Published online Jun 21, 2013. doi: 10.3748/wjg.v19.i23.3685
Revised: February 14, 2013
Accepted: March 15, 2013
Published online: June 21, 2013
Walled-off pancreatic necrosis and a pancreatic abscess are the most severe complications of acute pancreatitis. Surgery in such critically ill patients is often associated with significant morbidity and mortality within the first few weeks after the onset of symptoms. Minimal invasive approaches with high success and low mortality rates are therefore of considerable interest. Endoscopic therapy has the potential to offer safe and effective alternative treatment. We report here on 3 consecutive patients with infected walled-off pancreatic necrosis and 1 patient with a pancreatic abscess who underwent direct endoscopic necrosectomy 19-21 d after the onset of acute pancreatitis. The infected pancreatic necrosis or abscess was punctured transluminally with a cystostome and, after balloon dilatation, a non-covered self-expanding biliary metal stent was placed into the necrotic cavity. Following stent deployment, a nasobiliary pigtail catheter was placed into the cavity to ensure continuous irrigation. After 5-7 d, the metal stent was removed endoscopically and the necrotic cavity was entered with a therapeutic gastroscope. Endoscopic debridement was performed via the simultaneous application of a high-flow water-jet system; using a flush knife, a Dormia basket, and hot biopsy forceps. The transluminal endotherapy was repeated 2-5 times daily during the next 10 d. Supportive care included parenteral antibiotics and jejunal feeding. All patients improved dramatically and with resolution of their septic conditions; 3 patients were completely cured without any further complications or the need for surgery. One patient died from a complication of prolonged ventilation severe bilateral pneumonia, not related to the endoscopic procedure. No procedure related complications were observed. Transluminal endoscopic necrosectomy with temporary application of a self-expanding metal stent and a high-flow water-jet system shows promise for enhancing the potential of this endoscopic approach in patients with walled-off pancreatic necrosis and/or a pancreatic abscess.
Core tip: The endoscopic transluminal management of pancreatic necrosis and/or pancreatic abscess is associated with good initial and long-term clinical success, with acceptable morbidity and mortality rates. The advantages of endoscopic management are related to its minimal invasiveness. The combination of multiple endoscopic approaches is designed to achieve the goals of any treatment strategy.
- Citation: Hritz I, Fejes R, Székely A, Székely I, Horváth L, Sárkány &, Altorjay &, Madácsy L. Endoscopic transluminal pancreatic necrosectomy using a self-expanding metal stent and high-flow water-jet system. World J Gastroenterol 2013; 19(23): 3685-3692
- URL: https://www.wjgnet.com/1007-9327/full/v19/i23/3685.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i23.3685
Acute pancreatitis (AP) is a disease characterized by significant morbidity and mortality. Pancreatic necrosis (PN), occurring as diffuse or focal areas of non-viable pancreatic parenchyma as defined by the 1992 Atlanta classification, is a serious complication that can develop within a few days following the onset of AP[1].
Acute necrotizing pancreatitis (ANP) complicates 15%-20% of all cases with AP and, in patients in whom the most severe infectious complication infected necrosis develops, the mortality can be as high as 25%-70%[2].
“Organized” or “walled-off pancreatic necrosis” (WOPN), described as relatively well-circumscribed areas of PN, evolves during the several weeks after an episode of severe ANP. This process reflects the evolution from early diffuse PN to an encapsulated, loculated form[3].
Similarly, a pancreatic abscess (PA), a serious complication of AP, can be defined as a circumscribed intra-abdominal collection of purulent material adjacent to the pancreas, containing little or no PN, but circumscribed within a well-defined wall[1].
The conventional treatment of infected WOPN and PA necessitates open surgical debridement, together with sump drainage and continuous lavage. The key objectives of the surgical approach are the removal of all pancreatic- and peripancreatic necrotic tissue, the evacuation of purulent infected material, and the provision of continuous and adequate drainage to promote resolution of the inflammatory processes. The best current therapeutic approach comprises complete removal of the necrotic material, but preservation of all the viable pancreatic tissue, together with the best possible supportive care, preferably at an intensive care unit (ICU).
