Published online Jul 7, 2018. doi: 10.3748/wjg.v24.i25.2741
Peer-review started: March 29, 2018
First decision: May 9, 2018
Revised: June 1, 2018
Accepted: June 25, 2018
Article in press: June 25, 2018
Published online: July 7, 2018
To perform a comprehensive review and provide an up-to-date synopsis of the incidence and trends of inflammatory bowel disease (IBD).
We systematically searched the MEDLINE (source PubMed), EMBASE and Cochrane Library databases in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (period: 1985-2018) to identify studies reporting population-based data on the incidence of pediatric-onset (< 19 years at diagnosis) IBD in full manuscripts. Two authors carried out screening and data extraction. Choropleth interactive maps and temporal trends were used to illustrate the international differences and incidences of and changes in IBD and subtypes.
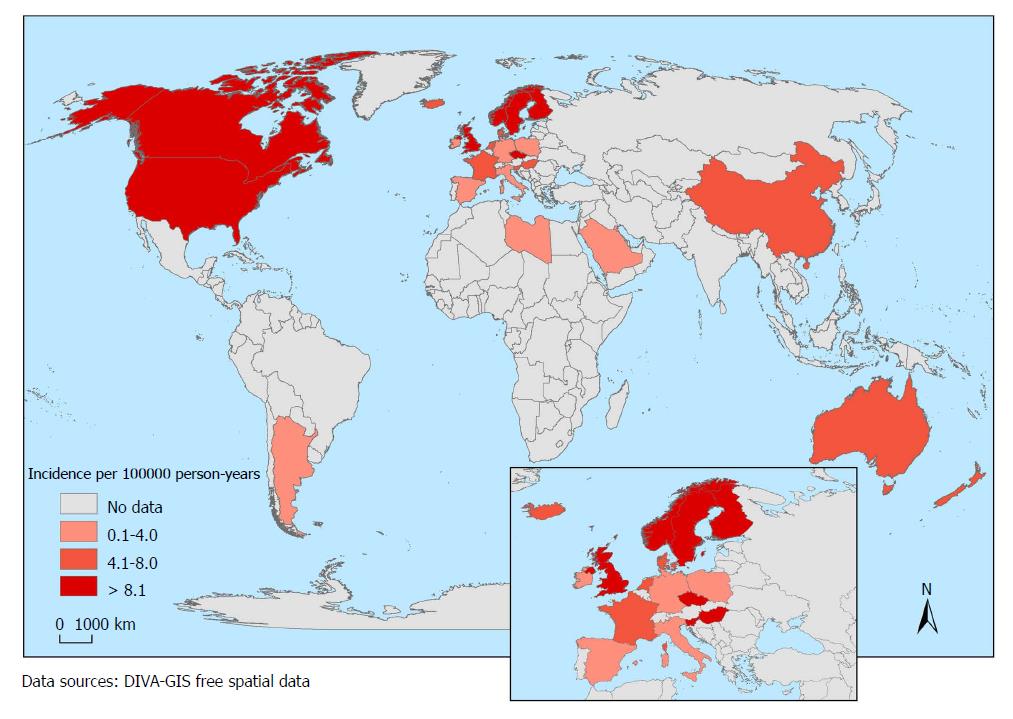
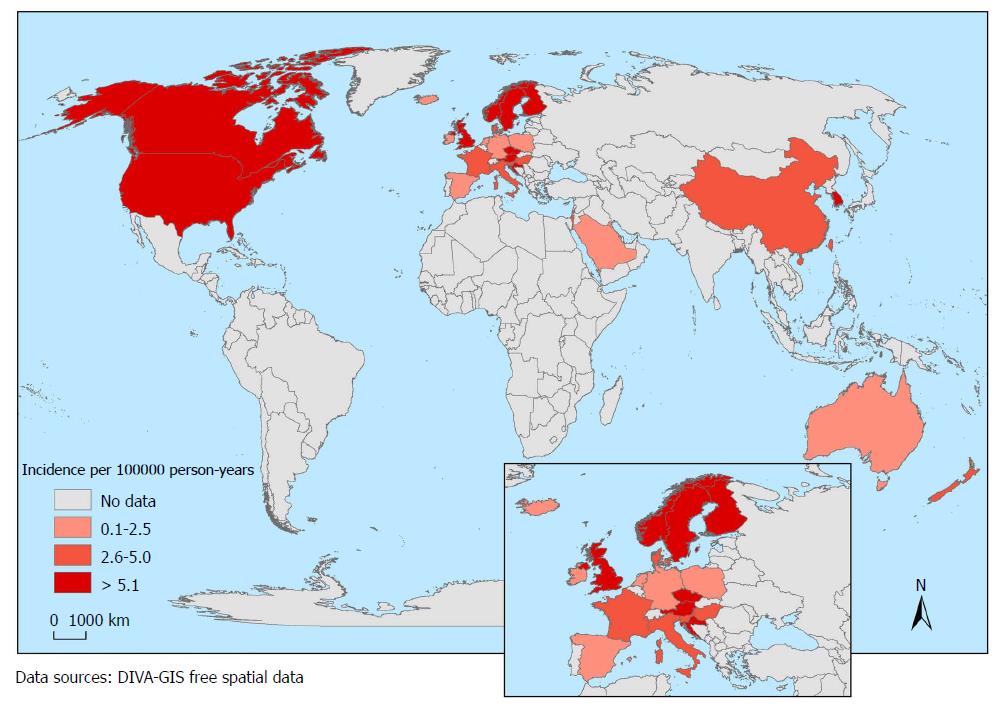
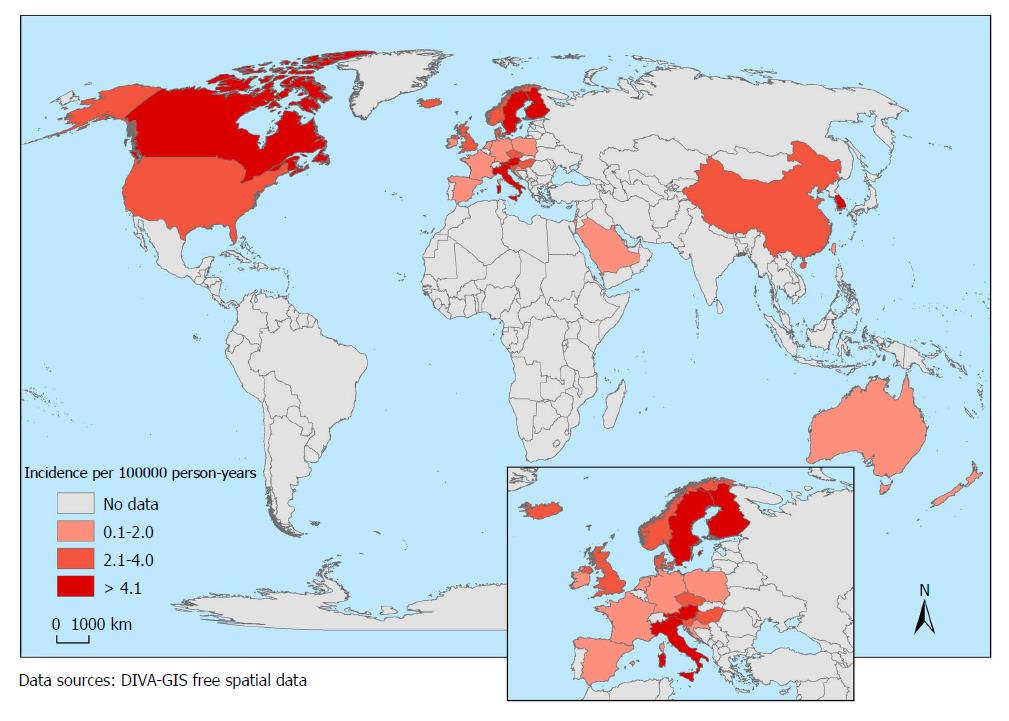
In total, one hundred forty studies reporting data from 38 countries were considered in this review. The highest annual pediatric incidences of IBD were 23/100000 person-years in Europe, 15.2/100000 in North America, and 11.4/100000 in Asia/the Middle East and Oceania. The highest annual incidences of Crohn’s disease (CD) were 13.9/100000 in North America and 12.3/100000 in Europe. The highest annual incidences of ulcerative colitis (UC) were 15.0/100000 in Europe and 10.6/100000 in North America. The highest annual incidences of IBD-unclassified (IBD-U) were 3.6/100000 in Europe and 2.1/100000 in North America. In the time-trend analyses, 67% of CD, 46% of UC and 11% of IBD-U studies reported an increasing incidence (P < 0.05). The risk of IBD is increasing among first-generation of migrant populations.
Globally, the incidence of IBD varies greatly by geographical areas. The steadily increasing incidence of pediatric IBD over time indicates its emergence as a global disease, suggesting that studies should investigate the environmental risk factors among pediatric cohorts.
Core tip: The incidence of inflammatory bowel disease (IBD) is unclear in the pediatric literature. We comprehensively reviewed and critically evaluated population-based and national cohort studies investigating the incidence of IBD and its global trends. One hundred forty studies met the inclusion criteria. The incidence of pediatric-onset IBD has been steadily increasing over time in different geographical areas in both developed and developing regions worldwide, whereas those in the West may have reached a plateau. This indicates the emergence of an IBD epidemic; however, incidence data from developing regions are limited. Exploring the changes and increasing incidence of pediatric IBD may provide new insights into the potential etiology of IBD.
- Citation: Sýkora J, Pomahačová R, Kreslová M, Cvalínová D, Štych P, Schwarz J. Current global trends in the incidence of pediatric-onset inflammatory bowel disease. World J Gastroenterol 2018; 24(25): 2741-2763
- URL: https://www.wjgnet.com/1007-9327/full/v24/i25/2741.htm
- DOI: https://dx.doi.org/10.3748/wjg.v24.i25.2741
Inflammatory bowel disease (IBD) encompasses the following three forms of idiopathic inflammation of the gut: ulcerative colitis (UC), Crohn’s disease (CD) and IBD-unclassified (IBD-U). The differentiation of IBD-U from CD and UC remains difficult; thus, the incidence of IBD-U must be explored. Approximately 25% of patients first present symptoms before the age of 18 years[1], and the incidence in children over 10 years of age is clearly increasing[2]. The incidence of IBD in industrialized countries is higher than that in developing countries; however, overwhelming data suggest that its prevalence is increasing worldwide in both adult[3] and pediatric populations[4] and that its distribution is uneven among regions[5]. Many explanations have been proposed, but the hypothesis that exposure to environmental and genetic factors is a fundamental contributor to the development of IBD has been challenged by several new observations[6-10]. Whether the etiology of pediatric-onset IBD differs from that of adult-onset IBD remains unknown[11]. Thus, there is a great need to summarize global information regarding the pediatric IBD incidence and disease burden in different settings and perform subsequent analyses of the underlying factors.
The aim of this review is to delineate the incidence of pediatric IBD (defined as onset at an age < 19 years) and summarize the latest incidence trends based on a comprehensive review of credible studies.
We conducted a systematic literature search according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)[12]. Articles were identified using computer-stored databases and manual search. A search of English and non-English language journals in the MEDLINE (source PubMed), EMBASE via OvidSP, and Cochrane Library databases was conducted to identify original studies investigating IBD incidence published between January 1, 1985 and March 2018. Suitable papers were subsequently catalogued. The Cochrane library was reviewed. We searched for the following strings according to the MeSH headings: “pediatric”[MeSH Terms] and “inflammatory bowel disease”[All Fields] or “Crohn´s disease”[All Fields] or “ulcerative colitis”[All Fields] or “inflammatory bowel disease unclassified”[All Fields] or “indeterminate colitis”[All Fields] and “incidence”[All Fields] or “children”[MeSH terms] or “adolescents”[MeSH terms] and “population-based studies”[All Fields] or “national registries”[All Fields] or “health administrative database”[All Fields] and “individual country names”[MeSH terms]. The detailed search strategy is shown in Figure 1.
The data extraction was performed independently by two researchers (Schwarz J and Sýkora J) using set criteria to analyze the title and abstract and extract all relevant study-specific data required for the analysis. This strategy was performed iteratively until no more relevant publications were found. During the full-text screening stage, we analyzed papers for inclusion suitability and adequate information regarding the incidence of IBD or calculation of incidence, and a final decision was made about inclusion/exclusion. In addition, the references in the relevant articles were checked by the first author. From each study, the data were extracted and sorted by the following variables: name of the study, leading author, journal, publication year, study period, type of study, age, study location, crude incidence rate and time trends. Double-extracted data were verified, when a disagreement occurred related to data extraction, this was resolved by consensus. Attempts were made to clarify missing data and any uncertainties or errors found in the studies by consulting with the corresponding authors. The rates were extrapolated from the figures if the articles reported data without specifying the numeric rates. The average incidence was calculated if the incidence rates were reported separately for females and males or by race/ethnicity. Reports focusing on immigrant/indigenous populations were analyzed separately based on the respective general population of the area. After the selection of appropriate articles extrapolating the incidence of IBD, a comprehensive review was performed. We only included original studies with a clear case ascertainment. A meta-analysis was not undertaken due to the variability in the included records; thus, a narrative review was conducted. The incidence was measured as the number of cases/100000 person-years, and the temporal evolution was examined to measure the effect of the calendar year on the incidence. All studies were organized by world and regional distribution and subsequently analyzed based on the United Nations (UN) geoscheme devised by the UN Statistics Division[13] regarding the assignment of countries or areas to specific groupings as follows: North America, Europe, Oceania, Asia, South America and Africa. We created interactive (choropleth) maps to visualize the resulting data worldwide and extrapolate the incidence at the country level. Choropleth maps depicting differences in the incidence rates by changes in color were employed. If data were reported for only a region within a country, the entire country was shown on the map.
