cell therapy types function in healing chronic wounds. It is critical that we comprehensively consider a variety of stem cell therapies for the treatment of a diverse scope of non-healing wounds in order to provide maximal clinical benefit to wound care specialists and providers.
To investigate the scope of a variety of stem cell therapies, including adipose-derived stem cells (ADSCs), bone marrow-derived stem cells (BMMSCs), bone marrow-derived mononuclear cells (BMMNCs), epidermally-derived mesenchymal stem cells (EMSCs), fibroblast stem cells (FSCs), keratinocyte stem cells (KSCs), placental mesenchymal stem cells (PMSCs), umbilical cord mesenchymal stem cells (UMSCs), and embryonic stem cells (ESCs), for the treatment of chronic, non-healing wounds.
We performed a systematic review of the literature according to the 2009 PRISMA guidelines. Five authors conducted a search in five databases (PubMed, EMBASE, Cochrane Library, Web of Science, and Scopus) to identify relevant publications, articles, and abstracts reporting clinical stem cell therapy use for chronic wounds from the years 2000 to 2019.
A total of 43 studies were included in this review. The studies reported that ADSCs and BMMSCs have been tested in the widest scope of clinical applications, including the treatment of severe radiation-associated wounds, venous leg ulcers, chronic fistulae, chronic diabetic ulcers, and advanced pressure ulcers from spinal cord injury. Enhanced testing of other stem cell therapy types has provided informative guidelines for therapy optimization, including seeding stem cells into artificial dermal, wound matrix, and hydrogel scaffolds for improved cell survival and proliferation in the wound beds. FSCs and KSCs can be delivered with additives, including fibrin, to strengthen cell properties of adherence, migration, and epithelial monolayer formation. Improved wound healing with each of the stem cell therapy types was determined on the basis of histological and functional parameters. No studies reported significant complications with clinical use of any of the investigated therapies.
Stem cells promote healing of chronic wounds by restoring impaired signaling pathways for growth factors, ensuring delivery of important cytokines and chemokines, inducing vascularization and innervation, and modulating inflammatory processes. Selecting optimal therapy for various wounds is contingent on patient variables, including age, sex, and stem cell donor site, as well as processing variables, such as culturing after cell harvest and potential for malignant transformation. Additional clinical studies are required to replicate the strength of the literature findings for ADSCs and BMMSCs and substantiate use of a wider scope of stem cell therapies for treating non-healing wounds.
There is limited clinical evidence examining the use of EMSCs, FSCs, KSCs, PMSCs, UMSCs, and ESCs for the treatment of chronic wounds, though these stem cells have demonstrated potential in preclinical in vitro and in vivo work. Further studies exploring the use of these therapies for clinically diverse patient wounds would be highly informative. In addition to aforementioned artificial dermal, wound matrix, and hydrogel scaffolds, there is ongoing development with newer cell delivery constructs, such as electrospun fiber scaffolds to facilitate creation of layered skin substitute dressings. As the etiology of chronic wounds varies from patient to patient, it is necessary to personalize therapeutic approaches. The goal of future investigations will be to further realize improved patient wound closure and histologic markers of wound healing, as well as decreased pain, disfigurement, and healthcare system burden.
Manuscript source: Invited manuscript
Corresponding Author's Membership(s) in Professional Societies: Plastic Surgery Research Council; American Board of Plastic Surgery; American Society for Reconstructive Microsurgery; American College of Surgeons; World Society of Reconstructive Microsurgery; California Society of Plastic Surgeons; Association for Academic Surgery; American Association of Plastic Surgeons.