The recommended surgical procedure of open necrosectomy is associated with a high postoperative complication rate (95%)[4], and the median mortality rate has been reported to be 25% (range 6%-56%)[5].
Minimally invasive approaches with high success and lower mortality rates are therefore of considerable interest, and various percutaneous and endoscopic techniques for the management of PN and PAs have been described during the last decade.
Transluminal retroperitoneal endoscopy for the debridement of infected WOPN can be considered one of the first clinical applications of natural orifice transluminal endoscopic surgery. However, the history of endoscopic necrosectomy began with the endoscopic transmural drainage of pancreatic pseudocysts and abscesses[3].
Endoscopic interventions are typically performed under conscious sedation without the need for general anesthesia. Endoscopic therapy for PN may involve endoscopic retrograde cholangiopancreatography (ERCP) with sphincterotomy, stenting, and nasojejunal feeding tube placement, while a more aggressive and potent therapeutic approach is transmural (transgastric or transduodenal) endoscopic drainage[3], with or without endoscopic ultrasonographic guidance[6,7], followed by balloon dilation of the cystogastrostoma or cystoduodenostoma, and repeated direct endoscopic necrosectomies through the dilated fistulous tract using different accessories (e.g., a polypectomy snare, Dormia basket, hot biopsy forceps, or tripod grasper)[8,9], and stent and/or catheter placement into the necrotic cavity, followed by continuous lavage and irrigation[10] and, if required, repeated balloon dilations of the fistulous tract.
Two large, multicenter retrospective studies have demonstrated that the direct transluminal endoscopic management of PN is associated with good long-term maintenance of the high initial success rate[11,12]. Nevertheless, all of the current endoscopic techniques have obvious inherent limitations, such as the risk of air embolism, endoscopically uncontrollable bleeding, and inadequate drainage through multiple plastic stents, together with an early occlusion of the fistulous tract.
To overcome these difficulties, we demonstrate here a new and successful method of endoscopic transluminal necrosectomy (ETN); a combination of the temporary placement of a self-expanding metal stent (SEMS) into the fistulous tract and daily irrigations of the necrotic cavity with a high-flow water-jet system, using a flush knife.
Direct endoscopic therapy was performed on 4 patients. The indication for the endoscopic transmural approach was infected PN in 3 cases and a PA in 1 case.
A 59-year-old female was admitted to our emergency sub-ICU with AP. At admission, she had severe upper abdominal pain that had started 24 h before. Abdominal ultrasonography revealed multiple small stones in the gallbladder, with concomitant dilation of the extrahepatic bile ducts. Laboratory tests demonstrated leukocytosis (26 g/L), and elevated blood glucose (13.4 mmol/L), liver function tests [aspartate aminotransferase (AST): 76 U/L, alanine aminotransferase (ALT): 106 U/L, gamma-glutamyl transferase (GGT): 389 U/L], and serum amylase (2262 U/L). The calculated Glasgow score was 7, which predicted a severe attack of pancreatitis. Emergency ERCP was performed, and after endoscopic sphincterotomy a small (5 mm in diameter) impacted gallstone was successfully removed from the common bile duct. Despite maximum conservative therapy, an abdominal computed tomography (CT) scan 72 h later demonstrated severe APN with extensive necrosis and peripancreatic fluid accumulation was observed (Balthazar score: 6). After transmission to the ICU she received maximum supportive care, with mechanical ventilation, parenteral antibiotics (Imipenem and Metronidazole), parenteral volume replacement and nasojejunal feeding, epidural anesthesia and plasmapheresis. Despite the optimum therapy, her general condition was still deteriorating at the end of the third week, due to a high fever, sepsis, and multi-organ failure. Laboratory tests showed elevated C-reactive protein (CRP) and procalcitonin (PCT) levels (226 mg/L and 2.72 μg/L, respectively). A repeated CT scan demonstrated an approximately 14 cm × 11 cm × 12 cm volume of not clearly demarcated peripancreatic necrosis with accumulated fluid located between the stomach and the body of the pancreas; clinically, infection of the necrosis was assumed. After repeated surgical consultations, we decided to attempt ETN rather than open surgical necrosectomy in order to minimize the level of invasiveness in this critically-ill patient on day 21 after the onset of ANP.