The quality of the incidence studies was assessed by whether the diagnostic criteria were clearly defined or recognized criteria (Lennard-Jones, Copenhagen criteria) were implemented[14]. The Critical Appraisal Skills Programme was used to appraise the principles of the studies based on the specific methodological designs of the included studies[15]. We extrapolated the incidence at the country level and summarized the incidence rates by jurisdiction in each study. The quality characteristics of each paper are summarized in Table 1. The assessment of the studies yielded either an A or B quality ranking based on the representativeness of the cases. A representative study population was a prerequisite for a study to be included in this systematic review (A). Studies that included populations that were not sampled across the entire country or had a smaller population size were given a B quality ranking. Because all papers were considered to contribute to the determination of the incidence and differences in incidence estimates, we included all studies without a quality assessment of each manuscript.
| Country/Region/Province | Age (yr) | Study period | IBD incidence | CD incidence | UC incidence | IBDU incidence | Setting | Year of publication | First author /reference | Representativeness of the case |
| Western and North Europe | ||||||||||
| Austria/Styria | < 18 | 1997-2007 | 6.7 a | 4.8a | R | 2013 | Petritsch W et al[122] | B | ||
| Denmark/Eastern | < 15 | 1998-2000 | 4.3. | 2.3. | 1.8. | 0.2 | R | 2002 | Urne et al[108] | B |
| Denmark | < 17 | 1995-2013 | P | 2016 | Larsen et al[109] | A | ||||
| 2013 | 9.7a | 6.7a | ||||||||
| Denmark/Eastern | < 15 | 1998-2009 | 6.4a | 3.2a,b | 3.1a,b | 0.2a | R | 2011 | Jakobsen et al[110] | B |
| Denmark | < 15 | 1995-2012 | 3.1a | 2.7a | P | 2014 | Norgard et al[111] | A | ||
| Denmark/Eastern | < 15 | 1998-2000 | 4.3a | 2.3a | 1.8a | 0.2a | P | 2008 | Jakobsen et al[33] | B |
| 2002-2004 | 6.1 | 3.1 | 2.7 | 0.3 | ||||||
| Denmark | < 15 | 1980-2013 | 2.4a | 3.3a | P | 2017 | Lophaven et al[103] | A | ||
| Denmark/Copenhagen County | < 19 | 1962-1987 | 8.6-13.3a | P | 1991 | Langholz et al[106] | B | |||
| Denmark/Copenhagen County | < 14 | 1962-1987 | 2.2a | 0.2a | 2.0a | P | 1997 | Langholz et al[105] | B | |
| Denmark/Copenhagen County | < 14 | 1962-1987 | 0.2 | R | 1992 | Munkholm et al[62] | B | |||
| Denmark/Copenhagen County | < 15 | 1962-1987 | 0.2-3.1a | 2 - 1.6a | P | 2009 | Jakobsen et al[34] | B | ||
| Denmark | < 15 | 1981-1992 | 0.8a | 2.5a | P | 1997 | Fonager et al[107] | A | ||
| Denmark/Northern | < 14 | 1978-2002 | 1.5 | 2.7 | P | 2006 | Jacobsen et al[104] | B | ||
| Denmark/Copenhagen County | < 17 | 2003-2005 | 4.4 | 5.5 | P | 2006 | Vind et al[102] | B | ||
| Finland | < 18 | 1987-2003 | 5a | 2a | 4a | P | 2011 | Lehtinen et al[65] | A | |
| 2003 | 15 | 5 | 9.1 | |||||||
| Finland | < 17 | 1987-2003 | P | 2006 | Turunen et al[64] | A | ||||
| 1987 | 3.9a | 1.7a | 2.2a | |||||||
| 2003 | 7 | 3.6 | 4.8 | 1 | ||||||
| Finland | < 17 | 1986-1992 | 1.0a | R | 1998 | Pebody et al[88] | A | |||
| Finland | < 18 | 1987-2003 | 6.5 | 2.1 | 4.1 | P | 2016 | Lehtinen et al[89] | A | |
| Finland | < 19 | 1987-2014 | 7-23a | 6-8a | 10-15a | R | 2017 | Virta et al[66] | A | |
| < 16 | 2011-2014 | 13 | 8 | |||||||
| France | < 17 | 1988-2011 | 3.1-6.3a | P | 2016 | Bequet et al[126] | A | |||
| France/Northern | < 17 | 1998-1999 | 3.1a | 2.3a | 0.8a | 0.12a | P | 2005 | Auvin et al[43] | B |
| France/Northern | < 17 | 1988-2011 | 4.4a | 3.2a | 1.1a | 0.1a | P | 2017 | Ghione et al[131] | B |
| France/Brittany | < 17 | 1994-1997 | 2.5 | 1.6 | 0.57 | P | 2000 | Tourtelier et al[131] | B | |
| France/Northern | < 19 | 1988-2007 | 3.4-5.9a | P | 2011 | Chouraki et al[32] | B | |||
| France/Northern | < 17 | 1988-2002 | 2.6 | P | 2008 | Vernier-Massouille et al[127] | B | |||
| France/Northern | < 19 | 1988-2008 | 6.5-12.9a | P | 2013 | Gower-Rousseau et al[125] | B | |||
| France/Corsica | < 19 | 2002-2003 | 12.3 | 6.2 | P | 2007 | Abakar-Mahamat et al[128] | B | ||
| France/Nord-Pas-de-Calais | < 17 | 1988-1989 | 3.1 | 2.1 | 0.5 | P | 1991 | Gottrand et al[130] | B | |
| The Farose Islands | < 19 | 1960-2014 | 9.02a | 2.52a | 52a | 1.52a | P,R | 2016 | Hammer et al[19] | A |
| Iceland | < 16 | 1950-2010 | P,R | 2013 | Agnarsson et al[29] | A | ||||
| 1951-1960 | 1.2 | 0.2 | 1.1 | |||||||
| 1961-1970 | 0.9 | 0.1 | 0.7 | |||||||
| 1971-1980 | 0.9 | 0.1 | 0.7 | |||||||
| 1981-1990 | 2.5a | 1.2a | 1.2a | 0.1a | ||||||
| 1991-2000/2001-2010 | 5.6/5.0 | 2.5/2.3 | 2.5/2.3 | 0.3/0.3 | ||||||
| Iceland | < 19 | 1990-1994 | 4.5 | 5 | P | 2000 | Bjornsson et al[112] | A | ||
| Ireland | < 16 | 2000-2010 | 2.5-5.6a | 2.3a | 1.1a | R | 2012 | Hope et al[120] | A | |
| Ireland | < 10 | 2000-2014 | 0.8-3.3a | P,R | 2017 | Coughlan et al[121] | A | |||
| The Netherlands | < 18 | 1999-2001 | 5.2 | 2.1 | 1.6 | 3.6 | P | 2004 | van der Zaag-Loonen et al[124] | A |
| Germany/Southern | < 15 | 2004-2006 | 4.0a | 2.4a | 1.1a | P | 2008 | Ott et al[123] | B | |
| Norway/Southeastern | < 16 | 1990-1994 | 4.2 | 2 | 2.1 | P | 2002 | Bentsen et al[85] | B | |
| Norway/Southeastern | < 14 | 1990-1993 | 1.3 | P | 1996 | Moum et al[95] | B | |||
| Norway/Southeastern | < 16 | 1990-1993 | 4.7 | 2.7 | 2 | P | 2004 | Stordal et al[93] | B | |
| Norway/Southeastern | < 18 | 2005-2007 | 10.9b | 6.8b | 3.6b | 0.6 | P | 2009 | Perminow et al[92] | B |
| Norway/Southeastern | < 16 | 1993-2004 | 5.6-5.7a | 2.0-3.6a | 3.7-2.1a | P,R | 2006 | Perminow et al[60] | B | |
| Norway/Western | < 16 | 1984-1985 | 6.8 | 2.5 | 4.3 | 0 | P | 1989 | Olafsdottir et al[97] | B |
| Norway/Western | < 14 | 1984-1985 | 2.5 | P | 1988 | Haug et al[163] | B | |||
| Norway/Souteastern | < 14 | 1990 | 3 | 1.2 | 0.6 | P | 1995 | Moum et al[94] | B | |
| Norway/Southeastern | < 14 | 1990-1993 | 0.94 | P | 1996 | Moum et al[96] | B | |||
| Sweden/Northern Stockholm | < 17 | 1990-1998 | 6.9a | 3.8a | 2.1a | 1.1a | R | 1999 | Askling et al[99] | B |
| Sweden/Northern Stockholm | < 15 | 1990-2001 | 7.4a | 4.9a | 2.2a | 0.2a | P | 2003 | Hildebrand et al[59] | B |
| Sweden/Soutwestern | < 16 | 1983-1987 | 5.3 | 2.7 | 2.6 | 0.7 | P | 1994 | Hildebrand et al[164] | B |
| Sweden/Stockholm | < 15 | 2002-2007 | 12.8a,b | 9.2a,b | 2.8a,b | P | 2013 | Malmborg et al[38] | B | |
| Sweden | < 15 | 1984-1995 | 4.6-7.0a | 1.2-1.3a | 1.4-3.2a | 1.1a | P | 2000 | Lindberg et al[68] | A |
| Sweden | < 14 | 1963-1967 | 1.4 - 0.7a | R | 1991 | Lindberg et al[98] | A | |||
| 1983-1987 | 0.7 | |||||||||
| Sweden/the Uppsala region | < 17 | 2005-2009 | 18.9 | 8.9 | P | 2013 | Sjoberg et al[100] | B | ||
| Sweden/the Uppsala region | < 17 | 2005-2009 | 10 | P | 2014 | Sjoberg et al[101] | B | |||
| Sweden/the Uppsala region | < 19 | 1965-1983 | R | 1991 | Ekbom et al[56] | B | ||||
| 1965-1970 | 5.9-5.0a | 5.2-4.8a | ||||||||
| 1975-1983 | 4.8-3.2 | 3.9-2.7 | ||||||||
| Sweden/Orebro | < 19 | 1963-1987 | 6 | R | 1992 | Tysk et al[165] | B | |||
| Sweden/Stockholm County | < 14 | 1955-1983 | 4.1 | R | 1997 | Lapidus et al[35] | B | |||
| Sweden | < 15 | 1984-1985 | 4.8 | 1.7 | 1.7 | P | 1991 | Hildebrand et al[166] | A | |
| UK/ Scotland | < 16 | 2003-2008 | 7.8a | 4.8a | 2.1a | 1.0a | P,R | 2012 | Henderson et al[2] | A |
| 1990-1995 | 4.5 | 2.9 | 1.6 | |||||||
| UK/Wesex Southern England | < 16 | 2002-2012/2013-2017 | 2014, 2018 | Ashton et al Ashton et al[116,117] | B | |||||
| 2002-2006 | 6.4a,b | 3.8a,b | 2.0a,b | P | ||||||
| 2008-2012 | 9.4 | 5.9 | 2.6 | |||||||
| UK/Scotland | < 16 | 1981-1995 | 3.4 | 2.3a | 1.2a | R | 2004 | Armitage et al[114] | A | |
| UK/Wales | < 16 | 1996-2003 | 5.4 | 3.6 | 1.5 | P | 2006 | Ahmed et al[30] | B | |
| UK/Wales | < 16 | 1995-1997 | 2.6 | 1.4 | 0.8 | 0.5 | P | 2000 | Hassan et al[118] | B |
| UK/Wales | < 16 | 1981-1995 | 2.5a | 1.3a | R | 2001 | Armitage et al[53] | B | ||
| UK/Northeastern Scotland | < 16 | 1980-1999 | ||||||||
| 1980-1989 | 2.2 | 0.7 | R | 2002 | Watson et al[115] | B | ||||
| 1990-1999 | 4.4 | 1.5 | ||||||||
| UK/Scotland | < 16 | 1968-1983 | 0.7-2.3a | 1.9-1.6a | R | 1989 | Barton et al[61] | A | ||
| UK/Cardiff | < 16 | 1996-2005 | 2.7 | R | 2007 | Gunesh et al[119] | B | |||
| UK/Leicestershire | < 10 | 1972-1989 | 0.3a,d | R | 1992 | Probert et al[27] | B | |||
| UK/Wales South Glamorgan | < 16 | 1983-1993 | 2.2a | 0.7a | R | 1996 | Cosgrove et al[57] | B | ||
| 1983-1988 | 1.3 | 0.7 | ||||||||
| 1989-1993 | 3.1 | 0.7 | ||||||||
| UK/British Isles and Ireland | < 16 | 1998-1999 | 5.3a,d | 3.1a,d | 1.4a,d | 0.6d | P | 2001 | Sawczenko et al[16] | A |
| UK/Scotland | < 19 | 1990-1992 | 2.9a | R | 1999 | Armitage et al[113] | A | |||
| UK/East London | < 20 | 1997-2001 | 2.3-7.3a,d | 2.4-8.1a,d | R | 2004 | Tsironi et al[17] | |||
| Northern America | ||||||||||
| Canada/Manitoba | < 17 | 1978-2007 | 6.9a | 1.2-4.7a | 0.5-1.6a | P | 2014 | El-Matary et[79] | B | |
| Canada/Metropolitan Toronto | < 17 | 1991-1996 | 3.7 | 2.7 | R | 2004 | Griffiths[82] | B | ||
| Canada/ 5 Provinces | < 16 | 1999-2010 | 9.7a | 6.5a | 2.4a | R | 2017 | Benchimol et al[31] | A | |
| Alberta | 9.7 | 5.9 | 2.7 | |||||||
| Manitoba | 7.2 | 4.2 | 2.8 | |||||||
| Nova Scotia | 15.2 | 9.3 | 4.2 | |||||||
| Ontario | 9.3 | 5.5 | 3.2 | |||||||
| Quebec | 10.3 | 8.8 | 1 | |||||||
| Canada/Suothwestern Ontario | < 17 | 1997-2006 | 13.3a | 4.9a | 8.1a | 0.3a | R | 2009 | Grieci et al[81] | B |
| 1997-2001 | 14.3 | 3.5 | 10.6 | 0.2 | ||||||
| 2002-2006 | 12.4 | 6 | 6 | 0.4 | ||||||
| Canada/Ontario | < 18 | 1994-2005 | P | 2009 | Benchimol et al[54] | B | ||||
| 1994 | 9.5a | 6.2a | 4.4a | |||||||
| 2005 | 11.4 | 7 | 4.8 | |||||||
| Canada/Ontario | < 19 | 1999-2008 | P | 2014 | Benchimol et al[84] | B | ||||
| 0-9 | 2.9a | 1.3a | 1.3a | 0.3 | ||||||
| 10-19 | 21.5 | 12.8 | 7.6 | 1.1 | ||||||
| Canada/British Columbia | < 16 | 1985-2005 | 5.2d | 3.7d | 1.0d | 0.5d | R | 2007 | Pinsk et al[18] | B |
| 15.2 | 6.4 | 6.7 | 2.1 | |||||||
| Canada/Ontario | < 18 | 1994-2009 | 9.4-13.2a | 5.2-7.9a | 3.9-4.1a | R | 2014 | Benchimol et al[84] | B | |
| Canada/Eastern | < 20 | 1996-2009 | 14-11a2c | 4-6a2c | 0-1.5a2c | P | 2014 | Leddin et al[83] | B | |
| Canada/Quebec | < 19 | 2001-2008 | R | 2014 | Bitton et al[78] | B | ||||
| 0-9 | 2.0a | 1.0a | ||||||||
| 10-19 | 20 | 4 | ||||||||
| Canada/ 5 Provinces | < 19 | 1998-2000 | P | 2006 | Bernstein et al[39] | A | ||||
| Alberta | 9.4a | 4.1a | ||||||||
| British Columbia | 5.4 | 3.2 | ||||||||
| Manitoba | 6.9 | 4.5 | ||||||||
| Nova Scotia | 12 | 5.7 | ||||||||
| Saskatchewan | 7.9 | 4.2 | ||||||||
| Canada/Qubec | < 19 | 1993-2002 | 13.9 | P | 2009 | Lowe et al[80] | B | |||
| U.S.A./Northern Carolina | < 17 | 1996-2006 | 2.2-4.3a,d | 1.8-4.9a,d | 0.5d | R | 2010 | Abramson et al[20] | B | |
| U.S.A/Wisconsin | < 18 | 2000-2001 | 7.05d | 4.6d | 2.1d | P | 2003 | Kugathasan et al[21] | B | |
| U.S.A/Wisconsin | < 18 | 2000-2007 | 9.5a,d | 6.6a,d | 2.4a,d | 0.5a,d | P,R | 2013 | Adamiak et al[22] | B |
| U.S.A/Georgia | < 19 | 1986-1995 | 8.8a,d | 5.3a,d | R | 1998 | Ogunbi et al[23] | B | ||
| U.S.A/Texas | < 17 | 1991-2002 | R | 2010 | Malaty et al[52] | B | ||||
| 1991-1996 | 1.1a | 0.7a | 0.3a | 0.1a | ||||||
| 1997-2002 | 2.4 | 1.3 | 0.5 | 0.7 | ||||||
| U.S.A/Olmstedt Minesota | < 19 | 1940-2000 | 3.4a | 2.4a | R | 2007 | Loftus et al[58] | B | ||
| 1990-2000 | 4.8 | 3.2 | ||||||||