Specialty type: Cell and tissue engineering
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: He XH S-Editor: Gong ZM L-Editor: A E-Editor: Xing YX
| 1. | Nunan R, Harding KG, Martin P. Clinical challenges of chronic wounds: searching for an optimal animal model to recapitulate their complexity. Dis Model Mech. 2014;7:1205-1213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 318] [Article Influence: 31.8] [Reference Citation Analysis (0)] |
| 2. | Nussbaum SR, Carter MJ, Fife CE, DaVanzo J, Haught R, Nusgart M, Cartwright D. An Economic Evaluation of the Impact, Cost, and Medicare Policy Implications of Chronic Nonhealing Wounds. Value Health. 2018;21:27-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 533] [Cited by in RCA: 725] [Article Influence: 103.6] [Reference Citation Analysis (0)] |
| 3. | Mustoe TA, O'Shaughnessy K, Kloeters O. Chronic wound pathogenesis and current treatment strategies: a unifying hypothesis. Plast Reconstr Surg. 2006;117:35S-41S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 363] [Cited by in RCA: 395] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 4. | Jacobson LK, Johnson MB, Dedhia RD, Niknam-Bienia S, Wong AK. Impaired wound healing after radiation therapy: A systematic review of pathogenesis and treatment. JPRAS Open. 2017;13:92-105. [RCA] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 67] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 5. | Mekkes JR, Loots MA, Van Der Wal AC, Bos JD. Causes, investigation and treatment of leg ulceration. Br J Dermatol. 2003;148:388-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 206] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 6. | Frykberg RG, Banks J. Challenges in the Treatment of Chronic Wounds. Adv Wound Care (New Rochelle). 2015;4:560-582. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1534] [Cited by in RCA: 1405] [Article Influence: 140.5] [Reference Citation Analysis (0)] |
| 7. | Granick M, Boykin J, Gamelli R, Schultz G, Tenenhaus M. Toward a common language: surgical wound bed preparation and debridement. Wound Repair Regen. 2006;14 Suppl 1:S1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 50] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 8. | Scherer SS, Pietramaggiori G, Mathews JC, Prsa MJ, Huang S, Orgill DP. The mechanism of action of the vacuum-assisted closure device. Plast Reconstr Surg. 2008;122:786-797. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 211] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 9. | Halim AS, Khoo TL, Saad AZ. Wound bed preparation from a clinical perspective. Indian J Plast Surg. 2012;45:193-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 60] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 10. | Leaper DJ, Schultz G, Carville K, Fletcher J, Swanson T, Drake R. Extending the TIME concept: what have we learned in the past 10 years?(*). Int Wound J. 2012;9 Suppl 2:1-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 223] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 11. | Johnson MB, Niknam-Bienia S, Soundararajan V, Pang B, Jung E, Gardner DJ, Xu X, Park SY, Wang C, Chen X, Baker RY, Chen M, Hong YK, Li W, Wong AK. Mesenchymal Stromal Cells Isolated from Irradiated Human Skin Have Diminished Capacity for Proliferation, Differentiation, Colony Formation, and Paracrine Stimulation. Stem Cells Transl Med. 2019;8:925-934. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 12. | Meirelles Lda S, Fontes AM, Covas DT, Caplan AI. Mechanisms involved in the therapeutic properties of mesenchymal stem cells. Cytokine Growth Factor Rev. 2009;20:419-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 951] [Cited by in RCA: 1010] [Article Influence: 63.1] [Reference Citation Analysis (0)] |
| 13. | Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52948] [Cited by in RCA: 46855] [Article Influence: 2928.4] [Reference Citation Analysis (0)] |
| 14. | Akita S, Yoshimoto H, Akino K, Ohtsuru A, Hayashida K, Hirano A, Suzuki K, Yamashita S. Early experiences with stem cells in treating chronic wounds. Clin Plast Surg. 2012;39:281-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 15. | Amato B, Compagna R, Amato M, Butrico L, Fugetto F, Chibireva MD, Barbetta A, Cannistrà M, de Franciscis S, Serra R. The role of adult tissue-derived stem cells in chronic leg ulcers: a systematic review focused on tissue regeneration medicine. Int Wound J. 2016;13:1289-1298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 16. | Aronson A, Laageide L, Powers J. Use of stem cells in wound healing. Cur Dermatol Rep. 2018;7:278-286. [RCA] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 17. | Boháč M, Csöbönyeiová M, Kupcová I, Zamborský R, Fedeleš J, Koller J. Stem cell regenerative potential for plastic and reconstructive surgery. Cell Tissue Bank. 2016;17:735-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 18. | Branski LK, Gauglitz GG, Herndon DN, Jeschke MG. A review of gene and stem cell therapy in cutaneous wound healing. Burns. 