A 62-year-old female patient was transferred from a secondary care hospital to our gastroenterological sub-ICU with upper abdominal pain and an elevated serum amylase level (4000 U/L). She had experienced the first attack of abdominal pain 2 wk earlier, though she then became asymptomatic, but the day before admission she again developed severe abdominal pain after a fatty food intake. At admission, the calculated Glasgow score was 8, indicating a severe attack of AP. On abdominal ultrasonography a large (2 cm in diameter) solitary gallbladder stone was detected. Laboratory tests demonstrated leukocytosis (21 G/L), elevated blood glucose (21 mmol/L), liver function tests (AST: 439 U/L, ALT: 339 U/L, GGT: 308 U/L), and serum amylase (1152 U/L). Emergency ERCP was performed, but, apart from the known gallbladder stone, no cholangiographic signs of biliary obstruction or gallstone pancreatitis were seen. Two days later, due to high fever and sepsis, she was transferred to the ICU. An abdominal CT scan on day 4 of hospitalization revealed ANP with extensive necrosis, duodenal compression, and an accumulation of peripancreatic fluid 45 mm in width at the bursa omentalis (Balthazar score: 7). Maximum supportive care at the ICU included parenteral antibiotics (Imipenem + Amikacin, followed by Tigecycline and Metronidazole), parenteral volume replacement, nasojejunal feeding, and epidural anesthesia; there was no need for long-term mechanical ventilation. A repeated CT scan demonstrated an 11 cm × 7 cm × 7.5 cm volume of peripancreatic necrosis with the extensive accumulation of fluid mainly between the posterior wall of the stomach and the pancreas. At the end of the third week, she still had high fever, and presented extremely high CRP (326 mg/L) and elevated PCT (2.67 μg/L) levels. From the clinical signs and laboratory results, infection of the PN was evident and therefore, after repeated surgical consultations, we decided to perform ETN on day 20.
A 72-year-old female patient was admitted to our department with upper abdominal pain and vomiting that had persisted for 6 d before hospitalization. Her case history included hypertension and a vertebrobasilar stroke 7 years previously. At admission, laboratory tests revealed only mild leukocytosis (10.2 g/L), but high CRP (249.6 mg/L), elevated lactate dehydrogenase (741 U/L) and blood urea nitrogen (14.8 mmol/L), and mildly increased serum amylase (136 U/L). The liver function tests were normal, with the exception of mild GGT elevation (110 U/L). The clinical signs and abdominal ultrasonography (no gallstones or bile duct dilatation) led to a diagnosis of idiopathic AP. Despite maximum supportive care, with parenteral antibiotics (Ciprofloxacin and Metronidazole), parenteral volume replacement, nasojejunal feeding, and epidural anesthesia, an abdominal CT scan 3 wk later demonstrated severe ANP, with complete destruction of the pancreatic tissue, secondary antral and duodenal compression, and a 10 cm wide accumulation of peripancreatic fluid in the retrogastric region, from the liver hilum up to the spleen, but not clearly demarcated from the retroperitoneal tissue (Balthazar score: 9). After repeated surgical consultations, we decided on ETN rather than open surgical necrosectomy in order to minimize the invasiveness in this elderly patient with significant co-morbidity, on day 31 after the onset of ANP.