| U.S.A/Olmstedt Minesota | < 14 | 1943-1982 | 0.75a | R | 1988 | Gollop et al[167] | B | |||
| U.S.A/Olmstedt Minesota | < 19 | 1940-1993 | 2.5a | R | 1998 | Loftus et al[36] | B | |||
| U.S.A/Northern California | < 18 | 1996-2002 | 3 | 2.9 | R | 2008 | Herrinton et al[76] | B | ||
| U.S.A/Olmstedt Minesota | < 19 | 1940-1993 | 1.8 | R | 2000 | Loftus et al[168] | B | |||
| U.S.A/Rhode Island | < 19 | 2008-2010 | P | 2016 | Shapiro et al[77] | B | ||||
| 0-9 | 4.5a | 1.1a | 0.0a | |||||||
| 10-19 | 20.6 | 8.6 | 0.7 | |||||||
| Latin America | ||||||||||
| Argentina/Provinces | < 18 | 2012-2013 | ||||||||
| Argentina | 0.4 | P | 2017 | Vincentin et al[87] | A | |||||
| Buenos Aires | 0.3 | |||||||||
| CABA | 2.4 | |||||||||
| Chaco | 0.3 | |||||||||
| Cordoba | 0.3 | |||||||||
| Corrientes | 0.6 | |||||||||
| Entre Rios | 0.8 | |||||||||
| Mendoza | 0.2 | |||||||||
| Misiones | 0.9 | |||||||||
| San Juan | 1 | |||||||||
| Tucana | 0.2 | |||||||||
| Africa | ||||||||||
| Libya/Eastern/Benghazi | < 15 | 1997-2006 | R | 2009 | Ahmaida et al[146] | B | ||||
| 1997 | 0.0a | |||||||||
| 2000 | 0.2 | |||||||||
| 2006 | 0.9 | |||||||||
| Asia /Middle East | ||||||||||
| Saudi Arabia | < 14 | 2003-2012 | 0.5 | 0.3 | 0.2 | R | 2014 | El Mouzan et al[149] | A | |
| Saudi Arabia/Riyadh | < 18 | 1993-2002 | 0.5 | R | 2006 | El Mouzan et al[150] | B | |||
| Kuwait | < 15 | 1998-2008 | 2.2 | 1.5 | 0.6 | 0.03 | R | 2011 | Al-Quabandi[151] | A |
| Israel/Tel Aviv | < 19 | 1970-1980 | 1 | P | 1989 | Grossman et al[148] | B | |||
| Israel/Southeastern | < 19 | 1968-1992 | 3.6 | R | 1994 | Odes et al[147] | B | |||
| the Kingdom of Bahrain | < 19 | 1990-2015 | 3.7a | P,R | 2017 | Zayyani et al[152] | A | |||
| 1990-1995 | 1.8 | |||||||||
| 2010-2015 | 7.6 | |||||||||
| Taiwan | < 18 | 1979-2000 | R | 2004 | Tsai et al[154] | A | ||||
| 1979-1995 | 0.9a | 0.9a | ||||||||
| 1996-2000 | 2.6 | 1 | ||||||||
| Singapore | < 18 | 1996-2009 | R | 2013 | Chu et al[155] | A | ||||
| 2000 | 2.2a | |||||||||
| 2008 | 11.4 | |||||||||
| Taiwan | < 19 | 2000-2010 | R | 2015 | Chia Jung Kuo et al[169] | A | ||||
| 0-9 | 0.1a | 0.1a | ||||||||
| 10-19 | 0.2 | 0.2 | ||||||||
| Korea/South | < 19 | 2011-2014 | 2017 | Jung et al[157] | B | |||||
| 10-14 | 1.62 | 2.02 | P | |||||||
| 15-19 | 8.22 | 4.82 | ||||||||
| China/Shangahai | < 18 | 2000-2010 | 5.5a | 2.9a | 2.5a | R | 2013 | Wang et al[153] | B | |
| Korea/Seoul | < 19 | 1986-1997 | R | 2000 | Yang et al[156] | B | ||||
| 0-9 | 1986-1995/1995-1997 | 0-0.2 | ||||||||
| Australasia | 10-19 | 1986 -1995/1995-1997 | 0.2-0.9 | |||||||
| Australia/Randwick | < 16 | 1987-2011 | 33.1-4.3d | R | 2014 | Naidoo et al[24] | B | |||
| Australia/Victoria | < 16 | 1971-2001 | 0.1-2.0a | R | 2003 | Phavichitr et al[158] | B | |||
| Australia/Victoria | < 16 | 1983-1998 | 2.0a | R | 2009 | Ponsonby et al[37] | B | |||
| Australia/Victoria | < 16 | 1950-2009 | 0.4-1.6a | R | 2013 | Schildkraut et al[159] | B | |||
| New Zealand | < 16 | 2015 | 5.2 | 3.5 | 1 | 0.7 | P | 2017 | Lopez et al[160] | A |
| New Zealand/Canterburry | < 16 | 1996-2015 | 7.2a | P | 2018 | Lopez et al[161] | B | |||
| New Zealand | < 15 | 2002-2003 | 2.9d | 1.9d | 0.5d | P | 2008 | Yap et al[26] | A | |
| Sousthern Europe | ||||||||||
| Italy/Florence | < 15 | 1978-1992 | 1.9-3.4a | 3.8-9.6a | R | 1996 | Trallori et al[136] | B | ||
| Italy/Lazio | < 19 | 2008-2009 | R | 2014 | Domenicantonio et al[135] | B | ||||
| 0-9 | 2.5 | 2 | ||||||||
| 10-19 | 8.72 | 7.52 | ||||||||
| Italy | < 18 | 1996-2003 | 0.9-1.4a | P | 2008 | Castro et al[63] | A | |||
| Italy/Lombardia | < 14 | 1990-1993 | 1.2 | 1.2 | 1996 | Ranzi et al[134] | B | |||
| Spain | < 18 | 1996-2009 | 1.0-2.8a,b | 0.5-1.7a,b | 0.4-0.9a,b | R | 2013 | Martin de Carpi et al[42] | A | |
| Spain/the Vigo area | < 15 | 2010 | 18.3 | 10.3 | 8.7 | 1.2 | P | 2015 | Fernandéz et al[50] | B |
| Spain/Navarra | < 14 | 2001-2003 | 2.6 | 1.7 | 0.9 | 0.6 | P | 2008 | Letamendia et al[137] | B |
| Spain | < 18 | 1985-1995 | 0.2a1 | R | 2014 | Martin de Carpi et al[51] | A | |||
| Spain/Asturias | < 14 | 1993-2000 | 0.3 | 0.2 | 0.1 | P,R | 2004 | Fernandez Gonzalez[170] | B | |
| Spain/Northern | < 14 | 2000-2002 | 5.8 | 1.6 | P | 2004 | Rodrigo et al[44] | B | ||
| Spain/Aragon | < 14 | 1992-1995 | 0.4a | 0.3a | P | 1999 | Lopez Miguel et al[171] | B | ||
| Malta | < 15 | 1993-2005 | 1.3a | 7.9a | R | 2008 | Cachia et al[138] | A | ||
| Central and Eastern Europe | ||||||||||
| Croatia/Primorsko-Goranska | < 18 | 2000-2004 | 8.7 | 0.9 | P | 2006 | Sincic et al[47] | B | ||
| Czech Republic/Moravia | < 16 | 1998-2001 | 2.2a | 2.7a | 1.8a | 0.3a | R | 2004 | Kolek et al[140] | B |
| Czech Republic | < 15 | 1990-2001 | 0.3-.1.3a | P,R | 2006 | Pozler et al[139] | A | |||
| Czech Republic/West Bohemia | < 19 | 2000-2015 | 10.0a | 6.2a | 2.8a | 1.0a | P | 2017 | Schwarz et al[45] | B |
| < 15 | 7.3 | 4.6 | 2 | 0.7 | P | |||||
| Hungary | < 16 | 2007-2009 | 7.5 | 4.7 | 2.3 | 0.5 | P | 2013 | Muller et al[141] | A |
| Hungary/Veszprem | < 19 | 1977-2011 | 0.0-7.2a | 0.7-5.2a | P,R | 2014 | Lovasz et al[46] | B | ||
| Slovenia/Northestern | < 18 | 2002-2010 | 7.6 | 4.6 | 2.8 | R | 2014 | Urlep et al[49] | B | |
| 2002-2004 | 5.7 | 3.9 | 1.8 | |||||||
| 2008-2010 | 8.9 | 5 | 3.4 | |||||||
| Slovenia | < 18 | 2002-2010 | 7.6a,b | 4.5a,b | 2.9a,b | 0.2a,b | R | 2015 | Urlep et al[48] | A |
| 2002-2004 | 5.8 | 3.6 | 2.2 | |||||||
| 2005-2007 | 8.6 | 5.1 | 3.2 | |||||||
| 2008-2010 | 8.4 | 4.9 | 3.2 | |||||||
| Slovenia/Western | < 17 | 1994-2005 | 4.0a | 2.4a | 1.1a | 0.5a | R | 2009 | Orel et al[143] | B |
| 1994-1999 | 3 | 2 | 0.8 | 0.3 | ||||||
| 2000-2005 | 5.1 | 2.9 | 1.6 | 0.69 | ||||||
| Yugoslavia/Zagreb Croatia | < 14 | 1980-1989 | 1.76 | P,R | 1991 | Vucelic et al[145] | B | |||
| Yugoslavia/Zagreb Croatia | < 14 | 1980-1989 | 3.1 | P,R | 1991 | Vucelic et al[144] | B | |||
| Poland | < 18 | 2002-2004 | 2.7 | 0.6 | 1.3 | 0.8 | P | 2009 | Karolewska-Bochenek et al[67] | A |
| Hungary/Western | < 18 | 1977-2008 | 7.5a | 5.5a | P | 2011 | Lakatos et al[142] | B |
Population-based or national/subnational cohort studies reporting incidence were included in the analysis of the temporal evolution of IBD. The inclusion criteria were as follows: (1) Population-based or national cohort/health care administrative database studies (a study was considered population-based if all residents within specific areas were included, and the study population was representative of the catchment area; studies were considered at the national level as stated in the report or if the study was multicenter involving multiple regions in a country, sub-national level if only a particular region was evaluated, or city level); (2) published full-text manuscripts; (3) studies defining pediatric patients as patients younger than 19 years of age; (4) studies describing patients in the entire age range; and (5) studies performed within the geographical regions outlined by the UN. The exclusion criteria were as follows: (1) Studies with a sample size < 5 patients; (2) studies that did not report original data (e.g., review articles, meta-analyses, conference presentations, and guidelines); (3) studies that only demonstrated the incidence of adult-onset IBD (IBD onset after the age of 19 years); (4) studies reporting hospital surveys; (5) studies reporting only the number of pediatric cases (no incidence per population) and prevalence studies because our interest was disease incidence; (6) studies without defined study periods; and (7) duplicate studies reporting the same outcome in an identical cohort. To avoid selection bias due to cohort overlap, only the most recent study was included.
In total, 140 papers were retrieved, and a significant proportion of the studies was conducted in European countries (96 in Europe, 23 in North America, and the remaining 21 in Latin America, Africa, Asia/Middle East and Oceania); overall, the studies reported the IBD incidence in 38 countries. Moreover, wide variability in study design was observed. The upper age limit defining the pediatric population differed across the studies and ranged mostly between 15 and 19 years. The characteristics, distribution and detailed results of the 140 incidence studies, including references, are summarized in Table 1. As shown in Table 1, the ratio of CD to UC and IBD-U varied geographically. Of the included papers, 99 (71%) reported the IBD incidence in children, while the remaining 41 (29%) reported the rates of IBD in non-pediatric studies, but distinctions were made between adults and children in reporting the data. Of the 140 studies, the sample frame was prospective in 72 (51%) studies and retrospective in 57 (41%) studies. The data of the 11 (8%) studies were combined for this analysis.
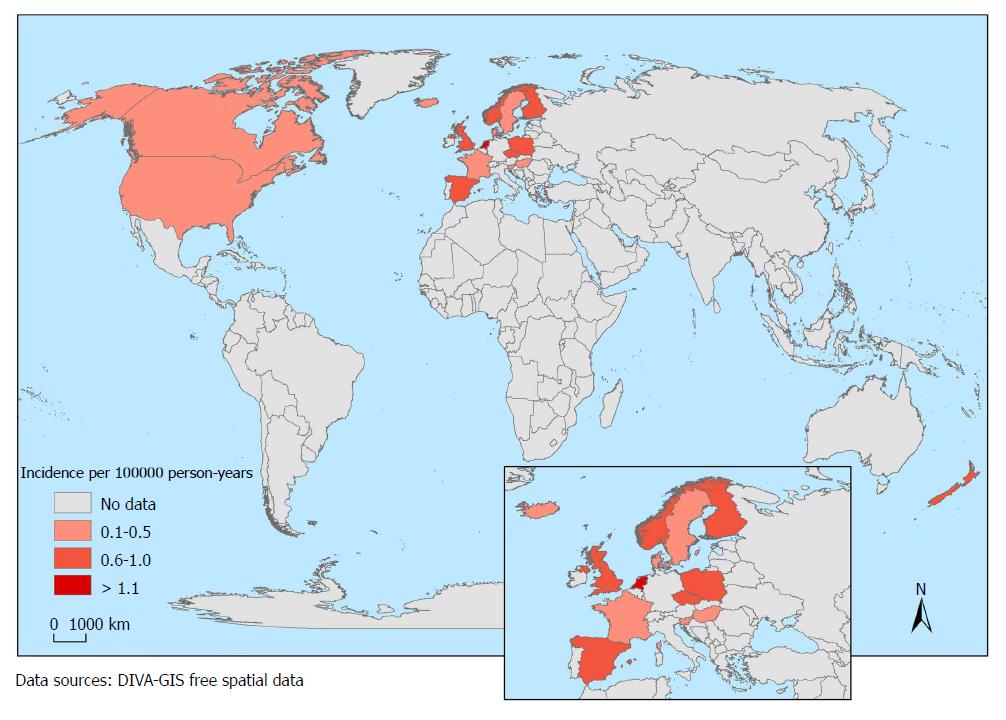
Broad variation was observed in the incidence rates, which ranged from 0.5 to 23/100000 for IBD, 0.1 to 13.9/100000 for CD, 0.3 to 15.0/100000 for UC, and 0.0 to 3.6/100000 for IBD-U. As shown in Table 2, the incidence of IBD greatly varied based on the geographical region. The regions with the highest IBD burden were Europe (0.2-23/100000) and North America (1.1-15.2/100000). The regions with the lowest reported IBD incidence were Oceania (2.9-7.2/100000), Asia (0.5-11.4/100000), Latin America (0.2-2.4/100000) and Africa (0.0-0.9/100000). The regions with the highest reported incidence of CD were North America (13.9/100000) and Europe (12.9/100000), while the highest rates of UC were 15.0/100000 in Europe and 10.6/100000 in North America. The highest incidence of IBD-U was 3.6/100000 in Europe and 2.1/100000 in North America. The current global status of the IBD incidence is shown in Figures 2-5.
| Regions | IBD incidence | UC incidence | CD incidence | IBD-U incidence | Time period |
| Europe | |||||
| North/West | 0.5-23 | 0.3-15 | 0.2-12.3 | 0.2-3.6 | 1951-2017 |
| East | 2.7-10.0 | 0.9-5.2 | 0.25-8.6 | 0.3-1 | 1997-2015 |
| South | 0.1-18.3 | 0.1-9.6 | 0.5-10.3 | 0.6-1.2 | 1978-2005 |
| North America | 1.1-15.2 | 0.5-10.6 | 0.7-13.9 | 0.2-2.1 | 1940-2010 |
| Latin America | 0.2-2.4 | NR | NR | NR | 2012-2013 |
| Africa | 0.0-0.9 | NR | NR | NR | 1997-2006 |
| Asia/Middle East | 0.5-11.4 | 0.2-3.9 | 0.3-3.7 | 0.1 | 1968-2012 |
| Australasia | 2.9-7.3 | 0.4-1 | 0.1-3.5 | 0.7 | 1971-2015 |
The incidence of IBD has been increasing worldwide. The time-trend analysis illustrated an increasing or stable incidence in North America, Europe and Oceania and an increasing incidence in newly industrialized countries in Asia, the Middle East and Africa. Of the 41 articles reporting the statistical significance (P < 0.05) of a time-trend analysis of the overall IBD incidence, 30 (73%) studies reported an increasing incidence, 11 (27 %) studies reported no significant changes, and no studies reported a decreasing incidence. Of the 71 studies that calculated the CD incidence, 48 (67%) studies reported a significant increase, 2 (3%) studies reported a significant decrease, and 21 (30%) studies observed no significant changes. Of the 63 UC studies, 29 (46%) studies reported a significant increase, 29 (46 %) studies reported no significant changes, and 5 (8%) studies reported a significant decreasing trend. Of the 19 studies calculating the IBD-U incidence, 2 (11%) studies reported a significant increase, and 17 (89%) studies reported no significant change.