2009;35:171-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 170] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 19. | Bronckaers A, Hilkens P, Martens W, Gervois P, Ratajczak J, Struys T, Lambrichts I. Mesenchymal stem/stromal cells as a pharmacological and therapeutic approach to accelerate angiogenesis. Pharmacol Ther. 2014;143:181-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 263] [Article Influence: 23.9] [Reference Citation Analysis (0)] |
| 20. | Brower J, Blumberg S, Carroll E, Pastar I, Brem H, Chen W. Mesenchymal stem cell therapy and delivery systems in nonhealing wounds. Adv Skin Wound Care. 2011;24:524-532; quiz 533-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 21. | Cha J, Falanga V. Stem cells in cutaneous wound healing. Clin Dermatol. 2007;25:73-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 120] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 22. | Coalson E, Bishop E, Liu W, Feng Y, Spezia M, Liu B, Shen Y, Wu D, Du S, Li AJ, Ye Z, Zhao L, Cao D, Li A, Hagag O, Deng A, Liu W, Li M, Haydon RC, Shi L, Athiviraham A, Lee MJ, Wolf JM, Ameer GA, He TC, Reid RR. Stem cell therapy for chronic skin wounds in the era of personalized medicine: From bench to bedside. Genes Dis. 2019;6:342-358. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 44] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 23. | Dabiri G, Heiner D, Falanga V. The emerging use of bone marrow-derived mesenchymal stem cells in the treatment of human chronic wounds. Expert Opin Emerg Drugs. 2013;18:405-419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 24. | Deng C, Wang L, Feng J, Lu F. Treatment of human chronic wounds with autologous extracellular matrix/stromal vascular fraction gel: A STROBE-compliant study. Medicine (Baltimore). 2018;97:e11667. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 42] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 25. | Deng C, Yao Y, Liu Z, Li H, Yang Z, Wang D, Wei Z. Chronic wound treatment with high-density nanofat grafting combined with negative pressure wound therapy. Int J Clin Exp Med. 2019;12:1402-1411. |
| 26. | Domaszewska-Szostek A, Krzyżanowska M, Siemionow M. Cell-Based Therapies for Chronic Wounds Tested in Clinical Studies: Review. Ann Plast Surg. 2019;83:e96-e109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 27. | Duscher D, Barrera J, Wong VW, Maan ZN, Whittam AJ, Januszyk M, Gurtner GC. Stem Cells in Wound Healing: The Future of Regenerative Medicine? A Mini-Review. Gerontology. 2016;62:216-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 157] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 28. | Ennis WJ, Sui A, Bartholomew A. Stem Cells and Healing: Impact on Inflammation. Adv Wound Care (New Rochelle). 2013;2:369-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 85] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 29. | Falanga V, Iwamoto S, Chartier M, Yufit T, Butmarc J, Kouttab N, Shrayer D, Carson P. Autologous bone marrow-derived cultured mesenchymal stem cells delivered in a fibrin spray accelerate healing in murine and human cutaneous wounds. Tissue Eng. 2007;13:1299-1312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 533] [Cited by in RCA: 519] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 30. | Farivar BS, Toursavadkohi S, Monahan TS, Sharma J, Ucuzian AA, Kundi R, Sarkar R, Lal BK. Prospective study of cryopreserved placental tissue wound matrix in the management of chronic venous leg ulcers. J Vasc Surg Venous Lymphat Disord. 2019;7:228-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 31. | Gauglitz GG, Jeschke MG. Combined gene and stem cell therapy for cutaneous wound healing. Mol Pharm. 2011;8:1471-1479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 32. | Gizaw M, Faglie A, Pieper M, Poudel S, Chou SF. The Role of Electrospun Fiber Scaffolds in Stem Cell Therapy for Skin Tissue Regeneration. Med One. 2019;4:e190002. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 33. | Sarasúa JG, López SP, Viejo MA, Basterrechea MP, Rodríguez AF, Gutiérrez AF, Gala JG, Menéndez YM, Augusto DE, Arias AP, Hernández JO. Treatment of pressure ulcers with autologous bone marrow nuclear cells in patients with spinal cord injury. J Spinal Cord Med. 2011;34:301-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 36] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 34. | Goodarzi P, Alavi-Moghadam S, Sarvari M, Tayanloo Beik A, Falahzadeh K, Aghayan H, Payab M, Larijani B, Gilany K, Rahim F, Adibi H, Arjmand B. Adipose Tissue-Derived Stromal Cells for Wound Healing. Adv Exp Med Biol. 2018;1119:133-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 35. | Gupta GJ, Karki K, Jain P, Saxena AK. Autologous Bone Marrow Aspirate Therapy for Skin Tissue Engineering and Tissue Regeneration. Adv Wound Care (New Rochelle). 2017;6:135-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 36. | Hanson SE, Bentz ML, Hematti P. Mesenchymal stem cell therapy for nonhealing cutaneous wounds. Plast Reconstr Surg. 2010;125:510-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 104] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 37. | Hashemi SS, Mohammadi AA, Kabiri H, Hashempoor MR, Mahmoodi M, Amini M, Mehrabani D. The healing effect of Wharton's jelly stem cells seeded on biological scaffold in chronic skin ulcers: A randomized clinical trial. J Cosmet Dermatol. 2019;18:1961-1967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 57] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 38. | Hassan WU, Greiser U, Wang W. Role of adipose-derived stem cells in wound healing. Wound Repair Regen. 2014;22:313-325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 261] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 39. | Hocking AM, Gibran NS. Mesenchymal stem cells: paracrine signaling and differentiation during cutaneous wound repair. Exp Cell Res. 2010;316:2213-2219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 317] [Cited by in RCA: 284] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 40. | Holm JS, Toyserkani NM, Sorensen JA. Adipose-derived stem cells for treatment of chronic ulcers: current status. Stem Cell Res Ther. 2018;9:142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 42] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 41. | Jadlowiec C, Brenes RA, Li X, Lv W, Protack CD, Collins MJ, Dardik A. Stem cell therapy for critical limb ischemia: what can we learn from cell therapy for chronic wounds? Vascular. 2012;20:284-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 42. | Kosaric N, Kiwanuka H, Gurtner GC. Stem cell therapies for wound healing. Expert Opin Biol Ther. 2019;19:575-585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 112] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 43. | Larsen L, Tchanque-Fossuo CN, Gorouhi F, Boudreault D, Nguyen C, Fuentes JJ, Crawford RW, Dahle SE, Whetzel T, Isseroff RR. Combination therapy of autologous adipose mesenchymal stem cell-enriched, high-density lipoaspirate and topical timolol for healing chronic wounds. J Tissue Eng Regen Med. 2018;12:186-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 44. | Locke M, Windsor J, Dunbar PR. Human adipose-derived stem cells: isolation, characterization and applications in surgery. ANZ J Surg. 2009;79:235-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 184] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 45. | Marfia G, Navone SE, Di Vito C, Ughi N, Tabano S, Miozzo M, Tremolada C, Bolla G, Crotti C, Ingegnoli F, Rampini P, Riboni L, Gualtierotti R, Campanella R. Mesenchymal stem cells: potential for therapy and treatment of chronic non-healing skin wounds. Organogenesis. 2015;11:183-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 81] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 46. | Maxson S, Lopez EA, Yoo D, Danilkovitch-Miagkova A, Leroux MA. Concise review: role of mesenchymal stem cells in wound repair. Stem Cells Transl Med. 2012;1:142-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 604] [Cited by in RCA: 563] [Article Influence: 43.3] [Reference Citation Analysis (0)] |
| 47. | Mulder GD, Lee DK, Jeppesen NS. Comprehensive review of the clinical application of autologous mesenchymal stem cells in the treatment of chronic wounds and diabetic bone healing. Int Wound J. 2012;9:595-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 48. | Ojeh N, Pastar I, Tomic-Canic M, Stojadinovic O. Stem Cells in Skin Regeneration, Wound Healing, and Their Clinical Applications. Int J Mol Sci. 2015;16:25476-25501. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 172] [Cited by in RCA: 194] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 49. | Pourmoussa A, Gardner DJ, Johnson MB, Wong AK. An update and review of cell-based wound dressings and their integration into clinical practice. Ann Transl Med. 2016;4:457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 50. | Rezaie F, Momeni-Moghaddam M, Naderi-Meshkin H. Regeneration and Repair of Skin Wounds: Various Strategies for Treatment. Int J Low Extrem Wounds. 2019;18:247-261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 51. | Roşca AM, Ţuţuianu R, Titorencu ID. Mesenchymal stromal cells derived exosomes as tools for chronic wound healing therapy. Rom J Morphol Embryol. 2018;59:655-662. [PubMed] |
