修回日期: 2003-03-15
接受日期: 2003-03-26
在线出版日期: 2003-09-15
探讨乙型肝炎病毒(HBV) X基因变异在肝癌发生中的作用.
应用聚合酶链反应(PCR)技术从5例肝细胞癌患者及12例慢性乙型肝炎患者血清中扩增X基因, 然后将纯化的PCR产物进行序列测定, 对获得的序列进行变异性及同源性分析.
肝细胞癌患者中得到的HBV X基因序列与Genebank中多个已发表的HBV各血清亚型序列进行同源性比较, 发现核苷酸同源性在89-96 %之间, 血清亚型以adw为主. 与非肝癌组的比较发现X基因序列的替换突变中存在4种形式. 6个位点的变异仅出现于肝癌组, 12个热点变异多见于肝癌组而少见于非肝癌组.
X基因序列的突变与肝细胞癌的发生有关.
引文著录: 代志琰, 徐启桓, 李刚, 马会慧, 汤正好, 舒欣, 姚集鲁. 肝癌患者乙型肝炎病毒X基因变异的研究. 世界华人消化杂志 2003; 11(9): 1349-1352
Revised: March 15, 2003
Accepted: March 26, 2003
Published online: September 15, 2003
To study the association of mutations of hepatitis B virus(HBV) X gene with hepatocellular carcinoma(HCC).
The complete X region of HBV from the serum of 5 patients with HCC and 12 patients with chronic hepatitis B (CH) was amplified by PCR and then the purified PCR products were sequenced.
A comparison of 492bp nucleotide sequence of the HBV X gene from 5 HCC patients with that of the several previously reported isolates of different serotype in Genebank showed that the nucleotide homology was 89-96 % and most of the strains were similar to serotype adw. The number of nucleotide changes in the X gene of HBV from HCC group (19.4±12.1) was significantly greater than that from the CH group (4.4±2.4, P =0.00 093).There were 4 models of substitutions in X gene and 6 hot spots found in HCC group and 12 hot spots found in HCC group.
The mutations of HBV X gene may be closely associated with the development of HCC.
- Citation: Dai ZY, Xu QH, Li G, Ma HH, Tang ZH, Shu X, Yao JL. Study on X gene mutation of hepatitis B virus from patients with hepatocellular carcinoma. Shijie Huaren Xiaohua Zazhi 2003; 11(9): 1349-1352
- URL: https://www.wjgnet.com/1009-3079/full/v11/i9/1349.htm
- DOI: https://dx.doi.org/10.11569/wcjd.v11.i9.1349
乙型肝炎病毒(HBV)基因组含有4个开放读码框架[1], 分别为S, C, P, X区. 对HBV X基因的研究集中其在肝癌发生中的重要作用[2-8]. 我们以X区为研究靶区域, 应用聚合酶链反应(PCR)扩增肝细胞癌(HCC)患者血清中的HBV X基因, 将获得的序列与来自基因库及非HCC的慢性乙型肝炎患者血清中HBVX基因序列进行比较, 了解X基因突变与HCC的关系.
我院2002-01-12 住院患者HCC组5例, 年龄48±17岁, 男4例, 女1例, ALT102.6±40.0 IU/L, HBVDNA 1.1±1.9×107 copies/mL, 两对半(A为HBsAg、HBeAg、抗-HBc阳性; B为HBsAg、抗-HBe、抗-HBc阳性)A3例, B2例; 非HCC组 12例, 年龄29±10岁, 男12例, 女1例, ALT 257.9±625.4 IU/L, HBVDNA 5.4±7.0×108 copies/mL , 两对半A10例, B2例. 诊断标准按照2000年西安第10次全国病毒性肝炎及肝病学术会议制定的《病毒性肝炎防治方案》, 患者均无应用HBV疫苗史和抗病毒治疗史, 其他肝炎病毒标志物检测阴性.