A 52-year-old male patient was admitted to our department with upper abdominal pain and nausea. The symptoms had developed after a spicy and fatty food intake. The case history revealed two attacks of AP one year previously, with the consequent development of a pancreatic pseudocyst. At admission, laboratory tests indicated mild leukocytosis (12.2 g/L), CRP (15.2 mg/L), and mildly elevated blood glucose (6.5 mmol/L) levels. Liver function tests were normal, but the serum level of amylase was increased (653 U/L). On abdominal ultrasonography a large (11 cm × 4 cm) pancreatic pseudocyst was seen, without signs of bile duct dilation. A temporary improvement was achieved on supportive care, but on day 7 of hospitalization a sudden deterioration was observed and the patient was transferred to the ICU. An abdominal CT scan revealed a PA (8.5 cm × 5 cm). Despite maximum supportive care with mechanical ventilation, parenteral antibiotics (Imipenem and Metronidazole, followed by Amikacin), parenteral volume replacement, and nasojejunal feeding, no improvement was achieved. After repeated surgical consultations, we decided to perform ETN on day 13 after the onset of AP.
All patients provided informed consent before each endoscopic intervention. The endoscopic procedures were performed with the patients under conscious sedation with intravenous benzodiazepine and/or opioid administration. ERCP was carried out when indicated.
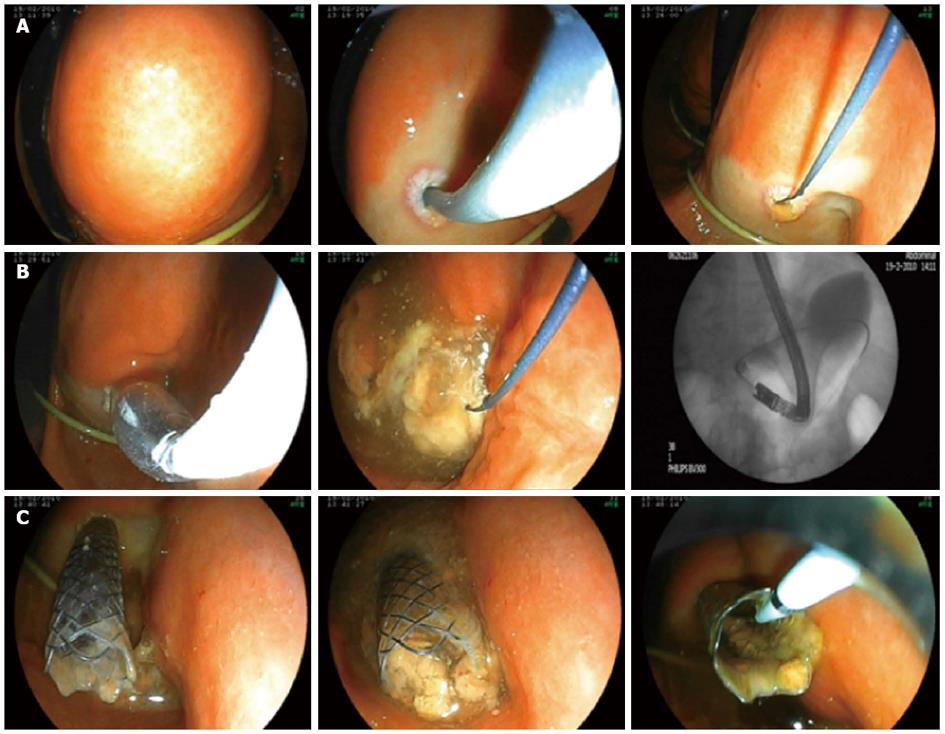
The feasibility of endoscopic drainage was assessed by endoscopic ultrasonography with the application of a linear echoendoscope (Fujinon EG-530UT). The optimum drainage site was selected, and vessel interposition was excluded by color-flow Doppler. After infiltration of the gastric wall with 20 mL of diluted adrenaline (1:10000 in saline), the cavity was punctured with a 22-gauge needle for sample collection (for microbiological analysis). Next, the gastric wall was perforated with a cystostome (Wilson-Cook, 60 W, forced coagulation) to access the necrotic cavity, followed by dilatation of the fistulous