| 52. | Teng M, Huang Y, Zhang H. Application of stems cells in wound healing--an update. Wound Repair Regen. 2014;22:151-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 53. | Veith AP, Henderson K, Spencer A, Sligar AD, Baker AB. Therapeutic strategies for enhancing angiogenesis in wound healing. Adv Drug Deliv Rev. 2019;146:97-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 586] [Article Influence: 97.7] [Reference Citation Analysis (0)] |
| 54. | Wettstein R, Savic M, Pierer G, Scheufler O, Haug M, Halter J, Gratwohl A, Baumberger M, Schaefer DJ, Kalbermatten DF. Progenitor cell therapy for sacral pressure sore: a pilot study with a novel human chronic wound model. Stem Cell Res Ther. 2014;5:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 55. | Yao B, Huang S, Gao D, Xie J, Liu N, Fu X. Age-associated changes in regenerative capabilities of mesenchymal stem cell: impact on chronic wounds repair. Int Wound J. 2016;13:1252-1259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 56. | Zou JP, Huang S, Peng Y, Liu HW, Cheng B, Fu XB, Xiang XF. Mesenchymal stem cells/multipotent mesenchymal stromal cells (MSCs): potential role in healing cutaneous chronic wounds. Int J Low Extrem Wounds. 2012;11:244-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 57. | You HJ, Han SK, Rhie JW. Randomised controlled clinical trial for autologous fibroblast-hyaluronic acid complex in treating diabetic foot ulcers. J Wound Care. 2014;23:521-522, 524, 526-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 41] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 58. | Digirolamo CM, Stokes D, Colter D, Phinney DG, Class R, Prockop DJ. Propagation and senescence of human marrow stromal cells in culture: a simple colony-forming assay identifies samples with the greatest potential to propagate and differentiate. Br J Haematol. 1999;107:275-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 670] [Cited by in RCA: 656] [Article Influence: 25.2] [Reference Citation Analysis (0)] |
| 59. | Sterodimas A, de Faria J, Nicaretta B, Pitanguy I. Tissue engineering with adipose-derived stem cells (ADSCs): current and future applications. J Plast Reconstr Aesthet Surg. 2010;63:1886-1892. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 187] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 60. | Bourin P, Bunnell BA, Casteilla L, Dominici M, Katz AJ, March KL, Redl H, Rubin JP, Yoshimura K, Gimble JM. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: a joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy. 2013;15:641-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1199] [Cited by in RCA: 1358] [Article Influence: 113.2] [Reference Citation Analysis (2)] |
| 61. | Kim WS, Park BS, Sung JH, Yang JM, Park SB, Kwak SJ, Park JS. Wound healing effect of adipose-derived stem cells: a critical role of secretory factors on human dermal fibroblasts. J Dermatol Sci. 2007;48:15-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 602] [Cited by in RCA: 626] [Article Influence: 34.8] [Reference Citation Analysis (0)] |
| 62. | Trottier V, Marceau-Fortier G, Germain L, Vincent C, Fradette J. IFATS collection: Using human adipose-derived stem/stromal cells for the production of new skin substitutes. Stem Cells. 2008;26:2713-2723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 163] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 63. | Rigotti G, Marchi A, Galiè M, Baroni G, Benati D, Krampera M, Pasini A, Sbarbati A. Clinical treatment of radiotherapy tissue damage by lipoaspirate transplant: a healing process mediated by adipose-derived adult stem cells. Plast Reconstr Surg. 2007;119:1409-1422; discussion 1423-1424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 782] [Cited by in RCA: 765] [Article Influence: 42.5] [Reference Citation Analysis (0)] |