Ten studies stratified according to migrant and race/ethnicity in South Asian (SA), Asian, African-American, Hispanic, Caucasian and Polynesian populations[16-26]. The two studies performed in Canada described the SA pediatric population, and one study compared non-immigrants to SA immigrants[18,25]. The three UK studies described the incidence in children with an Asian background[16,17,27]. The four studies performed in the US investigated Hispanic, Asian, African-American and Caucasian populations[20-22,28]. One study performed in New Zealand (NZ) described Polynesian children[26], one study performed in Australia compared Middle Eastern ethnicities[24], one study described the Faroese Islands[19], and the same authors described the risk of IBD in first-generation Faroese immigrants.
This rigorous and timely review has several important contributions. First, wide geographical and temporal variations were observed globally. Second, North America, Northern Europe and the UK have the highest incidence worldwide. The incidences of IBD in Southern and Eastern Europe and the Southern Hemisphere also appeared to be high, whereas the incidence was lower, but climbing, in Africa, South America, and Asia. Third, the incidence of IBD is substantially increasing in worldwide regions; however, data published during the previous two decades demonstrate the plateauing incidence of IBD in the Western world after a previously documented increase[22,29-38], but incidence remains high. Currently, the incidence might be sharply increasing in Southern and Eastern Europe and in Oceania. IBD is emerging in other parts of the world (i.e., certain parts of Asia/the Middle East, Africa) approaching the rates reported in westernized nations, but given the accelerating incidence found in many of these areas, the incidence is expected to increase. These trends clearly parallel those of the West that occurred along with the increasing development more than four decades ago indicating an emerging epidemic of IBD worldwide. This gap is considerably less pronounced in 2018, and the narrowing differential gap may have important implications for the worldwide IBD sequelae. The incidence has increased in recent decades up to 23 for IBD in Finland, 13.9 for CD in Canada, 15 for UC in Finland, and 3.6/100000 for IBD-U in the Netherlands. Few studies using north-south/west-east gradients have demonstrated particularly high rates in the north[39-42], except for northern France[43] and Spain[44]. Although our review did not specifically investigate disease incidence gradients, this phenomenon has been less prominent over the last three decades[45-51].
The global incidence rates of IBD have risen during the 20th century but data obtained during the previous two decades are conflicting. The incidence of IBD has increased mainly due to pediatric CD[42,51,52], whereas the incidence of UC has remained stable, although an inverse distribution of CD and UC has been reported[2,4,20,31,32,38,53-57]. Since 1950, 60% of CD and 20% of UC pediatric studies have shown a significant increase in incidence[4]. 75% of CD and 60% of UC studies show increasing incidence in adults[55]. In our systematic review, since 1985, 67% of studies investigating CD, 46% of studies investigating UC, and 11 % of studies investigating IBD-U have reported significant increasing in incidence worldwide. Thus, the incidence rates might be increasing in virtually all regions worldwide. However, the increase in UC was more modest. We intriguingly suggest that the rising incidence of UC may be attributable to the fact that in the emerging areas with a low incidence of IBD, UC has emerged first, followed by CD after a variable period[58], similar to trends in earlier studies from the West[4,56,59-63].
CD predominates over UC and IBD-U in areas with a high IBD incidence. Recent data indicate higher rates of pediatric CD than UC in Europe and North America, except in Scandinavia[64-66], Northern California[20], Southern[63] and Eastern Europe[67], where the incidence of UC exceeds that of CD. The reasons for these striking differences in the rates among the three subtypes of IBD remain uncertain[28,68,69]. IBD-U is more frequently found in children than adults (children 12.7% vs adults 6.0%, P < 0.0001)[70].
Considerable geographical diversity in IBD is observed. Moreover, several studies have reported different incidence rates within a country[31,65], highlighting the role of the environment[25]. Global data exploring the similarities and differences might call for future studies to extensively study genetic-environment interactions[71] providing an opportunity to further identify the contributing factors in locations where IBD is emerging rapidly[72]. Rates among ethnical/racial groups raise further questions[7] regarding why the children of immigrants from the developing world have increased rates of IBD. For example, the incidences of IBD among immigrants of SA origin in Canada[18,25], the UK[16,27], and in immigrants of Middle-Eastern descent in Australia have been reported[24]. In the state of Georgia, the highest incidence of CD in African American children was reported[23]. The key factor of migration influencing disease onset is likely exposure to a different environment than that in the country of origin[7,73]. Additionally, indigenous populations in developed countries have a much lower IBD incidence[74].
The incidence of IBD in North America is among the highest in the world and is increasing[4,20,22]. There are no national cohorts of pediatric IBD in the USA because the health system is not ideal for conducting population studies[75]. In general, data have been obtained from single regions and must be extrapolated to other geographical regions of the United States. The incidence of IBD in the entire state of Wisconsin (2000-2007) was 9.5/100000[22], which is similar to the incidence reported in Ontario, Canada[54], but the incidence of both CD and UC has remained stable. Regarding the members of the Kaiser Permanente Northern California health plan (KPNC) (1996-2006), the data somewhat differed likely due to the different mix of races/ethnicities than that observed in other parts of the United States. The incidence of UC demonstrated a significant 2.7-fold increase, and CD remained stable[20]. Hispanic and SA children had predominantly UC, suggesting the presence of possible etiological differences among ethnicities. Another study conducted in KPNC reported similar levels of IBD[76], while the incidence in children of African-American origin in Georgia was much higher (7.1/100000)[23]. In a Texas cohort, an increasing incidence of IBD among children with evidence of more CD than UC and IBD-U from 1991 to 2002 has been reported. Caucasians had a higher IBD incidence rate than African-Americans or Hispanics, and African Americans had predominantly CD[52]. Comparable values have been reported in Wisconsin. The mean incidence of CD was double that of UC. An equal IBD incidence was observed among all ethnic groups[21]. In the Olmsted County population, the incidence was 4.8 for CD and 3.2/100000 for UC, and remained stable between 1990 and 2000[36,58]. Data from Texas, Georgia, Wisconsin and from comparable studies in KPNC found rates similar to those in Olmsted County[20,76]. However, the ratio of CD/UC cases was greater in Olmsted County, Atlanta and Wisconsin, compared to the greater incidence of UC in KPNC. These results may be attributable to the ethnic demographics of the respective populations. In contrast, a much higher rat was found in the state of Rhode Island[77] compared with older cohorts from other parts of the U.S.[21,23,36,52].
The incidence of IBD in Canada is among the highest reported to date as documented in previous single-province studies[18,78-82] and large multi-province trials[31,39,83]. The incidence in Ontario has steadily increased from 9.5 (in 1994) to 11.4/100000 (in 2005). However, the incidence of UC was stable[54], and the incidence significantly increased from 9.4 to 13.2/100000 (1994-2009)[84]. Although the incidence of IBD in Ontario decreased from 14.3 to 12.4/100000 in 1997-2006, the incidence of CD nearly doubled[81], which is similar to another Canadian study[39]. Among the best studies in the region, 3 studies determined the incidence of IBD in five provinces of Canada[31,83,84]. The overall incidence was 9.7/100000, and CD was the predominant form of IBD similar to data from Nordic countries[85]. The incidence of IBD was the lowest in Manitoba and the highest in Nova Scotia. The incidence of IBD remained stable after stratifying into CD and UC during the first decade of the twenty-first century, except for an increase in incidence among the youngest group. By extrapolating the results to the entire country, approximately 650 children are diagnosed with IBD yearly, affecting up to 2695 children (< 16 years) in 2008[31]. Similarly, a study from Ontario observed the most rapid increase in children (< 10 years) between 1994 and 2008[86]. Recent data from Canada suggest that the rate of incidence of pediatric IBD is plateauing, indicating a reversal after a long period of ongoing increase[18,31,78,83].
In Argentina, IBD remains uncommon according to a study conducted from 2012 to 2013[87]. The total incidence was 0.4/100000 ranging from 0.2 to 2.4/100000. We could not find other data related to Argentinian children or children in other countries in Latin America for comparison.
The incidence rates reported in Scandinavia are among the highest rates published to date indicating higher rates of CD than UC and IBD-U but Finland is among the few countries that reportedly show a predominance of UC, whereas the incidence of CD has become relatively greater than that of UC in North America and the UK[20,64,66]. The incidence of IBD has almost doubled between 1987 and 2003[64] and tripled between 1987 and 2014, with a steeper increase in the incidence of UC compared to that of CD[66]. The incidence of UC increased from 4 to 9/100000 and that of CD increased from 2 to 5/100000[65,88-90] confirming a strongly increasing trend in Finland since the late 1980s. A Norwegian study covered the period of 1993-2004[60]. The incidence of IBD did not change and a trend towards an increase in CD and reduction in UC was recorded, which is similar to the finding of the IBSEN study of 1990-1994[91]. A subsequent study (2005-2007) performed in the same catchment area[92] showed that the incidence of IBD was 10.6/100000 indicating a marked increase in the incidence of CD although incidence of UC has been stabilized in Southeastern Norway compared with the rates over the previous 15 years[60,93-96]. Similarly, Canada had high a incidence of 9.7/100000[31]. The incidence of CD between 1984 and 1985 was 2.5/100000, whereas the incidence of UC was 4.3/100000 with a lower incidence of UC in Western Norway compared to that in Southeastern Norway[97], which is similar to the results reported in a subsequent study in Southeastern Norway (1990-1994)[85]. An increase in the incidence of CD was observed in Northern Stockholm (1990-2001), while the incidence of UC was stable, and a significant increase was observed in the overall incidence of IBD[59]. In a follow-up paper (2002-2007), the incidence of IBD had plateaued. The incidence rate was 9.2 for CD and 2.8/100000 for UC[38] and the incidence of UC significantly increased but not of CD. The incidence of IBD significantly increased between 2000 and 2007 compared to that reported in an earlier study conducted in the same region (1990-2001)[38]. These rates are relatively higher than those reported in other studies on pediatric IBD, although the rates are similar to those reported in studies conducted in Canada[54,81], Norway[92], and Finland[65]. Lindberg et al[68] suggested that the incidence of UC increased (from 1.4 to 3.2/100000), whereas that of CD and IBD-U remained stable (1984-1995), which is similar to the results of a study conducted from 1963-1987[98]. The incidence of IBD, CD, UC and IBD-U was 6.9, 3.8, 2.1, and 1.1/100000, respectively, in Northern Stockholm (1990-1998). After more than a decade of a stable incidence of IBD in Scandinavia[35], the incidence of CD significantly increased from 1990 to 1998, while the corresponding incidence of UC and IBD-U remained unchanged[99]. As a part of the Swedish ICURE study (2005-2009), the incidence of pediatric IBD in Uppsala County, just north of Stockholm, was among the highest reported in Europe[100,101]. In Denmark, the incidence of IBD has steadily increased from the 1960s until 2013[102-104], except for in one study[105]. An increase in IBD from the 1980s to 2013 was observed, but the incidence rates increased the most in patients (< 15 years) with CD[103]. The incidence rate of UC increased from 1962 to 1987 in the county of Copenhagen[106], and Fonager et al[107] discovered an increasing incidence of CD but a rather stable incidence of UC with a tendency towards decreasing from 1987 to 1992. Compared to earlier Danish investigations, the incidence of CD had increased nearly 15-fold, whereas the incidence of UC remained stable between 1962 and 2006[34,102,108]. Another study observed an insignificant increase in the incidence of IBD between 1998 and 2004[33], indicating that the previously observed increasing incidence might be levelling. A significant increase in the incidence of IBD was also reported in recent Danish nationwide comparisons from 1995 to 2013 [109-111]. The IBD incidence (< 19 years) has increased in isolated regions as the Faroe Islands (part of the Danish realm) (1960-2014[19] with the predominance of UC comparable to findings obtained in the Nordic countries[64,66,105]. The incidence among Icelandic children is closest to that observed in Denmark[34] and Sweden[59] but lower than that observed in Norway[54,92]. Between 1980 and 2010, a sharp increase in IBD incidence was observed, however, the incidence levelled from 2000 to 2010[29,112] similar to the findings in Denmark and Wisconsin[22,34] but lower compared to other Northern countries.
Sawczenko et al[16] discovered an incidence of 5.3/100000 in the British Isles; CD was twice as common as UC, accounting for approximately 700 new cases/year in the UK and the Republic of Ireland. A greater proportion of SA children had UC than non-immigrants[16]. In Scotland, incidence data spanning over 40 years showed a dramatic increase in IBD with a marginal decrease in the incidence of UC but an increasing incidence of CD from 1968 to 1983. In follow-up studies, the increase in CD continued between 1990 and 1992[61,113]. In contrast, the incidence of CD continues to increase, and the incidence of UC is also apparently increasing from 1981 to 1995[53]. Other Scottish studies showed an increase in the incidence of CD from 1981 to 1995 but no difference was observed in the incidence of UC[16,114,115]. By comparing the periods 1990-1995 to 2003-2008, significant increases were observed in the incidence of IBD, CD and UC[2]. Data obtained in Wessex, England reported an incidence of 9.37/100000 which significantly increased from 2002 to 2012[116]. The most recent figures (2013-2017) were reported and compared to previously published Wessex data, demonstrating the most contemporary incidence and trend over 16 years[117]. Similar findings have been observed among Welsh children[57] and appears to have plateaued between 1995 and 1997[118]. In 1995-2003, the overall incidence of IBD was 5.4/100000 and had reached a plateau[30,118], but data obtained during 1996-2005 show that the incidence of CD is continuing to slowly increase in Cardiff[119]. An increasing incidence of IBD in Irish children was observed between 1998 and 2014[16,120,121], which is consistent with the global trends[2,4]. Another study also confirms the continuous increase in the incidence of IBD, particularly UC[121].
In Austria, the overall incidence of CD and UC have increased from 1997 to 2007[122]. This finding is in contrast to Germany[123] and the Netherlands[124]. For example, a German study did not show any significant change in the IBD incidence[123]. Worldwide, strikingly, the high proportion of IBD-U was observed in the Netherlands[124] and the incidence of IBD cases is comparable with that reported in other European countries. In France, the incidence of CD significantly increased, while the incidence of UC remains unchanged from 1988 to 2011[32,43,125-128]. The most remarkable observation (the EPIMAD Registry) has been a striking increase from 1988 to 2008 in the incidence of CD (< 19 years)[125], and the rates also significantly rose from 1988 to 2011[32,126,127] Surprisingly, in Corsica, using the same registry (EPIMAD), the incidence of CD was close to that observed in other metropolitan French regions; however, the incidence of IBD for UC in Corsica is two-fold higher than that reported in other French regions[128]. In Brittany, the incidence of IBD in childhood was similar to data obtained in Northern France and Nord-Pas-de-Calais[129,130]. Between 1988 and 2011, a dramatic increase was observed in the incidence of both UC and CD in French adolescents[131]. Other than the Swiss IBD Cohort Study (SIBDCS)[132] and the Belgian registry for pediatric CD (BELCRO)[133], up-to-date data regarding the incidence and trends are lacking.