取血清100 mL加TES 300 mL (10 mmol/L Tris-HCl, pH 8.0, 0.5 mmol/L EDTA, 5 g/L SDS, 150 mg/L蛋白酶K), 65 °C 3 h, 常规酚:氯仿:异戊醇 (V:V:V; 25:24:1)抽提, 乙醇沉淀, DNA溶解于双蒸水20 mL中, -20 °C保存备用. 设计引物, P1: 5-CCA AGC TTC ATC ATT TCC ATG G-3; P2: 5-TGG ATC CAA GAG ATG ATT AGG-3; P3: 5-TTT TGC TCG CAG CCG GTC T-3; P4: 5 - TGC TTG AAC AGT AGG ACA TG-3. 引物由北京赛百盛公司合成. 肝癌组扩增用巢式PCR(其中仅1号用1次PCR, 引物为P3、P4), 即外引物为P3、P4, 内引物为P1、P2, 目的片段长度为492个bp; 非肝癌组用1次PCR, 引物为P3、P4 , 目的片段长度为578个bp. PCR参数如下: 94 °C 5 min预变性, 94 °C 30 s变性, 53 °C40 s退火, 72 °C 1 min延伸, 共32个循环, 72 °C总延伸10 min. 选HBV全基因组质粒(本室保存)相同条件扩增产物作为阳性对照, 健康人血清(两对半及血清HBVDNA均阴性)作为阴性对照. 将PCR产物在20 g/L的琼脂糖凝胶中电泳, 切取目的片段, 经QIAqiuck Gel Extraction Kit (QIAGEN, 德国 )回收DNA, 测序由大连宝生物工程公司完成. 序列变异性及同源性分析用DNAstar软件分析.
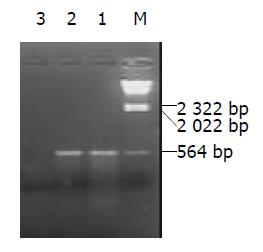
从患者血清中扩增出HBV X 基因, 电泳后可见PCR产物长度约578 bp, 与预期大小相符(图1). 肝癌组与非肝癌组X基因序列同源性比较的百分比值如表1.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | |
| 1 | 92.9 | 97.8 | 92.9 | 91.7 | 95.7 | 90.4 | 90.9 | 90.7 | 91.1 | 97.6 | 90.0 | 88.4 | 91.3 | 91.1 | 90.7 | 91.3 | |
| 2 | 91.7 | 97.8 | 96.1 | 90.4 | 94.7 | 97.0 | 96.7 | 96.7 | 90.7 | 95.5 | 94.1 | 97.0 | 96.7 | 96.1 | 95.7 | ||
| 3 | 91.7 | 91.5 | 95.7 | 90.2 | 90.7 | 90.4 | 90.9 | 95.9 | 89.8 | 88.2 | 90.9 | 91.1 | 90.4 | 91.1 | |||
| 4 | 97.0 | 89.4 | 95.3 | 96.7 | 96.1 | 96.1 | 90.4 | 94.9 | 93.7 | 96.3 | 96.1 | 95.7 | 96.5 | ||||
| 5 | 90.5 | 97.4 | 98.1 | 97.9 | 97.9 | 91.7 | 97.1 | 93.1 | 98.3 | 97.9 | 97.8 | 98.4 | |||||
| 6 | 90.0 | 90.8 | 91.3 | 91.7 | 97.9 | 90.7 | 86.9 | 91.7 | 91.3 | 91.0 | 90.8 | ||||||
| 7 | 97.2 | 97.6 | 97.4 | 91.0 | 96.9 | 93.3 | 97.8 | 97.8 | 97.1 | 97.9 | |||||||
| 8 | 98.8 | 98.8 | 90.8 | 97.9 | 94.1 | 99.1 | 98.8 | 98.6 | 98.3 | ||||||||
| 9 | 99.3 | 91.3 | 98.1 | 94.1 | 99.3 | 99.0 | 98.6 | 98.4 | |||||||||
| 10 | 91.7 | 98.1 | 94.1 | 99.3 | 99.0 | 98.8 | 98.4 | ||||||||||
| 11 | 90.7 | 86.9 | 91.7 | 91.3 | 91.2 | 91.9 | |||||||||||
| 12 | 94.1 | 98.4 | 98.4 | 97.9 | 97.6 | ||||||||||||
| 13 | 94.5 | 94.5 | 93.9 | 93.6 | |||||||||||||
| 14 | 99.3 | 99.1 | 98.8 | ||||||||||||||
| 15 | 98.8 | 98.4 | |||||||||||||||
| 16 | 98.6 |
肝癌组 5例所得序列与Genebank 中HBV多个已发表的各血清亚型序列进行同源性比较, 发现5例患者与多个已知分离株的相应序列的核苷酸同源性在89-96 %之间, 推导的氨基酸序列同源性在80-99%, 其中 3例为adw亚型, 2例为ayr亚型. 非肝癌组12例慢性乙型肝炎患者与多个已知分离株的相应序列的核苷酸同源性在87-96 %之间, 推导的氨基酸序列同源性在86-98 %, 其中 10例为adw亚型, 2例为ayr亚型.