| 64. | Han SK, Kim HR, Kim WK. The treatment of diabetic foot ulcers with uncultured, processed lipoaspirate cells: a pilot study. Wound Repair Regen. 2010;18:342-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 86] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 65. | Lee HC, An SG, Lee HW, Park JS, Cha KS, Hong TJ, Park JH, Lee SY, Kim SP, Kim YD, Chung SW, Bae YC, Shin YB, Kim JI, Jung JS. Safety and effect of adipose tissue-derived stem cell implantation in patients with critical limb ischemia: a pilot study. Circ J. 2012;76:1750-1760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 195] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 66. | Raposio E, Bertozzi N, Bonomini S, Bernuzzi G, Formentini A, Grignaffini E, Pio Grieco M. Adipose-derived Stem Cells Added to Platelet-rich Plasma for Chronic Skin Ulcer Therapy. Wounds. 2016;28:126-131. [PubMed] |
| 67. | Carstens MH, Gómez A, Cortés R, Turner E, Pérez C, Ocon M, Correa D. Non-reconstructable peripheral vascular disease of the lower extremity in ten patients treated with adipose-derived stromal vascular fraction cells. Stem Cell Res. 2017;18:14-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 42] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 68. | Chopinaud M, Labbé D, Creveuil C, Marc M, Bénateau H, Mourgeon B, Chopinaud E, Veyssière A, Dompmartin A. Autologous Adipose Tissue Graft to Treat Hypertensive Leg Ulcer: A Pilot Study. Dermatology. 2017;233:234-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 19] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 69. | Konstantinow A, Arnold A, Djabali K, Kempf W, Gutermuth J, Fischer T, Biedermann T. Therapy of ulcus cruris of venous and mixed venous arterial origin with autologous, adult, native progenitor cells from subcutaneous adipose tissue: a prospective clinical pilot study. J Eur Acad Dermatol Venereol. 2017;31:2104-2118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 70. | Darinskas A, Paskevicius M, Apanavicius G, Vilkevicius G, Labanauskas L, Ichim TE, Rimdeika R. Stromal vascular fraction cells for the treatment of critical limb ischemia: a pilot study. J Transl Med. 2017;15:143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 71. | Marino G, Moraci M, Armenia E, Orabona C, Sergio R, De Sena G, Capuozzo V, Barbarisi M, Rosso F, Giordano G, Iovino F, Barbarisi A. Therapy with autologous adipose-derived regenerative cells for the care of chronic ulcer of lower limbs in patients with peripheral arterial disease. J Surg Res. 2013;185:36-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 97] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 72. | García-Olmo D, García-Arranz M, Herreros D, Pascual I, Peiro C, Rodríguez-Montes JA. A phase I clinical trial of the treatment of Crohn's fistula by adipose mesenchymal stem cell transplantation. Dis Colon Rectum. 2005;48:1416-1423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 604] [Cited by in RCA: 571] [Article Influence: 28.6] [Reference Citation Analysis (0)] |
| 73. | Garg RK, Rennert RC, Duscher D, Sorkin M, Kosaraju R, Auerbach LJ, Lennon J, Chung MT, Paik K, Nimpf J, Rajadas J, Longaker MT, Gurtner GC. Capillary force seeding of hydrogels for adipose-derived stem cell delivery in wounds. Stem Cells Transl Med. 2014;3:1079-1089. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 79] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 74. | Friedenstein AJ, Piatetzky-Shapiro II, Petrakova KV. Osteogenesis in transplants of bone marrow cells. J Embryol Exp Morphol. 1966;16:381-390. [PubMed] |
| 75. | Humpert PM, Bärtsch U, Konrade I, Hammes HP, Morcos M, Kasper M, Bierhaus A, Nawroth PP. Locally applied mononuclear bone marrow cells restore angiogenesis and promote wound healing in a type 2 diabetic patient. Exp Clin Endocrinol Diabetes. 2005;113:538-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 76. | Ren H, Cao Y, Zhao Q, Li J, Zhou C, Liao L, Jia M, Zhao Q, Cai H, Han ZC, Yang R, Chen G, Zhao RC. Proliferation and differentiation of bone marrow stromal cells under hypoxic conditions. Biochem Biophys Res Commun. 2006;347:12-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 156] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 77. | Stoff A, Rivera AA, Sanjib Banerjee N, Moore ST, Michael Numnum T, Espinosa-de-Los-Monteros A, Richter DF, Siegal GP, Chow LT, Feldman D, Vasconez LO, Michael Mathis J, Stoff-Khalili MA, Curiel DT. Promotion of incisional wound repair by human mesenchymal stem cell transplantation. Exp Dermatol. 2009;18:362-369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 90] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 78. | Badiavas EV, Falanga V. Treatment of chronic wounds with bone marrow-derived cells. Arch Dermatol. 2003;139:510-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 321] [Cited by in RCA: 281] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 79. | Lu D, Chen B, Liang Z, Deng W, Jiang Y, Li S, Xu J, Wu Q, Zhang Z, Xie B, Chen S. Comparison of bone marrow mesenchymal stem cells with bone marrow-derived mononuclear cells for treatment of diabetic critical limb ischemia and foot ulcer: a double-blind, randomized, controlled trial. Diabetes Res Clin Pract. 