A registry in Italy (1996-2003) showed a significant increase in the incidence of IBD from 0.89 to 1.39/100000 in all 3 pathologies[63], which is comparable to the incidence in Lombardia (1990-1993)[134]. Of note, Italy had a low incidence of IBD earlier, with an initial increase in UC exceeding CD and IBD-U, followed by an increase in the CD incidence, while the UC incidence was stable from 1998-2003[63]. In contrast, the incidence of IBD in central Italy (1978-1992) was comparable to that in the Nordic countries[135,136]. The incidence of IBD was shown to have significantly increased in Spain[44,137]. The SPIRIT (1996-2009) and EXPERIENCE registries (1985-1995) contribute to the complete description of the changes in pediatric IBD in Spain. A three-fold collective increase in IBD (1996-2009) was observed, with another three-fold increase in CD and a two-fold increase in UC, while a lower proportion of IBD-U was described[42]. According to the two latter studies extending the trends to a full 25-year period, these registries showed a sixteen-fold increase in Spain[42,51]. Notably, the incidence of IBD (mainly CD) (18.3/100000) in the Vigo area was the highest compared to that in former Spanish pediatric cohorts[50]. In Malta, UC showed an almost significant increasing trend, but no significant trend in CD was observed[138].
A sharp increase in the incidence of IBD is particularly noticeable in the Czech Republic, Hungary, Slovenia and Croatia, but not in Poland. The results of the 3 studies conducted in the Czech Republic are comparable to the West. Pozler et al[139] showed a five-fold increase in the incidence of CD. Kolek et al[140] published results from Moravia (the eastern part of the Czech Republic) showing increasing incidence of CD and UC between 1999 and 2001. The Czech Republic has among the highest rates of IBD worldwide as recently observed by our group (2000-2015)[45] (10.0, 6.2, 2.8 and 1.0/100000 for IBD, CD, UC and IBD-U, respectively) and have been shown to be increasing in future projections[45]. In neighboring Poland, UC incidence was higher than that of CD with significant regional differences, but the incidence was markedly lower than that observed in the West. Of note the incidence of IBD-U was surprisingly high[67]. An increasing incidence of IBD has been reported in Hungary[46,141,142] comparable to the rate of Slovenia from 1994 to 2010[48,49,143]. One other publication[47] reported much higher rates in Croatia (2000-2004) compared to previous reports[144,145].
Expectedly, knowledge regarding the incidence of IBD in the entire African pediatric population is limited, but the incidence of IBD increased from 0.0/100000 in 1997 to 0.2/100000 in 2002 and 0.9/100000 in 2006 in Libya[146].
In Israel, the estimated incidence of CD and UC was 3.7 and 0.9/100000, respectively[147,148], which are comparable, but at the lower end, with those in the West[16,21]. 2 studies reported that the IBD incidence (0.5/100000) is lower than that in Western countries among children in Saudi Arabia from 1993 to 2012[149,150]. A higher incidence was reported in Kuwait and Bahrain (states neighboring Saudi Arabia). In Kuwait, the incidence was more than triple that reported in neighboring Saudi Arabia[151]. This finding provides an annual incidence of 2.16/100000 for IBD, whereas the incidence of CD is 1.53, UC is 0.6 and IBD-U is 0.03/100000. A remarkable finding reported in the Arabian Gulf region is the high incidence of CD in Bahrain[152], which is comparable to Western areas. The lower incidence of IBD in Asia compared with that in Western countries is not universal, and the incidence in China is higher than that in other regions in Asia. This incidence is considered low compared to that in North America and the Nordic countries but not that low compared to the incidence in Scotland[53,113,114] and France[43] and is higher than that in Italy[63]. In China, a multicenter audit of over a decade of experience with childhood IBD between 2000 and 2010 in Shanghai has shown a steadily increasing trend (< 14 years). The incidence of IBD in 2010 was 6.1/100000, which is 12-fold higher than the incidence in 2001[153]. Recent data on Taiwan also demonstrated a substantial increase in the incidence of IBD, which is mainly attributable to CD, while the incidence of UC did not change significantly[154]. Singapore also witnessed a remarkable increase from 2000-2008; the incidence rates were 5.2-fold greater than those assessed 9 years earlier (from 2.2 to 11.4/100000)[155], which is similar to a report from Scandinavia[66]. The incidence of IBD has been increasing in Korea recently[156,157].
Early studies conducted in Australia and NZ (collectively termed Australasia) mirror the incidence observed in the Northern Hemisphere[54,65]. Two Victorian studies from the same area of Australia clearly show increasing rates of both CD and UC. The incidence of CD increased 10-fold over 30 years until 2001[158]. Additionally, an eleven-fold increase was seen in UC with particular increases in the early 1990s, and the incidence has yet to plateau[159]. Using the 1983-1998 population data, the incidence was estimated to be 2.01/100000 for CD among Victorian children with a documented increase[37]. A study conducted in the Sydney area showed much higher rates of IBD in children of Middle-Eastern descent[24]. In contrast, low rates of IBD were observed in the following indigenous populations: Aborigines and Maori[74]. Yap et al[26] calculated the incidence of IBD, CD and UC to be 2.9, 1.9 and 0.5/100000, respectively in NZ, which is at the lower end compared with the incidence in Europe. Recently, Lopez et al[160] provided important data pertaining to the incidence of pediatric IBD in NZ. The incidence of IBD, CD, UC and IBD-U in NZ in 2015 were 5.2, 3.5, 1.0 and 0.7/100000, respectively. A 4-fold increase was observed in the incidence of childhood IBD in the Canterbury Province of NZ between 1996 and 2015. The annual incidence rate was 7.18/100000 with the preponderance of CD over UC (8.4:1)[160,161].
Limitations include heterogeneity in population characteristics among the different studies which were also conducted at different times. Most countries lack accurate estimates of the incidence of pediatric IBD. A direct inter-region comparison may be limited due to the use of different diagnostic criteria and geographic distribution[25]. Also, study quality, case ascertainment, different database capture systems, and methodological problems demonstrated heterogeneity, emphasizing the importance of nationwide registries for retrieval of specific health data[62] including a well-established referral system with uniform criteria[40]. This recommendation underlines the need for uniform diagnostic guidelines for the accuracy of comparison among populations[38,162]. Some studies observed crude incidence rates, while other studies reported age- and/or sex-adjusted rates. Some studies did not reflect the countries’ true incidence, because selected areas of the country were sampled instead of the entire country. We should target large databases at the national/international levels providing a more comprehensive analysis[41]. The other limitation rests with the retrospective design of a number of studies and a better categorisation of migrants. The differences in the various age brackets with great impact on incidence rate should be considered as studies involving a higher defined age limit had higher incidence figures[92]. Despite these limitations, these considerations are unlikely to have had major effects on the reporting the changes in the incidence of IBD across time and geography.
The incidence of pediatric IBD (mainly CD) has recently dramatically increased emerging as a globally important changing pediatric disease. In a rapidly changing world, the dramatically increasing incidence of IBD has been observed in the Western world, Oceania and Eastern and Southern Europe, despite a stabilization of incidence rates in the West. The incidence appears to be rising both in newly industrialized and developing countries, and among first-generation of immigrants. Regarding IBD, knowledge of its etiology is limited, and awareness of the patterns of its global incidence could offer new clues, but the complex interplay of genetic and environmental factors remains unclear. Investigations of IBD performed where it is rapidly emerging provide an opportunity to identify the contributing factors. Efforts at global co-ordination for more prospective, population-based studies in children should be encouraged.
The incidence of inflammatory bowel disease (IBD) is increasing globally. Multiple studies have reported the pediatric IBD incidence in individuals over the past few years. However, the global and regional IBD incidences in childhood and their trends over time are not well reported. The highest pediatric incidence is traditionally observed in industrialized countries in North America and Western Europe. The incidence of IBD is increasing in both developed and developing countries. The variations in the disease incidence may reflect differences in the distribution of various environmental triggers for the disease within specific areas. The changing incidence of pediatric IBD worldwide provides an opportunity to study disease etiology.
The incidence of IBD is increasing worldwide in both adults and children. Thus, epidemiological knowledge is essential for defining new etiological hypotheses and predictors of the development of IBD to better define how environmental factors might influence disease onset and to guide future studies. Furthermore, additional evidence has recently become available due to the publication of previous reviews, but many incidence rates have since changed. Consequently, currently, a window of opportunity exists for the completion of a new, rigorous pediatric systematic review.
The authors aimed to summarize up-to-date studies investigating the incidence of pediatric-onset IBD and track the changes over time based on a comprehensive search of credible published pediatric studies and current knowledge regarding pediatric IBD incidence.
A systematic review was performed using Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. We searched electronic databases (MEDLINE, EMBASE, and Cochrane Library). Studies investigating the incidence and trends of pediatric IBD over time were eligible for inclusion. Interactive maps and temporal trends were used to illustrate the incidences of and changes in IBD.
One hundred forty studies met the inclusion criteria, demonstrating a substantial increase in the incidence of pediatric IBD and great geographic variations. The incidence of IBD remains the highest in the northern populations of Europe and America and has remained stable or even decreased. Rising rates of pediatric IBD were observed in previously low-incidence areas and much of the developing world, and among children of immigrants. The incidence rates of Crohn’s disease (CD) and ulcerative colitis (UC) vary worldwide between 0.2/100000 and 13.9/100000 and between 0.1/100000 and 15/100000, respectively. In the time-trend analyses, 67% of CD and 46% of UC studies reported a significant increase.
This study is among the most comprehensive studies to summarize the global IBD incidence. The IBD incidence is increasing or stable over time in both developed and developing regions of the world, indicating an emerging epidemic of the disease outside the Western world, whereas those in Northern Europe may have reached a plateau. The reasons contributing to these continued increases remain unclear. Whether genetic or environmental factors are the cause of these differences remains to be determined. Investigations of IBD performed in locations where it is emerging rapidly provide an opportunity to further identify the causative factors within specific populations. Whether the incidence of IBD in children will continue to increase or remain static is unclear.
Our data may serve as an essential resource for future studies and can be used to prioritize public health efforts in areas with the highest incidence. Knowing the increasing incidence of IBD and different geographic distribution may provide new insight into the etiology of pediatric IBD and direct future investigative studies. We must find ways to match genetic and environmental factors to pediatric IBD. An understanding of the early evolution of IBD is important and must be further investigated to unravel its etiology. This understanding is particularly important for preventing or curing the disease during the early stages. Attempts to perform studies with global coordination and additional prospective, population-based studies should be encouraged.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Czech Republic
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Prato A, Kirsaclioglu CT S- Editor: Wang JL L- Editor: A E- Editor: Huang Y
| 1. | Abraham BP, Mehta S, El-Serag HB. Natural history of pediatric-onset inflammatory bowel disease: a systematic review. J Clin Gastroenterol. 2012;46:581-589. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 132] [Cited by in F6Publishing: 129] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 2. | Henderson P, Rogers P, Gillett P, Wilson D. The epidemiology and natural history of paediatric inflammatory bowel disease in a UK region: a prospective 14-year study. Arch Dis Child. 2012;97:A53.2-A54. [DOI] [Cited in This Article: ] |
| 3. | Ng SC, Shi HY, Hamidi N, Underwood FE, Tang W, Benchimol EI, Panaccione R, Ghosh S, Wu JCY, Chan FKL. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet. 2018;390:2769-2778. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2677] [Cited by in F6Publishing: 3172] [Article Influence: 453.1] [Reference Citation Analysis (0)] |
| 4. | Benchimol EI, Fortinsky KJ, Gozdyra P, Van den Heuvel M, Van Limbergen J, Griffiths AM. Epidemiology of pediatric inflammatory bowel disease: a systematic review of international trends. Inflamm Bowel Dis. 2011;17:423-439. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 640] [Cited by in F6Publishing: 662] [Article Influence: 50.9] [Reference Citation Analysis (0)] |
| 5. | da Silva BC, Lyra AC, Rocha R, Santana GO. Epidemiology, demographic characteristics and prognostic predictors of ulcerative colitis. World J Gastroenterol. 2014;20:9458-9467. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 167] [Cited by in F6Publishing: 162] [Article Influence: 16.2] [Reference Citation Analysis (1)] |
| 6. | Van Limbergen J, Russell RK, Drummond HE, Aldhous MC, Round NK, Nimmo ER, Smith L, Gillett PM, McGrogan P, Weaver LT. Definition of phenotypic characteristics of childhood-onset inflammatory bowel disease. Gastroenterology. 2008;135:1114-1122. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 632] [Cited by in F6Publishing: 630] [Article Influence: 39.4] [Reference Citation Analysis (0)] |
| 7. | Misra R, Faiz O, Munkholm P, Burisch J, Arebi N. Epidemiology of inflammatory bowel disease in racial and ethnic migrant groups. World J Gastroenterol. 2018;24:424-437. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 34] [Cited by in F6Publishing: 27] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 8. | Girardelli M, Basaldella F, Paolera SD, Vuch J, Tommasini A, Martelossi S, Crovella S, Bianco AM. Genetic profile of patients with early onset inflammatory bowel disease. Gene. 2018;645:18-29. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 9. | Jakobsen C, Cleynen I, Andersen PS, Vermeire S, Munkholm P, Paerregaard A, Wewer V. Genetic susceptibility and genotype-phenotype association in 588 Danish children with inflammatory bowel disease. J Crohns Colitis. 2014;8:678-685. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 10. | Fang YH, Luo YY, Yu JD, Lou JG, Chen J. Phenotypic and genotypic characterization of inflammatory bowel disease in children under six years of age in China. World J Gastroenterol. 2018;24:1035-1045. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 29] [Cited by in F6Publishing: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 11. | von Allmen D. Pediatric Crohn‘s Disease. Clin Colon Rectal Surg. 2018;31:80-88. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 12. | Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6:e1000100. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9970] [Cited by in F6Publishing: 10260] [Article Influence: 684.0] [Reference Citation Analysis (0)] |
| 13. | United Nations. Composition of macro Geographical (Continental) Regions, Geographical Sub-Regions, and Selected Economic and Other Groupings. Available from: http://unstats.un.org/unsd/methods/m49/m49regin.htm. [Cited in This Article: ] |
| 14. | Lennard-Jones JE. Classification of inflammatory bowel disease. Scand J Gastroenterol Suppl. 1989;170:2-6; discussion 16-19. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1396] [Cited by in F6Publishing: 1420] [Article Influence: 40.6] [Reference Citation Analysis (0)] |
| 15. | Sanderson S, Tatt ID, Higgins JP. Tools for assessing quality and susceptibility to bias in observational studies in epidemiology: a systematic review and annotated bibliography. Int J Epidemiol. 2007;36:666-676. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1078] [Cited by in F6Publishing: 1134] [Article Influence: 66.7] [Reference Citation Analysis (0)] |