tract up to 12 F with the outer dilatation catheter of the same device. Contrast medium was then injected to assess the extent and borders of the cavity. After this, a 480 cm long, 0.035 inch diameter guidewire (Tracer Wire Guide; Wilson-Cook) was carefully advanced through the cystostome to form at least two loops inside the cavity, so as to facilitate safe catheter exchange. The puncture site was dilated with a wire-guided biliary balloon up to 10 mm and 1.5 psi (Olympus Endoscopy). Finally, a 6 cm long, 10 mm wide throughout, non-covered, biliary SEMS (Micro-Tech Europe) was positioned and opened inside the fistulous tract in order to make a permanent and wide connection between the necrotic cavity and the stomach. After opening of the SEMS, a large volume of purulent fluid and necrotic material emptied spontaneously into the stomach, and this was further facilitated with continuous flushing and suction via the endoscope. We finalized the first stage of the necrosectomy process with the placement of a 12 F nasobiliary pigtail catheter into the necrotic cavity through the SEMS. During the next 5 d, physiological saline was irrigated through this catheter at a flow rate of 100 mL/h to facilitate continuous lavage and optimum drainage of the necrotic material.
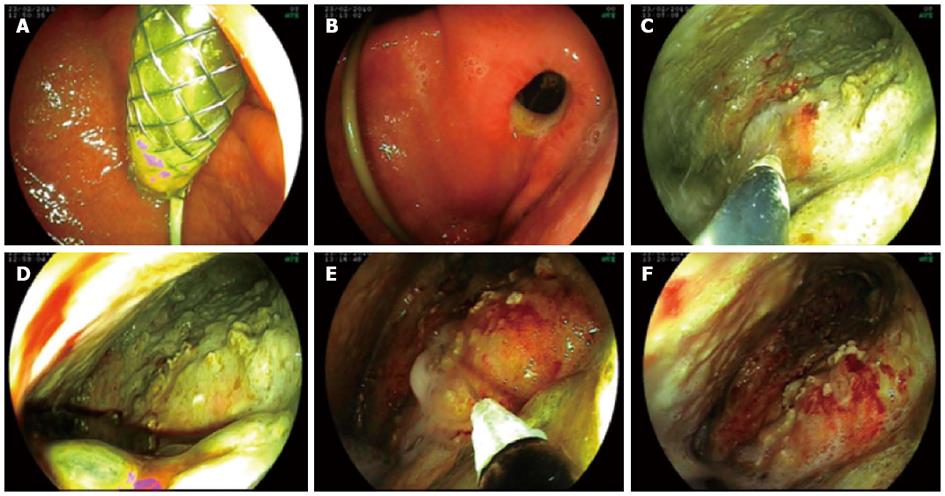
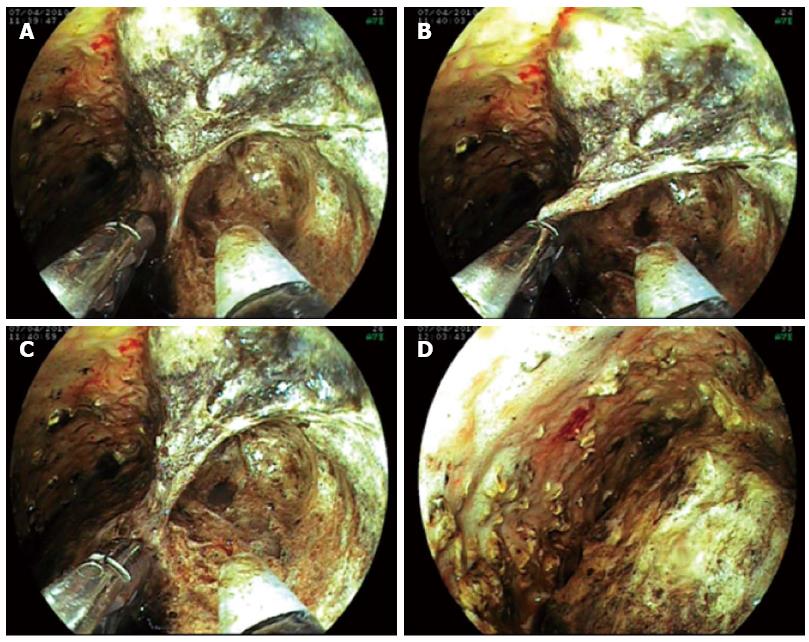
We entered the stomach with a double-channel therapeutic gastroscope (Fujinon EG-530D), and removed the SEMS from the necrotic cavity without any difficulty with standard foreign body forceps 5-7 d later. ETN was performed in the following steps: (1) the therapeutic gastroscope (Fujinon EG-530D) was inserted directly into the cavity; (2) with the application of a flush knife (Fujinon Flush Knife), the purulent debris was flushed from the inner cavity wall, using the high-flow water-jet system (Fujinon JW-2 Water Pump). For high-flow irrigation, we used Betadine solution (1:10 dilution in saline); continuous suction was achieved through the wide-channel therapeutic endoscope; (3) the necrotic remnant was removed by simultaneous application of the flush knife, a Dormia basket, and hot biopsy forceps; and (4) finally, placement of a 12 F nasobiliary catheter into the necrotic cavity and a suction drain into the stomach provided continuous lavage between the endoscopic sessions.