2011;92:26-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 390] [Cited by in RCA: 326] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 80. | Dash NR, Dash SN, Routray P, Mohapatra S, Mohapatra PC. Targeting nonhealing ulcers of lower extremity in human through autologous bone marrow-derived mesenchymal stem cells. Rejuvenation Res. 2009;12:359-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 227] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 81. | Rogers LC, Bevilacqua NJ, Armstrong DG. The use of marrow-derived stem cells to accelerate healing in chronic wounds. Int Wound J. 2008;5:20-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 82. | Vojtassák J, Danisovic L, Kubes M, Bakos D, Jarábek L, Ulicná M, Blasko M. Autologous biograft and mesenchymal stem cells in treatment of the diabetic foot. Neuro Endocrinol Lett. 2006;27 Suppl 2:134-137. [PubMed] |
| 83. | Yoshikawa T, Mitsuno H, Nonaka I, Sen Y, Kawanishi K, Inada Y, Takakura Y, Okuchi K, Nonomura A. Wound therapy by marrow mesenchymal cell transplantation. Plast Reconstr Surg. 2008;121:860-877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 187] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 84. | Ravari H, Hamidi-Almadari D, Salimifar M, Bonakdaran S, Parizadeh MR, Koliakos G. Treatment of non-healing wounds with autologous bone marrow cells, platelets, fibrin glue and collagen matrix. Cytotherapy. 2011;13:705-711. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 61] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 85. | Vahidy FS, Rahbar MH, Zhu H, Rowan PJ, Bambhroliya AB, Savitz SI. Systematic Review and Meta-Analysis of Bone Marrow-Derived Mononuclear Cells in Animal Models of Ischemic Stroke. Stroke. 2016;47:1632-1639. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 61] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 86. | Jain P, Perakath B, Jesudason MR, Nayak S. The effect of autologous bone marrow-derived cells on healing chronic lower extremity wounds: results of a randomized controlled study. Ostomy Wound Manage. 2011;57:38-44. [PubMed] |
| 87. | Yamaguchi Y, Yoshida S, Sumikawa Y, Kubo T, Hosokawa K, Ozawa K, Hearing VJ, Yoshikawa K, Itami S. Rapid healing of intractable diabetic foot ulcers with exposed bones following a novel therapy of exposing bone marrow cells and then grafting epidermal sheets. Br J Dermatol. 2004;151:1019-1028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 41] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 88. | Wong VW, Levi B, Rajadas J, Longaker MT, Gurtner GC. Stem cell niches for skin regeneration. Int J Biomater. 2012;2012:926059. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 67] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 89. | Yang R, Liu F, Wang J, Chen X, Xie J, Xiong K. Epidermal stem cells in wound healing and their clinical applications. Stem Cell Res Ther. 2019;10:229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 104] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 90. | Bauer JW, Koller J, Murauer EM, De Rosa L, Enzo E, Carulli S, Bondanza S, Recchia A, Muss W, Diem A, Mayr E, Schlager P, Gratz IK, Pellegrini G, De Luca M. Closure of a Large Chronic Wound through Transplantation of Gene-Corrected Epidermal Stem Cells. J Invest Dermatol. 2017;137:778-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 101] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 91. | Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17989] [Cited by in RCA: 18081] [Article Influence: 951.6] [Reference Citation Analysis (0)] |
| 92. | Yonezawa M, Tanizaki H, Inoguchi N, Ishida M, Katoh M, Tachibana T, Miyachi Y, Kubo K, Kuroyanagi Y. Clinical study with allogeneic cultured dermal substitutes for chronic leg ulcers. Int J Dermatol. 2007;46:36-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 93. | Krishnaswamy VR, Korrapati PS. Role of dermatopontin in re-epithelialization: implications on keratinocyte migration and proliferation. Sci Rep. 2014;4:7385. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 94. | Lampert IA, Suitters AJ, Chisholm PM. Expression of Ia antigen on epidermal keratinocytes in graft-versus-host disease. Nature. 1981;293:149-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 195] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 95. | Vanscheidt W, Ukat A, Horak V, Brüning H, Hunyadi J, Pavlicek R, Emter M, Hartmann A, Bende J, Zwingers T, Ermuth T, Eberhardt R. Treatment of recalcitrant venous leg ulcers with autologous keratinocytes in fibrin sealant: a multinational randomized controlled clinical trial. Wound Repair Regen. 