| 16. | Sawczenko A, Sandhu BK, Logan RF, Jenkins H, Taylor CJ, Mian S, Lynn R. Prospective survey of childhood inflammatory bowel disease in the British Isles. Lancet. 2001;357:1093-1094. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 260] [Cited by in F6Publishing: 304] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 17. | Tsironi E, Feakins RM, Probert CS, Rampton DS, Phil D. Incidence of inflammatory bowel disease is rising and abdominal tuberculosis is falling in Bangladeshis in East London, United Kingdom. Am J Gastroenterol. 2004;99:1749-1755. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 82] [Cited by in F6Publishing: 85] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 18. | Pinsk V, Lemberg DA, Grewal K, Barker CC, Schreiber RA, Jacobson K. Inflammatory bowel disease in the South Asian pediatric population of British Columbia. Am J Gastroenterol. 2007;102:1077-1083. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 117] [Cited by in F6Publishing: 112] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 19. | Hammer T, Nielsen KR, Munkholm P, Burisch J, Lynge E. The Faroese IBD Study: Incidence of Inflammatory Bowel Diseases Across 54 Years of Population-based Data. J Crohns Colitis. 2016;10:934-942. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 43] [Cited by in F6Publishing: 46] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 20. | Abramson O, Durant M, Mow W, Finley A, Kodali P, Wong A, Tavares V, McCroskey E, Liu L, Lewis JD. Incidence, prevalence, and time trends of pediatric inflammatory bowel disease in Northern California, 1996 to 2006. J Pediatr. 2010;157:233-239.e1. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 89] [Cited by in F6Publishing: 90] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 21. | Kugathasan S, Judd RH, Hoffmann RG, Heikenen J, Telega G, Khan F, Weisdorf-Schindele S, San Pablo W Jr, Perrault J, Park R, Yaffe M, Brown C, Rivera-Bennett MT, Halabi I, Martinez A, Blank E, Werlin SL, Rudolph CD, Binion DG; Wisconsin Pediatric Inflammatory Bowel Disease Alliance. Epidemiologic and clinical characteristics of children with newly diagnosed inflammatory bowel disease in Wisconsin: a statewide population-based study. J Pediatr. 2003;143:525-531. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 441] [Cited by in F6Publishing: 478] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 22. | Adamiak T, Walkiewicz-Jedrzejczak D, Fish D, Brown C, Tung J, Khan K, Faubion W Jr, Park R, Heikenen J, Yaffee M, Rivera-Bennett MT, Wiedkamp M, Stephens M, Noel R, Nugent M, Nebel J, Simpson P, Kappelman MD, Kugathasan S. Incidence, clinical characteristics, and natural history of pediatric IBD in Wisconsin: a population-based epidemiological study. Inflamm Bowel Dis. 2013;19:1218-1223. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 77] [Cited by in F6Publishing: 64] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 23. | Ogunbi SO, Ransom JA, Sullivan K, Schoen BT, Gold BD. Inflammatory bowel disease in African-American children living in Georgia. J Pediatr. 1998;133:103-107. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 63] [Cited by in F6Publishing: 66] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 24. | Naidoo CM, Leach ST, Day AS, Lemberg DA. Inflammatory bowel disease in children of middle eastern descent. Int J Pediatr. 2014;2014:906128. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5] [Cited by in F6Publishing: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 25. | Benchimol EI, Mack DR, Guttmann A, Nguyen GC, To T, Mojaverian N, Quach P, Manuel DG. Inflammatory bowel disease in immigrants to Canada and their children: a population-based cohort study. Am J Gastroenterol. 2015;110:553-563. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 158] [Cited by in F6Publishing: 151] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 26. | Yap J, Wesley A, Mouat S, Chin S. Paediatric inflammatory bowel disease in New Zealand. N Z Med J. 2008;121:19-34. [PubMed] [Cited in This Article: ] |
| 27. | Probert CS, Jayanthi V, Pinder D, Wicks AC, Mayberry JF. Epidemiological study of ulcerative proctocolitis in Indian migrants and the indigenous population of Leicestershire. Gut. 1992;33:687-693. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 185] [Cited by in F6Publishing: 196] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 28. | Malaty HM, Mehta S, Abraham B, Garnett EA, Ferry GD. The natural course of inflammatory bowel disease-indeterminate from childhood to adulthood: within a 25 year period. Clin Exp Gastroenterol. 2013;6:115-121. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 25] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 29. | Agnarsson U, Björnsson S, Jóhansson JH, Sigurdsson L. Inflammatory bowel disease in Icelandic children 1951-2010. Population-based study involving one nation over six decades. Scand J Gastroenterol. 2013;48:1399-1404. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 30. | Ahmed M, Davies IH, Hood K, Jenkins HR. Incidence of paediatric inflammatory bowel disease in South Wales. Arch Dis Child. 2006;91:344-345. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 25] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 31. | Benchimol EI, Bernstein CN, Bitton A, Carroll MW, Singh H, Otley AR, Vutcovici M, El-Matary W, Nguyen GC, Griffiths AM. Trends in Epidemiology of Pediatric Inflammatory Bowel Disease in Canada: Distributed Network Analysis of Multiple Population-Based Provincial Health Administrative Databases. Am J Gastroenterol. 2017;112:1120-1134. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 190] [Cited by in F6Publishing: 210] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 32. | Chouraki V, Savoye G, Dauchet L, Vernier-Massouille G, Dupas JL, Merle V, Laberenne JE, Salomez JL, Lerebours E, Turck D. The changing pattern of Crohn‘s disease incidence in northern France: a continuing increase in the 10- to 19-year-old age bracket (1988-2007). Aliment Pharmacol Ther. 2011;33:1133-1142. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 121] [Cited by in F6Publishing: 88] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 33. | Jakobsen C, Wewer V, Urne F, Andersen J, Faerk J, Kramer I, Stagegaard B, Pilgaard B, Weile B, Paerregaard A. Incidence of ulcerative colitis and Crohn‘s disease in Danish children: Still rising or levelling out? J Crohns Colitis. 2008;2:152-157. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 26] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 34. | Jakobsen C, Paerregaard A, Munkholm P, Wewer V. Paediatric inflammatory bowel disease during a 44-year period in Copenhagen County: occurrence, course and prognosis--a population-based study from the Danish Crohn Colitis Database. Eur J Gastroenterol Hepatol. 2009;21:1291-1301. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 31] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 35. | Lapidus A, Bernell O, Hellers G, Persson PG, Löfberg R. Incidence of Crohn‘s disease in Stockholm County 1955-1989. Gut. 1997;41:480-486. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 103] [Cited by in F6Publishing: 114] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 36. | Loftus EV Jr, Silverstein MD, Sandborn WJ, Tremaine WJ, Harmsen WS, Zinsmeister AR. Crohn‘s disease in Olmsted County, Minnesota, 1940-1993: incidence, prevalence, and survival. Gastroenterology. 1998;114:1161-1168. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 458] [Cited by in F6Publishing: 427] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 37. | Ponsonby AL, Catto-Smith AG, Pezic A, Dupuis S, Halliday J, Cameron D, Morley R, Carlin J, Dwyer T. Association between early-life factors and risk of child-onset Crohn‘s disease among Victorian children born 1983-1998: a birth cohort study. Inflamm Bowel Dis. 2009;15:858-866. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 31] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 38. | Malmborg P, Grahnquist L, Lindholm J, Montgomery S, Hildebrand H. Increasing incidence of paediatric inflammatory bowel disease in northern Stockholm County, 2002-2007. J Pediatr Gastroenterol Nutr. 2013;57:29-34. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 68] [Cited by in F6Publishing: 64] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 39. | Bernstein CN, Wajda A, Svenson LW, MacKenzie A, Koehoorn M, Jackson M, Fedorak R, Israel D, Blanchard JF. The epidemiology of inflammatory bowel disease in Canada: a population-based study. Am J Gastroenterol. 2006;101:1559-1568. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 402] [Cited by in F6Publishing: 422] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 40. | Shivananda S, Lennard-Jones J, Logan R, Fear N, Price A, Carpenter L, van Blankenstein M. Incidence of inflammatory bowel disease across Europe: is there a difference between north and south? Results of the European Collaborative Study on Inflammatory Bowel Disease (EC-IBD). Gut. 1996;39:690-697. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 684] [Cited by in F6Publishing: 643] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 41. | Burisch J. Crohn‘s disease and ulcerative colitis. Occurrence, course and prognosis during the first year of disease in a European population-based inception cohort. Dan Med J. 2014;61:B4778. [PubMed] [Cited in This Article: ] |
| 42. | Martín-de-Carpi J, Rodríguez A, Ramos E, Jiménez S, Martínez-Gómez MJ, Medina E; SPIRIT-IBD Working Group of Sociedad Española de Gastroenterología, Hepatología y Nutricion Pediátrica. Increasing incidence of pediatric inflammatory bowel disease in Spain (1996-2009): the SPIRIT Registry. Inflamm Bowel Dis. 2013;19:73-80. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 84] [Cited by in F6Publishing: 85] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 43. | Auvin S, Molinié F, Gower-Rousseau C, Brazier F, Merle V, Grandbastien B, Marti R, Lerebours E, Dupas JL, Colombel JF. Incidence, clinical presentation and location at diagnosis of pediatric inflammatory bowel disease: a prospective population-based study in northern France (1988-1999). J Pediatr Gastroenterol Nutr. 2005;41:49-55. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 161] [Cited by in F6Publishing: 143] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 44. | Rodrigo L, Riestra S, Niño P, Cadahía V, Tojo R, Fuentes D, Moreno M, González Ballina E, Fernández E. A population-based study on the incidence of inflammatory bowel disease in Oviedo (Northern Spain). Rev Esp Enferm Dig. 2004;96:296-305. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 71] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 45. | Schwarz J, Sýkora J, Cvalínová D, Pomahačová R, Klečková J, Kryl M, Včelák P. Inflammatory bowel disease incidence in Czech children: A regional prospective study, 2000-2015. World J Gastroenterol. 2017;23:4090-4101. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 13] [Cited by in F6Publishing: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 46. | Lovasz BD, Lakatos L, Horvath A, Pandur T, Erdelyi Z, Balogh M, Szipocs I, Vegh Z, Veres G, Müller KE. Incidence rates and disease course of paediatric inflammatory bowel diseases in Western Hungary between 1977 and 2011. Dig Liver Dis. 2014;46:405-411. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 47. | Sincić BM, Vucelić B, Persić M, Brncić N, Erzen DJ, Radaković B, Mićović V, Stimac D. Incidence of inflammatory bowel disease in Primorsko-goranska County, Croatia, 2000-2004: A prospective population-based study. Scand J Gastroenterol. 2006;41:437-444. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 67] [Cited by in F6Publishing: 70] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 48. | Urlep D, Blagus R, Orel R. Incidence Trends and Geographical Variability of Pediatric Inflammatory Bowel Disease in Slovenia: A Nationwide Study. Biomed Res Int. 2015;2015:921730. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 49. | Urlep D, Trop TK, Blagus R, Orel R. Incidence and phenotypic characteristics of pediatric IBD in northeastern Slovenia, 2002-2010. J Pediatr Gastroenterol Nutr. 2014;58:325-332. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 22] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 50. | Fernández A, Hernández V, Martínez-Ares D, Sanromán L, de Castro ML, Pineda JR, Carmona A, González-Portela C, Salgado C, Martínez-Cadilla J. Incidence and phenotype at diagnosis of inflammatory bowel disease. Results in Spain of the EpiCom study. Gastroenterol Hepatol. 2015;38:534-540. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 51. | Martín-de-Carpi J, Rodríguez A, Ramos E, Jiménez S, Martínez-Gómez MJ, Medina E, Navas-López VM; SPIRIT-IBD Working Group of SEGHNP (Sociedad Española de Gastroenterología Hepatología Nutrición Pediátrica). The complete picture of changing pediatric inflammatory bowel disease incidence in Spain in 25 years (1985-2009): the EXPERIENCE registry. J Crohns Colitis. 2014;8:763-769. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 56] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 52. | Malaty HM, Fan X, Opekun AR, Thibodeaux C, Ferry GD. Rising incidence of inflammatory bowel disease among children: a 12-year study. J Pediatr Gastroenterol Nutr. 2010;50:27-31. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 153] [Cited by in F6Publishing: 151] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 53. | Armitage E, Drummond HE, Wilson DC, Ghosh S. Increasing incidence of both juvenile-onset Crohn‘s disease and ulcerative colitis in Scotland. Eur J Gastroenterol Hepatol. 2001;13:1439-1447. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 121] [Cited by in F6Publishing: 123] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 54. | Benchimol EI, Guttmann A, Griffiths AM, Rabeneck L, Mack DR, Brill H, Howard J, Guan J, To T. Increasing incidence of paediatric inflammatory bowel disease in Ontario, Canada: evidence from health administrative data. Gut. 2009;58:1490-1497. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 274] [Cited by in F6Publishing: 299] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 55. | Molodecky NA, Soon IS, Rabi DM, Ghali WA, Ferris M, Chernoff G, Benchimol EI, Panaccione R, Ghosh S, Barkema HW. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology. 2012;142:46-54.e42; quiz e30. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3134] [Cited by in F6Publishing: 3262] [Article Influence: 271.8] [Reference Citation Analysis (1)] |
| 56. | Ekbom A, Helmick C, Zack M, Adami HO. The epidemiology of inflammatory bowel disease: a large, population-based study in Sweden. Gastroenterology. 1991;100:350-358. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 322] [Cited by in F6Publishing: 323] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 57. | Cosgrove M, Al-Atia RF, Jenkins HR. The epidemiology of paediatric inflammatory bowel disease. Arch Dis Child. 1996;74:460-461. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 107] [Cited by in F6Publishing: 144] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 58. | Loftus CG, Loftus EV Jr, Harmsen WS, Zinsmeister AR, Tremaine WJ, Melton LJ 3rd, Sandborn WJ. Update on the incidence and prevalence of Crohn‘s disease and ulcerative colitis in Olmsted County, Minnesota, 1940-2000. Inflamm Bowel Dis. 2007;13:254-261. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 473] [Cited by in F6Publishing: 464] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 59. | Hildebrand H, Finkel Y, Grahnquist L, Lindholm J, Ekbom A, Askling J. Changing pattern of paediatric inflammatory bowel disease in northern Stockholm 1990-2001. Gut. 2003;52:1432-1434. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 161] [Cited by in F6Publishing: 172] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 60. | Perminow G, Frigessi A, Rydning A, Nakstad B, Vatn MH. Incidence and clinical presentation of IBD in children: comparison between prospective and retrospective data in a selected Norwegian population. Scand J Gastroenterol. 2006;41:1433-1439. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 61. | Barton JR, Gillon S, Ferguson A. Incidence of inflammatory bowel disease in Scottish children between 1968 and 1983; marginal fall in ulcerative colitis, three-fold rise in Crohn‘s disease. Gut. 1989;30:618-622. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 148] [Cited by in F6Publishing: 188] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 62. | Munkholm P, Langholz E, Nielsen OH, Kreiner S, Binder V. Incidence and prevalence of Crohn‘s disease in the county of Copenhagen, 1962-87: a sixfold increase in incidence. Scand J Gastroenterol. 1992;27:609-614. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 218] [Cited by in F6Publishing: 212] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 63. | Castro M, Papadatou B, Baldassare M, Balli F, Barabino A, Barbera C, Barca S, Barera G, Bascietto F, Berni Canani R. Inflammatory bowel disease in children and adolescents in Italy: data from the pediatric national IBD register (1996-2003). Inflamm Bowel Dis. 2008;14:1246-1252. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 93] [Cited by in F6Publishing: 73] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 64. | Turunen P, Kolho KL, Auvinen A, Iltanen S, Huhtala H, Ashorn M. Incidence of inflammatory bowel disease in Finnish children, 1987-2003. Inflamm Bowel Dis. 2006;12:677-683. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 129] [Cited by in F6Publishing: 110] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 65. | Lehtinen P, Ashorn M, Iltanen S, Jauhola R, Jauhonen P, Kolho KL, Auvinen A. Incidence trends of pediatric inflammatory bowel disease in Finland, 1987-2003, a nationwide study. Inflamm Bowel Dis. 2011;17:1778-1783. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 93] [Cited by in F6Publishing: 71] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 66. | Virta LJ, Saarinen MM, Kolho KL. Inflammatory Bowel Disease Incidence is on the Continuous Rise Among All Paediatric Patients Except for the Very Young: A Nationwide Registry-based Study on 28-Year Follow-up. J Crohns Colitis. 2017;11:150-156. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 67. | Karolewska-Bochenek K, Lazowska-Przeorek I, Albrecht P, Grzybowska K, Ryzko J, Szamotulska K, Radzikowski A, Landowski P, Krzesiek E, Ignys I. Epidemiology of inflammatory bowel disease among children in Poland. A prospective, population-based, 2-year study, 2002-2004. Digestion. 2009;79:121-129. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 68. | Lindberg E, Lindquist B, Holmquist L, Hildebrand H. Inflammatory bowel disease in children and adolescents in Sweden, 1984-1995. J Pediatr Gastroenterol Nutr. 2000;30:259-264. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 142] [Cited by in F6Publishing: 152] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 69. | Mahdi B, Mahdi BM. A Review Of Inflammatory Bowel Disease Unclassified --Indeterminate Colitis. J Gastroenterol Hepatol Res. 2012;1:241-246. [Cited in This Article: ] |