ETN and lavage were performed daily until complete evacuation of the necrotic and purulent material (Figures 1-3).
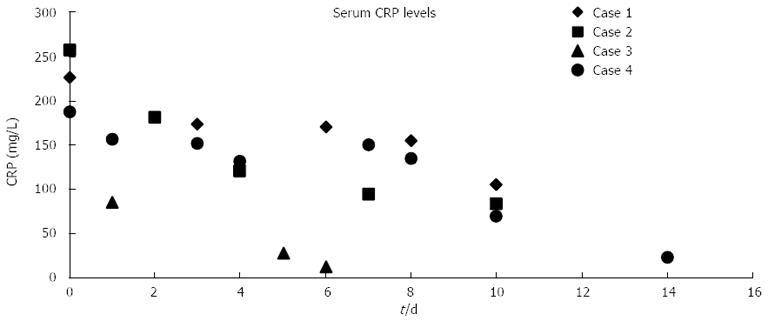
The puncture of the necrotic cavity and placement of the SEMS into the fistulous tract, together with nasopancreatic drainage, was successful in all patients. Access to the cavity was created by an endoscopic puncture into a clearly bulging lesion under endosonographic guidance in 3 cases, and by using a spontaneous perforation of the cyst to the gastrointestinal lumen in Case 4. Transgastric (Cases 1-3) or transduodenal access (Case 4) was chosen in 3 and 1 cases, respectively. After 5-7 days of lavage, the SEMSs were all safely removed. No SEMS-related complications were observed. After SEMS removal, the fistulous tract was sufficiently widely open (without any need for further balloon dilation) to be entered repeatedly with a double working channel therapeutic gastroscope; this allowed permanent access for transgastric necrosectomy during the next 2-3 wk. The average number of daily necrosectomies was 2.6 (range, 2-5). No endotherapy-related complications were observed. ERCP was performed in two patients. After endoscopic management, the Sequential Organ Failure Assessment score and the sepsis improved, the decreased level of consciousness ceased, CRP levels significantly decreased (Figure 4), and a CT scan showed no further evolution of peripancreatic fluid, necrosis, or abscess. All patients, except one (Case 2), were uneventfully and completely cured after endoscopic therapy and medical treatment. Following 6 wk of further basic supportive treatment and jejunal nutrition, they could leave the hospital. The average duration of hospitalization was 63 d (range 48-74 d). The artificial transluminal fistula spontaneously closed within 3 mo.