肝癌组序列分析发现, HBV X区的替换突变表现为散在分布, 但相对集中于核苷酸序列的nt 1 358-1 365, nt 1 490-1 502, nt 1 634-1 675, nt 1 721-1 801四个区域, 分析核苷酸序列时发现替换突变中存在4种形式, 第1种形式是突变具有广泛性, HBV X基因序列发生变异位点数肝癌组高于非肝癌组(分别为19.4±12.1, 4.4±2.4, P =0.00 093), 多个患者的核苷酸序列在某个位点均突变为同一的碱基, 如肝癌组5例标本中有4例nt 1 359 A→C, nt 1 362 T→G, nt 1 363 A→C, nt 1 365 A→T, nt 1 813 T→C, nt 1 859 A→T, 且此类变异仅出现于肝癌组而不见于非肝癌组; 第2种形式为突变具有个体特异性, 如患者5在nt 1 440 G→T, 在nt 1 705 A→G, 患者14在nt 1 564 C→T, 患者17在nt 1 599 C→G; 第3种形式存在热点变异, 我们结合两组病例, 对492个碱基替换的频率进行统计, 有12处位点为高发突变位点(17例中有5例以上在该位点发生替换, 频率大于28.8%), 即热点变异, 且此类变异多出现于肝癌组而少见于非肝癌组见表2; 第4种形式为散在的随机替换, 最为常见. HBV X区的直接重复序列 (DR2, nt 1 590-1 600; DR1, nt 1 824-1 834)、基本核心启动子(BCP)的第3个AT富集区(nt 1 789-1 795) 在全部肝癌组5例和非肝癌组12例中均无变异发生. T1 764 A1 766联合突变见于肝癌组4例和非肝癌组3例中.
| 碱基变异位点 | 肝癌组 | 非肝癌组 | 合计 |
| nt 1 361 G→A | 4 | 2 | 6 |
| nt 1 466 C→T | 3 | 2 | 5 |
| nt 1 490 C→T | 3 | 2 | 5 |
| nt 1 510 A→T | 3 | 2 | 5 |
| nt 1 640 C→T | 3 | 2 | 5 |
| nt 1 644 G→A | 3 | 2 | 5 |
| nt 1 764 A→T | 4 | 3 | 7 |
| nt 1 766 G→A | 4 | 3 | 7 |
| nt 1 859 A→T | 5 | 2 | 7 |
HBV感染在HCC发生中起着重要作用[9-19]. HBV的变异是较普遍的现象, 可能与其聚合酶缺乏校正功能, 以及HBV 承受宿主巨大的免疫压力有关. HBV X 蛋白是一种具有反式作用的调节蛋白, 能直接或间接激活c-myc、c-fos、c-jun和ras等肿瘤基因[20-23], 他不仅能活化HBV 自身的启动子, 而且能活化一些异源病毒和细胞启动子, 参与对细胞的基因表达的调控[24-29], 从而与HCC的发生发展相关.
HBV进入人体后病毒基因组发生相对较为特殊的突变[30,31], 以最大限度地适应宿主环境, 从而得以长期在体内持续存在. 我们通过应用PCR扩增、序列测定等分子生物学技术, 获得5份HCC及12份非HCC标本中HBV X基因的核苷酸序列, 与Genebank 中HBV多个已发表的各血清亚型序列进行同源性比较, 发现HCC组与非HCC组的HBV亚型分布类似, 核苷酸及推导的氨基酸序列同源性分析均提示以adw 亚型为主. HBV X基因序列的变异性分析则发现HCC组与非HCC组的HBV X区突变有所不同, HBV X基因序列发生变异的位点数HCC组高于非HCC组, HCC标本中HBV X基因序列的变异具有一定的特征, 有6处变异仅出现于HCC组而不见于非HCC组, 有12处位点为高发突变位点, 多见于HCC组, 而少见于非HCC组, 由此推断这些热点位点的突变可能在HCC的发生中起重要作用.
T1764A1766联合突变常见于HCC患者, 本研究也在HCC组4例和非HCC组3例中测出此类变异, 而这两处突变正处在增强子2和C启动子序列中, 位于反式转录激活必需序列的上游. 目前认为X 蛋白的反式转录激活作用还包括很强的调节作用, N端1/3为负调节区, C端为正调节区. 因此. 我们推测这两处突变类型可能使反式转录激活作用增强, 包括HBV X 蛋白表达增加, 在致病或致癌过程中起重要作用.