2007;15:308-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 50] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 96. | Moustafa M, Bullock AJ, Creagh FM, Heller S, Jeffcoate W, Game F, Amery C, Tesfaye S, Ince Z, Haddow DB, MacNeil S. Randomized, controlled, single-blind study on use of autologous keratinocytes on a transfer dressing to treat nonhealing diabetic ulcers. Regen Med. 2007;2:887-902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 55] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 97. | Hartmann A, Quist J, Hamm H, Bröcker EB, Friedl P. Transplantation of autologous keratinocyte suspension in fibrin matrix to chronic venous leg ulcers: improved long-term healing after removal of the fibrin carrier. Dermatol Surg. 2008;34:922-929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 98. | De Luca M, Albanese E, Bondanza S, Megna M, Ugozzoli L, Molina F, Cancedda R, Santi PL, Bormioli M, Stella M. Multicentre experience in the treatment of burns with autologous and allogenic cultured epithelium, fresh or preserved in a frozen state. Burns. 1989;15:303-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 189] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 99. | Bayram Y, Deveci M, Imirzalioglu N, Soysal Y, Sengezer M. The cell based dressing with living allogenic keratinocytes in the treatment of foot ulcers: a case study. Br J Plast Surg. 2005;58:988-996. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 100. | Beele H, de la Brassine M, Lambert J, Suys E, De Cuyper C, Decroix J, Boyden B, Tobback L, Hulstaert F, De Schepper S, Brissinck J, Delaey B, Draye JP, De Deene A, De Waele P, Verbeken G. A prospective multicenter study of the efficacy and tolerability of cryopreserved allogenic human keratinocytes to treat venous leg ulcers. Int J Low Extrem Wounds. 2005;4:225-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 101. | Shukla VK, Tiwary SK, Barnwal S, Gulati AK, Pandey SS. Effect of autologous epidermal cell suspension transplantation in chronic nonhealing wounds: a pilot study. Can J Surg. 2010;53:6-10. [PubMed] |
| 102. | Guenou H, Nissan X, Larcher F, Feteira J, Lemaitre G, Saidani M, Del Rio M, Barrault CC, Bernard FX, Peschanski M, Baldeschi C, Waksman G. Human embryonic stem-cell derivatives for full reconstruction of the pluristratified epidermis: a preclinical study. Lancet. 2009;374:1745-1753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 168] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 103. | Devolder K, Savulescu J. The moral imperative to conduct embryonic stem cell and cloning research. Camb Q Healthc Ethics. 2006;15:7-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 104. | Okano H, Nakamura M, Yoshida K, Okada Y, Tsuji O, Nori S, Ikeda E, Yamanaka S, Miura K. Steps toward safe cell therapy using induced pluripotent stem cells. Circ Res. 2013;112:523-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 301] [Cited by in RCA: 300] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 105. | You HJ, Han SK. Cell therapy for wound healing. J Korean Med Sci. 2014;29:311-319. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 109] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 106. | Bura A, Planat-Benard V, Bourin P, Silvestre JS, Gross F, Grolleau JL, Saint-Lebese B, Peyrafitte JA, Fleury S, Gadelorge M, Taurand M, Dupuis-Coronas S, Leobon B, Casteilla L. Phase I trial: the use of autologous cultured adipose-derived stroma/stem cells to treat patients with non-revascularizable critical limb ischemia. Cytotherapy. 2014;16:245-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 216] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 107. | Yamada N, Uchinuma E, Kuroyanagi Y. Clinical trial of allogeneic cultured dermal substitutes for intractable skin ulcers. J Artif Organs. 2012;15:193-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 108. | Marcelo D, Beatriz PM, Jussara R, Fabiana B. Tissue therapy with autologous dermal and epidermal culture cells for diabetic foot ulcers. Cell Tissue Bank. 2012;13:241-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 109. | Teepe RG, Roseeuw DI, Hermans J, Koebrugge EJ, Altena T, de Coninck A, Ponec M, Vermeer BJ. Randomized trial comparing cryopreserved cultured epidermal allografts with hydrocolloid dressings in healing chronic venous ulcers. J Am Acad Dermatol. 1993;29:982-988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 45] [Article Influence: 1.4] [Reference Citation Analysis (0)] |