After a temporary improvement, one patient (Case 2) died 20 d after the first endoscopic intervention, on day 38 of ICU treatment. The autopsy revealed multifocal pulmonary abscesses and severe bilateral pneumonia (an obvious complication of prolonged ventilation), but no retroperitoneal abscess and only moderate inflammatory signs at the site of ETN.
Despite receiving maximum ICU therapy, patients with ANP can succumb rapidly. In terms of the clinical disease course, severe AP is biphasic. During the first week after the onset, there is an early mortality peak, which is known to be mainly due to the systemic inflammatory response syndrome and early multiorgan failure evoked by the exaggerated pro-inflammatory response[13,14]. In contrast, 2-3 wk after the onset of the symptoms, at the time of the maximum anti-inflammatory response and consequent immunosuppression, there is a second peak of mortality, which is known to be mainly due to the infectious complications caused by bacterial translocation, followed by sepsis and late multiorgan failure[15,16]. At this point, even maximum ICU therapy may not be able to halt or reverse the disease progression in some patients.
The standard treatment for infected and complicated WOPN is surgical intervention with open necrosectomy and drainage[17,18]. If it is considered that ANP is responsible for the multiple organ dysfunction syndrome, conventional early surgery (with its high complication rates) is the possible second hit, which may be the explanation, at least in part, for the high mortality. This concept is compounded by the fact that minimally invasive approaches, including minimally invasive retroperitoneal surgical necrosectomy[19], percutaneous catheter drainage[20], and endoscopic transluminal drainage (ETD)[3] ETN[6-12,21], are thought to induce less physiological stress and result in less activation of the inflammatory processes than equivalent open surgery, and may therefore improve the overall outcome of patients with infected PN.
Transgastric endoscopic stent placement, mainly under endoscopic ultrasonographic guidance, is helpful for the drainage of abscesses, but its success in cases of infected WOPN is rather limited. In 2000, Seifert et al[6] first described transgastric access with a standard endoscope into the retroperitoneal space for necrosectomy and debridement. The comparison of ETN with ETD by Gardner et al[7] indicated that ETN achieves higher rates of resolution relative to ETD, without concomitant changes in the number of endoscopic procedures, the complication rate, or the time to resolution. To date, numerous workgroups have reported success and convincing results, including the low morbidity and mortality of ETN in the treatment of infected WOPN in case series[8,9,22]. Moreover, two large, multicenter retrospective studies from Germany and the United States have demonstrated that transmural minimally-invasive endoscopic treatment of WOPN is an efficacious and reproducible technique with an acceptable safety profile[11,12]. However, all studies involving transluminal pancreatic necrosectomies are challenged by the limitations in the efficacy of the endoscopic devices and accessories used for debridement (e.g., Dormia baskets and polypectomy snares). Additional novel methods have recently been described that may facilitate debridement during ETN and improve the overall outcome. Belle et al[23] showed that, through the temporary use of a special partially-covered SEMS designed to keep the pancreaticogastrostomy open for the drainage of WOPN, clinical success (defined as the complete removal of necrotic masses without major complications) was achieved in all 4 of the study’s patients. Furthermore, Antillon et al[24] reported one case where placement of a large-diameter removable metallic esophageal stent into the necrotic cavity, in conjunction with intensive lavage facilitated drainage, was effective; when previous multiple sessions of ETN and drainage with plastic stents had failed.
We have presented here a new and highly efficient method of ETN as a combination of the temporary placement of a non-covered biliary SEMS into the fistulous tract and daily irrigations of the pancreatic necrotic cavity during direct necrosectomies with a high-flow water-jet system, using a flush knife.