HBV感染易导致慢性化, 而经慢性肝炎与肝硬化进展为HCC是一个缓慢的连续的过程. 通过比较HCC与非HCC标本中HBV X基因核苷酸序列将有助于了解HBV X基因在HCC发生中的作用, 发现HBV X基因在致癌过程中的变异规律是对HCC发病机制研究上的一个飞跃. 虽然, 现有结果不足以阐明病毒变异与病变的因果关系, 但已能明确, HBV X基因核苷酸序列的突变有其规律可循, 与HCC的发生发展有着密切的联系. 进一步的研究须以细胞转染和动物实验相结合, 结合机体免疫对变异株的应答来认识HBV X基因在HCC发生中的作用, 要完全阐明HBV X基因序列的变异与HCC的关系尚须研究更大量的标本.
| 1. | Murakami S. Hepatitis B virus X protein: structure, function and biology. Intervirology. 1999;42:81-99. [DOI] |
| 3. | Xu ZH, Zhao MJ, Li TP. p73 b inhibits transcriptional activities of enhancer I and X promoter in hepatitis B virus more efficiently than p73alpha. World J Gastroenterol. 2002;8:1094-1097. [DOI] |
| 4. | Wang XZ, Jiang XR, Chen XC, Chen ZX, Li D, Lin JY, Tao QM. Seek protein which can interact with hepatitis B virus X protein from human liver cDNA library by yeast two-hybrid system. World J Gastroenterol. 2002;8:95-98. [DOI] |
| 5. | Guo SP, Wang WL, Zhai YQ, Zhao YL. Expression of nuclear factor-kappa B in hepatocellular carcinoma and its relation with the X protein of hepatitis B virus. World J Gastroenterol. 2001;7:340-344. [DOI] |
| 6. | Guo SP, Ma ZS, Wang WL. Construction of eukaryotic expression vector of HBV x gene. World J Gastroenterol. 1999;5:351-352. [DOI] |
| 7. | Xiong J, Yao YC, Zi XY, Li JX, Wang XM, Ye XT, Zhao SM, Yan YB, Yu HY, Hu YP. Expression of hepatitis B virus X protein in transgenic mice. World J Gastroenterol. 2003;9:112-116. [DOI] |
| 8. | Lee JH, Ku JL, Park YJ, Lee KU, Kim WH, Park JG. Establishment and characterization of four human hepatocellular carcinoma cell lines containing hepatitis B virus DNA. World J Gastroenterol. 1999;5:289-295. [DOI] |
| 18. | Staib F, Hussain SP, Hofseth LJ, Wang XW, Harris CC. TP53 and liver carcinogenesis. Hum Mutat. 2003;21:201-216. [DOI] |
| 19. | Jayshree RS, Sridhar H, Devi GM. Surface, core, and X genes of hepatitis B virus in hepatocellular carcinoma: an in situ hybridization study. Cancer. 2003;99:63-67. [DOI] |
| 20. | Nijhara R, Jana SS, Goswami SK, Rana A, Majumdar SS, Kumar V, Sarkar DP. Sustained activation of mitogen-activated protein kinases and activator protein 1 by the hepatitis B virus X protein in mouse hepatocytes in vivo. J Virol. 2001;75:10348-10358. [DOI] |
| 22. | Lakhtakia R, Kumar V, Reddi H, Mathur M, Dattagupta S, Panda SK. Hepatocellular carcinoma in a hepatitis B 'x' transgenic mouse model: A sequential pathological evaluation. J Gastroenterol Hepatol. 2003;18:80-91. [DOI] |
| 23. | Chun E, Lee J, Cheong HS, Lee KY. Tumor eradication by hepatitis B virus X antigen-specific CD8+ T cells in xenografted nude mice. J Immunol. 2003;170:1183-1190. |
| 24. | Kim JH, Rho HM. Activation of the human transforming growth factor alpha (TGF-alpha) gene by the hepatitis B viral X protein (HBx) through AP-2 sites. Mol Cell Biochem. 2002;231:155-161. |
| 25. | Madden CR, Finegold MJ, Slagle BL. Altered DNA mutation spectrum in aflatoxin b1-treated transgenic mice that express the hepatitis B virus x protein. J Virol. 2002;76:11770-11774. [DOI] |
| 26. | Zhu Z, Zhu M, Ni C. Significance of p33(ING1b) and p53 gene expression in hepatocellular carcinoma. Zhonghua Yixue Zazhi. 2002;82:1332-1336. |
| 27. | Li QF, Ou-Yang GL, Peng XX, Hong SG. Effects of tachyplesin on the regulation of cell cycle in human hepatocarcinoma SMMC-7721 cells. World J Gastroenterol. 2003;9:454-458. [DOI] |
| 29. | Sohn S, Jaitovitch-Groisman I, Benlimame N, Galipeau J, Batist G, Alaoui-Jamali MA. Retroviral expression of the hepatitis B virus x gene promotes liver cell susceptibility to carcinogen-induced site specific mutagenesis. Mutat Res. 2000;460:17-28. [DOI] |