| 70. | Prenzel F, Uhlig HH. Frequency of indeterminate colitis in children and adults with IBD - a metaanalysis. J Crohns Colitis. 2009;3:277-281. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 86] [Cited by in F6Publishing: 115] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 71. | Legaki E, Gazouli M. Influence of environmental factors in the development of inflammatory bowel diseases. World J Gastrointest Pharmacol Ther. 2016;7:112-125. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 57] [Cited by in F6Publishing: 60] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 72. | Kamm MA. Rapid changes in epidemiology of inflammatory bowel disease. Lancet. 2018;390:2741-2742. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 54] [Cited by in F6Publishing: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 73. | M‘Koma AE. Inflammatory bowel disease: an expanding global health problem. Clin Med Insights Gastroenterol. 2013;6:33-47. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 151] [Cited by in F6Publishing: 181] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 74. | Leach ST, Day AS, Moore D, Lemberg DA. Low rate of inflammatory bowel disease in the Australian indigenous paediatric population. J Paediatr Child Health. 2014;50:328-329. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 6] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 75. | Gismera CS, Aladrén BS. Inflammatory bowel diseases: a disease (s) of modern times? Is incidence still increasing? World J Gastroenterol. 2008;14:5491-5498. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 58] [Cited by in F6Publishing: 57] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 76. | Herrinton LJ, Liu L, Lewis JD, Griffin PM, Allison J. Incidence and prevalence of inflammatory bowel disease in a Northern California managed care organization, 1996-2002. Am J Gastroenterol. 2008;103:1998-2006. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 122] [Cited by in F6Publishing: 132] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 77. | Shapiro JM, Zoega H, Shah SA, Bright RM, Mallette M, Moniz H, Grabert SA, Bancroft B, Merrick M, Flowers NT. Incidence of Crohn‘s Disease and Ulcerative Colitis in Rhode Island: Report from the Ocean State Crohn‘s and Colitis Area Registry. Inflamm Bowel Dis. 2016;22:1456-1461. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 16] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 78. | Bitton A, Vutcovici M, Patenaude V, Sewitch M, Suissa S, Brassard P. Epidemiology of inflammatory bowel disease in Québec: recent trends. Inflamm Bowel Dis. 2014;20:1770-1776. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 46] [Cited by in F6Publishing: 52] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 79. | El-Matary W, Moroz SP, Bernstein CN. Inflammatory bowel disease in children of Manitoba: 30 years‘ experience of a tertiary center. J Pediatr Gastroenterol Nutr. 2014;59:763-766. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 38] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 80. | Lowe AM, Roy PO, B-Poulin M, Michel P, Bitton A, St-Onge L, Brassard P. Epidemiology of Crohn‘s disease in Québec, Canada. Inflamm Bowel Dis. 2009;15:429-435. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 47] [Cited by in F6Publishing: 44] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 81. | Grieci T, Bütter A. The incidence of inflammatory bowel disease in the pediatric population of Southwestern Ontario. J Pediatr Surg. 2009;44:977-980. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 82. | Griffiths AM. Specificities of inflammatory bowel disease in childhood. Best Pract Res Clin Gastroenterol. 2004;18:509-523. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 253] [Cited by in F6Publishing: 257] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 83. | Leddin D, Tamim H, Levy AR. Decreasing incidence of inflammatory bowel disease in eastern Canada: a population database study. BMC Gastroenterol. 2014;14:140. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 44] [Cited by in F6Publishing: 48] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 84. | Benchimol EI, Manuel DG, Guttmann A, Nguyen GC, Mojaverian N, Quach P, Mack DR. Changing age demographics of inflammatory bowel disease in Ontario, Canada: a population-based cohort study of epidemiology trends. Inflamm Bowel Dis. 2014;20:1761-1769. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 136] [Cited by in F6Publishing: 148] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 85. | Bentsen BS, Moum B, Ekbom A. Incidence of inflammatory bowel disease in children in southeastern Norway: a prospective population-based study 1990-94. Scand J Gastroenterol. 2002;37:540-545. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 76] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 86. | Benchimol EI, Mack DR, Nguyen GC, Snapper SB, Li W, Mojaverian N, Quach P, Muise AM. Incidence, outcomes, and health services burden of very early onset inflammatory bowel disease. Gastroenterology. 2014;147:803-813.e7; quiz e14-e15. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 176] [Cited by in F6Publishing: 175] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 87. | Vicentín R, Wagener M, Pais AB, Contreras M, Orsi M. One-year prospective registry of inflammatory bowel disease in the Argentine pediatric population. Arch Argent Pediatr. 2017;115:533-540. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 4] [Cited by in F6Publishing: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 88. | Pebody RG, Paunio M, Ruutu P. Measles, measles vaccination, and Crohn‘s disease. Crohn‘s disease has not increased in Finland. BMJ. 1998;316:1745-1746. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 19] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 89. | Lehtinen P, Pasanen K, Kolho KL, Auvinen A. Incidence of Pediatric Inflammatory Bowel Disease in Finland: An Environmental Study. J Pediatr Gastroenterol Nutr. 2016;63:65-70. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 90. | Jussila A, Virta LJ, Kautiainen H, Rekiaro M, Nieminen U, Färkkilä MA. Increasing incidence of inflammatory bowel diseases between 2000 and 2007: a nationwide register study in Finland. Inflamm Bowel Dis. 2012;18:555-561. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 39] [Cited by in F6Publishing: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 91. | Solberg IC, Vatn MH, Høie O, Stray N, Sauar J, Jahnsen J, Moum B, Lygren I; IBSEN Study Group. Clinical course in Crohn‘s disease: results of a Norwegian population-based ten-year follow-up study. Clin Gastroenterol Hepatol. 2007;5:1430-1438. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 502] [Cited by in F6Publishing: 488] [Article Influence: 28.7] [Reference Citation Analysis (0)] |
| 92. | Perminow G, Brackmann S, Lyckander LG, Franke A, Borthne A, Rydning A, Aamodt G, Schreiber S, Vatn MH; IBSEN-II Group. A characterization in childhood inflammatory bowel disease, a new population-based inception cohort from South-Eastern Norway, 2005-07, showing increased incidence in Crohn‘s disease. Scand J Gastroenterol. 2009;44:446-456. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 87] [Cited by in F6Publishing: 94] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 93. | Størdal K, Jahnsen J, Bentsen BS, Moum B. Pediatric inflammatory bowel disease in southeastern Norway: a five-year follow-up study. Digestion. 2004;70:226-230. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 94. | Moum B, Vatn MH, Ekbom A, Fausa O, Aadland E, Lygren I, Sauar J, Schulz T. Incidence of inflammatory bowel disease in southeastern Norway: evaluation of methods after 1 year of registration. Southeastern Norway IBD Study Group of Gastroenterologists. Digestion. 1995;56:377-381. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 67] [Cited by in F6Publishing: 71] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 95. | Moum B, Vatn MH, Ekbom A, Aadland E, Fausa O, Lygren I, Sauar J, Schulz T, Stray N. Incidence of ulcerative colitis and indeterminate colitis in four counties of southeastern Norway, 1990-93. A prospective population-based study. The Inflammatory Bowel South-Eastern Norway (IBSEN) Study Group of Gastroenterologists. Scand J Gastroenterol. 1996;31:362-366. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 137] [Cited by in F6Publishing: 138] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 96. | Moum B, Vatn MH, Ekbom A, Aadland E, Fausa O, Lygren I, Stray N, Sauar J, Schulz T. Incidence of Crohn‘s disease in four counties in southeastern Norway, 1990-93. A prospective population-based study. The Inflammatory Bowel South-Eastern Norway (IBSEN) Study Group of Gastroenterologists. Scand J Gastroenterol. 1996;31:355-361. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 116] [Cited by in F6Publishing: 118] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 97. | Olafsdottir EJ, Fluge G, Haug K. Chronic inflammatory bowel disease in children in western Norway. J Pediatr Gastroenterol Nutr. 1989;8:454-458. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 52] [Cited by in F6Publishing: 52] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 98. | Lindberg E, Jörnerot G. The incidence of Crohn‘s disease is not decreasing in Sweden. Scand J Gastroenterol. 1991;26:495-500. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 63] [Cited by in F6Publishing: 62] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 99. | Askling J, Grahnquist L, Ekbom A, Finkel Y. Incidence of paediatric Crohn‘s disease in Stockholm, Sweden. Lancet. 1999;354:1179. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 49] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 100. | Sjöberg D, Holmström T, Larsson M, Nielsen AL, Holmquist L, Ekbom A, Rönnblom A. Incidence and natural history of ulcerative colitis in the Uppsala Region of Sweden 2005-2009 - results from the IBD cohort of the Uppsala Region (ICURE). J Crohns Colitis. 2013;7:e351-e357. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 60] [Cited by in F6Publishing: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 101. | Sjöberg D, Holmström T, Larsson M, Nielsen AL, Holmquist L, Ekbom A, Rönnblom A. Incidence and clinical course of Crohn‘s disease during the first year - results from the IBD Cohort of the Uppsala Region (ICURE) of Sweden 2005-2009. J Crohns Colitis. 2014;8:215-222. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 90] [Cited by in F6Publishing: 89] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 102. | Vind I, Riis L, Jess T, Knudsen E, Pedersen N, Elkjaer M, Bak Andersen I, Wewer V, Nørregaard P, Moesgaard F. Increasing incidences of inflammatory bowel disease and decreasing surgery rates in Copenhagen City and County, 2003-2005: a population-based study from the Danish Crohn colitis database. Am J Gastroenterol. 2006;101:1274-1282. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 379] [Cited by in F6Publishing: 379] [Article Influence: 21.1] [Reference Citation Analysis (0)] |
| 103. | Lophaven SN, Lynge E, Burisch J. The incidence of inflammatory bowel disease in Denmark 1980-2013: a nationwide cohort study. Aliment Pharmacol Ther. 2017;45:961-972. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 85] [Cited by in F6Publishing: 103] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 104. | Jacobsen BA, Fallingborg J, Rasmussen HH, Nielsen KR, Drewes AM, Puho E, Nielsen GL, Sørensen HT. Increase in incidence and prevalence of inflammatory bowel disease in northern Denmark: a population-based study, 1978-2002. Eur J Gastroenterol Hepatol. 2006;18:601-606. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 102] [Cited by in F6Publishing: 97] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 105. | Langholz E, Munkholm P, Krasilnikoff PA, Binder V. Inflammatory bowel diseases with onset in childhood. Clinical features, morbidity, and mortality in a regional cohort. Scand J Gastroenterol. 1997;32:139-147. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 186] [Cited by in F6Publishing: 186] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 106. | Langholz E, Munkholm P, Nielsen OH, Kreiner S, Binder V. Incidence and prevalence of ulcerative colitis in Copenhagen county from 1962 to 1987. Scand J Gastroenterol. 1991;26:1247-1256. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 182] [Cited by in F6Publishing: 188] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 107. | Fonager K, Sørensen HT, Olsen J. Change in incidence of Crohn‘s disease and ulcerative colitis in Denmark. A study based on the National Registry of Patients, 1981-1992. Int J Epidemiol. 1997;26:1003-1008. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 58] [Cited by in F6Publishing: 62] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 108. | Urne FU, Paerregaard A. [Chronic inflammatory bowel disease in children. An epidemiological study from eastern Denmark 1998-2000]. Ugeskr Laeger. 2002;164:5810-5814. [PubMed] [Cited in This Article: ] |
| 109. | Larsen L, Jensen MD, Larsen MD, Nielsen RG, Thorsgaard N, Vind I, Wildt S, Kjeldsen J. The Danish National Registry for Biological Therapy in Inflammatory Bowel Disease. Clin Epidemiol. 2016;8:607-612. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 110. | Jakobsen C, Paerregaard A, Munkholm P, Faerk J, Lange A, Andersen J, Jakobsen M, Kramer I, Czernia-Mazurkiewicz J, Wewer V. Pediatric inflammatory bowel disease: increasing incidence, decreasing surgery rate, and compromised nutritional status: A prospective population-based cohort study 2007-2009. Inflamm Bowel Dis. 2011;17:2541-2550. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 92] [Cited by in F6Publishing: 71] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 111. | Nørgård BM, Nielsen J, Fonager K, Kjeldsen J, Jacobsen BA, Qvist N. The incidence of ulcerative colitis (1995-2011) and Crohn‘s disease (1995-2012) - based on nationwide Danish registry data. J Crohns Colitis. 2014;8:1274-1280. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 59] [Cited by in F6Publishing: 62] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 112. | Björnsson S, Jóhannsson JH. Inflammatory bowel disease in Iceland, 1990-1994: a prospective, nationwide, epidemiological study. Eur J Gastroenterol Hepatol. 2000;12:31-38. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 90] [Cited by in F6Publishing: 91] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 113. | Armitage E, Drummond H, Ghosh S, Ferguson A. Incidence of juvenile-onset Crohn‘s disease in Scotland. Lancet. 1999;353:1496-1497. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 64] [Cited by in F6Publishing: 74] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 114. | Armitage EL, Aldhous MC, Anderson N, Drummond HE, Riemersma RA, Ghosh S, Satsangi J. Incidence of juvenile-onset Crohn‘s disease in Scotland: association with northern latitude and affluence. Gastroenterology. 2004;127:1051-1057. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 112] [Cited by in F6Publishing: 124] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 115. | Watson AJ, Johnston AT, Barker PM, Youngson GG, Bisset WM, Mahomed AA. The presentation and management of juvenile-onset chronic inflammatory bowel disease in Northeastern Scotland. J Pediatr Surg. 2002;37:83-86. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 116. | Ashton JJ, Wiskin AE, Ennis S, Batra A, Afzal NA, Beattie RM. Rising incidence of paediatric inflammatory bowel disease (PIBD) in Wessex, Southern England. Arch Dis Child. 2014;99:659-664. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 117. | Ashton JJ, Cullen M, Afzal NA, Coelho T, Batra A, Beattie RM. Is the incidence of paediatric inflammatory bowel disease still increasing? Arch Dis Child. 2018; Epub ahead of print. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 118. | Hassan K, Cowan FJ, Jenkins HR. The incidence of childhood inflammatory bowel disease in Wales. Eur J Pediatr. 2000;159:261-263. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 51] [Cited by in F6Publishing: 54] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 119. | Gunesh S, Thomas GA, Williams GT, Roberts A, Hawthorne AB. The incidence of Crohn‘s disease in Cardiff over the last 75 years: an update for 1996-2005. Aliment Pharmacol Ther. 2008;27:211-219. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 55] [Cited by in F6Publishing: 49] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 120. | Hope B, Shahdadpuri R, Dunne C, Broderick AM, Grant T, Hamzawi M, O‘Driscoll K, Quinn S, Hussey S, Bourke B. Rapid rise in incidence of Irish paediatric inflammatory bowel disease. Arch Dis Child. 2012;97:590-594. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 60] [Cited by in F6Publishing: 65] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 121. | Coughlan A, Wylde R, Lafferty L, Quinn S, Broderick A, Bourke B, Hussey S; DOCHAS Study. A rising incidence and poorer male outcomes characterise early onset paediatric inflammatory bowel disease. Aliment Pharmacol Ther. 2017;45:1534-1541. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 122. | Petritsch W, Fuchs S, Berghold A, Bachmaier G, Högenauer C, Hauer AC, Weiglhofer U, Wenzl HH. Incidence of inflammatory bowel disease in the province of Styria, Austria, from 1997 to 2007: a population-based study. J Crohns Colitis. 2013;7:58-69. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 123. | Ott C, Obermeier F, Thieler S, Kemptner D, Bauer A, Schölmerich J, Rogler G, Timmer A. The incidence of inflammatory bowel disease in a rural region of Southern Germany: a prospective population-based study. Eur J Gastroenterol Hepatol. 2008;20:917-923. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 75] [Cited by in F6Publishing: 68] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 124. | van der Zaag-Loonen HJ, Casparie M, Taminiau JA, Escher JC, Pereira RR, Derkx HH. The incidence of pediatric inflammatory bowel disease in the Netherlands: 1999-2001. J Pediatr Gastroenterol Nutr. 2004;38:302-307. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 63] [Cited by in F6Publishing: 46] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 125. | Gower-Rousseau C, Vasseur F, Fumery M, Savoye G, Salleron J, Dauchet L, Turck D, Cortot A, Peyrin-Biroulet L, Colombel JF. Epidemiology of inflammatory bowel diseases: new insights from a French population-based registry (EPIMAD). Dig Liver Dis. 2013;45:89-94. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 128] [Cited by in F6Publishing: 117] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 126. | Bequet E, Sarter H, Fumery M, Vasseur F, Armengol-Debeir L, Pariente B, Ley D, Spyckerelle C, Coevoet H, Laberenne JE. Incidence and Phenotype at Diagnosis of Very-early-onset Compared with Later-onset Paediatric Inflammatory Bowel Disease: A Population-based Study [1988-2011]. J Crohns Colitis. 2017;11:519-526. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 127. | Vernier-Massouille G, Balde M, Salleron J, Turck D, Dupas JL, Mouterde O, Merle V, Salomez JL, Branche J, Marti R. Natural history of pediatric Crohn‘s disease: a population-based cohort study. Gastroenterology. 2008;135:1106-1113. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 427] [Cited by in F6Publishing: 420] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 128. | Abakar-Mahamat A, Filippi J, Pradier C, Dozol A, Hébuterne X. Incidence of inflammatory bowel disease in Corsica from 2002 to 2003. Gastroenterol Clin Biol. 2007;31:1098-1103. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 64] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 129. | Tourtelier Y, Dabadie A, Tron I, Alexandre JL, Robaskiewicz M, Cruchant E, Seyrig JA, Heresbach D, Bretagne JF. Incidence of inflammatory bowel disease in children in Brittany (1994-1997). Arch Pediatr. 2000;7:377-384. [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 23] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 130. | Gottrand F, Colombel JF, Moreno L, Salomez JL, Farriaux JP, Cortot A. [Incidence of inflammatory bowel diseases in children in the Nord-Pas-de-Calais region]. Arch Fr Pediatr. 1991;48:25-28. [PubMed] [Cited in This Article: ] |
| 131. | Ghione S, Sarter H, Fumery M, Armengol-Debeir L, Savoye G, Ley D, Spyckerelle C, Pariente B, Peyrin-Biroulet L, Turck D. Dramatic Increase in Incidence of Ulcerative Colitis and Crohn‘s Disease (1988-2011): A Population-Based Study of French Adolescents. Am J Gastroenterol. 2018;113:265-272. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 83] [Cited by in F6Publishing: 101] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 132. | Pittet V, Juillerat P, Mottet C, Felley C, Ballabeni P, Burnand B, Michetti P, Vader JP; Swiss IBD Cohort Study Group. Cohort profile: the Swiss Inflammatory Bowel Disease Cohort Study (SIBDCS). Int J Epidemiol. 2009;38:922-931. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 121] [Cited by in F6Publishing: 125] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 133. | De Greef E, Mahachie John JM, Hoffman I, Smets F, Van Biervliet S, Scaillon M, Hauser B, Paquot I, Alliet P, Arts W, Dewit O, Peeters H, Baert F, D‘Haens G, Rahier JF, Etienne I, Bauraind O, Van Gossum A, Vermeire S, Fontaine F, Muls V, Louis E, Van de Mierop F, Coche JC, Van Steen K, Veereman G; IBD working group of the Belgian Society of Pediatric Gastroenterology, Hepatology and Nutrition (BeSPGHAN); Belgian IBD Research and Development. Profile of pediatric Crohn‘s disease in Belgium. J Crohns Colitis. 2013;7:e588-e598. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 134. | Ranzi T, Bodini P, Zambelli A, Politi P, Lupinacci G, Campanini MC, Dal Lago AL, Lisciandrano D, Bianchi PA. Epidemiological aspects of inflammatory bowel disease in a north Italian population: a 4-year prospective study. Eur J Gastroenterol Hepatol. 1996;8:657-661. [PubMed] [Cited in This Article: ] |
| 135. | Di Domenicantonio R, Cappai G, Arcà M, Agabiti N, Kohn A, Vernia P, Biancone L, Armuzzi A, Papi C, Davoli M. Occurrence of inflammatory bowel disease in central Italy: a study based on health information systems. Dig Liver Dis. 2014;46:777-782. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 33] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 136. | Trallori G, Palli D, Saieva C, Bardazzi G, Bonanomi AG, d‘Albasio G, Galli M, Vannozzi G, Milla M, Tarantino O. A population-based study of inflammatory bowel disease in Florence over 15 years (1978-92). Scand J Gastroenterol. 1996;31:892-899. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 84] [Cited by in F6Publishing: 87] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 137. | Arin Letamendia A, Borda Celaya F, Burusco Paternain MJ, Prieto Martínez C, Martínez Echeverría A, Elizalde Apestegui I, Laiglesia Izquierdo M, Macias Mendizábal E, Tamburri Moso P, Sánchez Valverde F. [High incidence rates of inflammatory bowel disease in Navarra (Spain). Results of a prospective, population-based study]. Gastroenterol Hepatol. 2008;31:111-116. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 80] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 138. | Cachia E, Calleja N, Aakeroy R, Degaetano J, Vassallo M. Incidence of inflammatory bowel disease in Malta between 1993 and 2005: a retrospective study. Inflamm Bowel Dis. 2008;14:550-553. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 139. | Pozler O, Maly J, Bonova O, Dedek P, Frühauf P, Havlickova A, Janatova T, Jimramovsky F, Klimova L, Klusacek D. Incidence of Crohn disease in the Czech Republic in the years 1990 to 2001 and assessment of pediatric population with inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 2006;42:186-189. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 43] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 140. | Kolek A, Janout V, Tichý M, Grepl M. The incidence of inflammatory bowel disease is increasing among children 15 years old and younger in the Czech Republic. J Pediatr Gastroenterol Nutr. 2004;38:362-363. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 141. | Müller KE, Lakatos PL, Arató A, Kovács JB, Várkonyi Á, Szűcs D, Szakos E, Sólyom E, Kovács M, Polgár M, Nemes É, Guthy I, Tokodi I, Tóth G, Horváth Á, Tárnok A, Csoszánszki N, Balogh M, Vass N, Bódi P, Dezsőfi A, Gárdos L, Micskey E, Papp M, Cseh Á, Szabó D, Vörös P, Veres G; Hungarian IBD Registry Group (HUPIR). Incidence, Paris classification, and follow-up in a nationwide incident cohort of pediatric patients with inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 2013;57:576-582. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 73] [Cited by in F6Publishing: 60] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 142. | Lakatos L, Kiss LS, David G, Pandur T, Erdelyi Z, Mester G, Balogh M, Szipocs I, Molnar C, Komaromi E. Incidence, disease phenotype at diagnosis, and early disease course in inflammatory bowel diseases in Western Hungary, 2002-2006. Inflamm Bowel Dis. 2011;17:2558-2565. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 115] [Cited by in F6Publishing: 120] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 143. | Orel R, Kamhi T, Vidmar G, Mamula P. Epidemiology of pediatric chronic inflammatory bowel disease in central and western Slovenia, 1994-2005. J Pediatr Gastroenterol Nutr. 2009;48:579-586. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 34] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 144. | Vucelić B, Korać B, Sentić M, Milicić D, Hadzić N, Juresa V, Bozikov J, Rotkvić I, Buljevac M, Kovacević I. Ulcerative colitis in Zagreb, Yugoslavia: incidence and prevalence 1980-1989. Int J Epidemiol. 1991;20:1043-1047. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 39] [Cited by in F6Publishing: 39] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 145. | Vucelić B, Korać B, Sentić M, Milicić D, Hadzić N, Juresa V, Bozikov J, Rotkvić I, Buljevac M, Kovacević I. Epidemiology of Crohn‘s disease in Zagreb, Yugoslavia: a ten-year prospective study. Int J Epidemiol. 1991;20:216-220. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 41] [Cited by in F6Publishing: 40] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 146. | Ahmaida A, Al-Shaikhi S. Childhood Inflammatory Bowel Disease in Libya: Epidemiological and Clinical features. Libyan J Med. 2009;4:70-74. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5] [Cited by in F6Publishing: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 147. | Odes HS, Locker C, Neumann L, Zirkin HJ, Weizman Z, Sperber AD, Fraser GM, Krugliak P, Gaspar N, Eidelman L. Epidemiology of Crohn‘s disease in southern Israel. Am J Gastroenterol. 1994;89:1859-1862. [PubMed] [Cited in This Article: ] |
| 148. | Grossman A, Fireman Z, Lilos P, Novis B, Rozen P, Gilat T. Epidemiology of ulcerative colitis in the Jewish population of central Israel 1970-1980. Hepatogastroenterology. 1989;36:193-197. [PubMed] [Cited in This Article: ] |
| 149. | El Mouzan MI, Saadah O, Al-Saleem K, Al Edreesi M, Hasosah M, Alanazi A, Al Mofarreh M, Asery A, Al Qourain A, Nouli K. Incidence of pediatric inflammatory bowel disease in Saudi Arabia: a multicenter national study. Inflamm Bowel Dis. 2014;20:1085-1090. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 150. | El Mouzan MI, Abdullah AM, Al Habbal MT. Epidemiology of juvenile-onset inflammatory bowel disease in central Saudi Arabia. J Trop Pediatr. 2006;52:69-71. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 36] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 151. | Al-Qabandi WA, Buhamrah EK, Hamadi KA, Al-Osaimi SA, Al-Ruwayeh AA, Madda J. Inflammatory bowel disease in children, an evolving problem in Kuwait. Saudi J Gastroenterol. 2011;17:323-327. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 152. | Zayyani NR, Malaty HM, Graham DY. Increasing Incidence of Crohn‘s Disease with Familial Clustering in the Kingdom of Bahrain: A 25-Year Population-based Study. Inflamm Bowel Dis. 2017;23:304-309. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 4] [Cited by in F6Publishing: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 153. | Wang XQ, Zhang Y, Xu CD, Jiang LR, Huang Y, Du HM, Wang XJ. Inflammatory bowel disease in Chinese children: a multicenter analysis over a decade from Shanghai. Inflamm Bowel Dis. 2013;19:423-428. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 52] [Cited by in F6Publishing: 45] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 154. | Tsai CH, Chen HL, Ni YH, Hsu HY, Jeng YM, Chang CJ, Chang MH. Characteristics and trends in incidence of inflammatory bowel disease in Taiwanese children. J Formos Med Assoc. 2004;103:685-691. [PubMed] [Cited in This Article: ] |
| 155. | Chu HP, Logarajah V, Tan N, Phua KB. Paediatric inflammatory bowel disease in a multiracial Asian country. Singapore Med J. 2013;54:201-205. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 156. | Yang SK, Hong WS, Min YI, Kim HY, Yoo JY, Rhee PL, Rhee JC, Chang DK, Song IS, Jung SA. Incidence and prevalence of ulcerative colitis in the Songpa-Kangdong District, Seoul, Korea, 1986-1997. J Gastroenterol Hepatol. 2000;15:1037-1042. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 140] [Cited by in F6Publishing: 141] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 157. | Jung YS, Han M, Kim WH, Park S, Cheon JH. Incidence and Clinical Outcomes of Inflammatory Bowel Disease in South Korea, 2011-2014: A Nationwide Population-Based Study. Dig Dis Sci. 2017;62:2102-2112. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 54] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 158. | Phavichitr N, Cameron DJ, Catto-Smith AG. Increasing incidence of Crohn‘s disease in Victorian children. J Gastroenterol Hepatol. 2003;18:329-332. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 78] [Cited by in F6Publishing: 87] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 159. | Schildkraut V, Alex G, Cameron DJ, Hardikar W, Lipschitz B, Oliver MR, Simpson DM, Catto-Smith AG. Sixty-year study of incidence of childhood ulcerative colitis finds eleven-fold increase beginning in 1990s. Inflamm Bowel Dis. 2013;19:1-6. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 160. | Lopez RN, Evans HM, Appleton L, Bishop J, Chin S, Mouat S, Gearry RB, Day AS. Prospective Incidence of Paediatric Inflammatory Bowel Disease in New Zealand in 2015: Results From the Paediatric Inflammatory Bowel Disease in New Zealand (PINZ) Study. J Pediatr Gastroenterol Nutr. 2018;66:e122-e126. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 161. | Lopez RN, Appleton L, Gearry RB, Day AS. Rising Incidence of Paediatric Inflammatory Bowel Disease in Canterbury, New Zealand, 1996-2015. J Pediatr Gastroenterol Nutr. 2018;66:e45-e50. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 162. | Levine A, Koletzko S, Turner D, Escher JC, Cucchiara S, de Ridder L, Kolho KL, Veres G, Russell RK, Paerregaard A. ESPGHAN revised porto criteria for the diagnosis of inflammatory bowel disease in children and adolescents. J Pediatr Gastroenterol Nutr. 2014;58:795-806. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 822] [Cited by in F6Publishing: 811] [Article Influence: 81.1] [Reference Citation Analysis (0)] |
| 163. | Haug K, Schrumpf E, Barstad S, Fluge G, Halvorsen JF. Epidemiology of ulcerative colitis in western Norway. Scand J Gastroenterol. 1988;23:517-522. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 46] [Cited by in F6Publishing: 42] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 164. | Hildebrand H, Brydolf M, Holmquist L, Krantz I, Kristiansson B. Incidence and prevalence of inflammatory bowel disease in children in south-western Sweden. Acta Paediatr. 1994;83:640-645. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 41] [Cited by in F6Publishing: 43] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 165. | Tysk C, Järnerot G. Ulcerative proctocolitis in Orebro, Sweden. A retrospective epidemiologic study, 1963-1987. Scand J Gastroenterol. 1992;27:945-950. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 74] [Cited by in F6Publishing: 70] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 166. | Hildebrand H, Fredrikzon B, Holmquist L, Kristiansson B, Lindquist B. Chronic inflammatory bowel disease in children and adolescents in Sweden. J Pediatr Gastroenterol Nutr. 1991;13:293-297. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 60] [Cited by in F6Publishing: 63] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 167. | Gollop JH, Phillips SF, Melton LJ 3rd, Zinsmeister AR. Epidemiologic aspects of Crohn‘s disease: a population based study in Olmsted County, Minnesota, 1943-1982. Gut. 1988;29:49-56. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 187] [Cited by in F6Publishing: 200] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 168. | Loftus EV Jr, Silverstein MD, Sandborn WJ, Tremaine WJ, Harmsen WS, Zinsmeister AR. Ulcerative colitis in Olmsted County, Minnesota, 1940-1993: incidence, prevalence, and survival. Gut. 2000;46:336-343. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 360] [Cited by in F6Publishing: 387] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 169. | Kuo CJ, Yu KH, See LC, Chiu CT, Su MY, Hsu CM, Kuo CF, Chiou MJ, Liu JR, Wang HW. The Trend of Inflammatory Bowel Diseases in Taiwan: A Population-Based Study. Dig Dis Sci. 2015;60:2454-2462. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 22] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 170. | González FN, García BC, Polo RE, Hernández CM. Inflammatory bowel disease in pediatric patients in Asturias, Spain (1993-2000): epidemiology and clinical features. Acta Pediatr Esp. 2004;62:466-472. [Cited in This Article: ] |
| 171. | Lopez Miguel C, Sicilia B, Sierra E, Lopez Zaborras J, Arribas F, Gomollon F. [Incidence of inflammatory bowel disease in Aragon: outcome of a prospective population-based study]. Gastroenterol Hepatol. 1999;22:323-328. [PubMed] [Cited in This Article: ] |