The temporary placement of the SEMS resulted in a sustained, open fistulous tract with a wide orifice between the stomach and pancreatic necrotic cavity, through which the ETN could easily be traced and re-performed (even 2 wk after removal) without any additional plastic stent insertion. The relatively short duration in place of the SEMS may prevent stent-related potential complications such as migration, stent-end rubbing-associated cavities, or stomach wall erosions, bleeding or perforation. The necrotic tissue, including large particles, and the purulent material could be removed and/or emptied to the stomach without any complication by washing the cavity with the high-flow water-jet system, using the flush knife, and providing continuous suction via the double-channel therapeutic gastroscope. Only for the removal of adhesive necrotic remnants did we use other accessories (a Dormia basket and hot biopsy forceps) to facilitate the debridement. During this effective, but from a surgical aspect relatively non-aggressive approach, we experienced no procedure-related complications.
Earlier, when we have performed ETN, several hours after each direct endoscopic debridement session we observed a temporary temperature increase in the patients (data not shown). It is speculated that this may be associated with the extreme endotoxin challenge resultant from the debris removed from the necrotic cavity to the gastrointestinal tract, and the impaired gastrointestinal barrier function. We hypothesize that continuous lavage performed constantly together with suction may prevent this event.
We also included a patient with a PA (Case 4) which consisted mainly of solid particles and we had therefore decided to perform direct ETN instead of drainage alone.
We believe that this new combination of methods may lead to clinical success as a consequence of its efficacy with fewer complications and an improvement in patient comfort. However, in order to confirm and optimize this endotherapy, further studies are necessary, preferably on a larger patient population.
The endoscopic transluminal management of PN and/or PA is associated with good initial and long-term clinical success, with acceptable morbidity and mortality rates. The advantages of endoscopic management are related to its minimal invasiveness. The combination of multiple endoscopic approaches is designed to achieve the goals of any treatment strategy.
P- Reviewer Han TQ S- Editor Gou SX L- Editor Rutherford A E- Editor Zhang DN
| 1. | Bradley EL. A clinically based classification system for acute pancreatitis. Summary of the International Symposium on Acute Pancreatitis, Atlanta, Ga, September 11 through 13, 1992. Arch Surg. 1993;128:586-590. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1929] [Cited by in F6Publishing: 1716] [Article Influence: 55.4] [Reference Citation Analysis (0)] |
| 2. | Forsmark CE, Baillie J. AGA Institute technical review on acute pancreatitis. Gastroenterology. 2007;132:2022-2044. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 494] [Cited by in F6Publishing: 480] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 3. | Baron TH, Thaggard WG, Morgan DE, Stanley RJ. Endoscopic therapy for organized pancreatic necrosis. Gastroenterology. 1996;111:755-764. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 316] [Cited by in F6Publishing: 276] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 4. | Connor S, Alexakis N, Raraty MG, Ghaneh P, Evans J, Hughes M, Garvey CJ, Sutton R, Neoptolemos JP. Early and late complications after pancreatic necrosectomy. Surgery. 2005;137:499-505. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 219] [Cited by in F6Publishing: 183] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 5. | Besselink MG, Verwer TJ, Schoenmaeckers EJ, Buskens E, Ridwan BU, Visser MR, Nieuwenhuijs VB, Gooszen HG. Timing of surgical intervention in necrotizing pancreatitis. Arch Surg. 2007;142:1194-1201. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 245] [Cited by in F6Publishing: 219] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 6. | Seifert H, Wehrmann T, Schmitt T, Zeuzem S, Caspary WF. Retroperitoneal endoscopic debridement for infected peripancreatic necrosis. Lancet. 2000;356:653-655. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 250] [Cited by in F6Publishing: 259] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 7. | Gardner TB, Chahal P, Papachristou GI, Vege SS, Petersen BT, Gostout CJ, Topazian MD, Takahashi N, Sarr MG, Baron TH. A comparison of direct endoscopic necrosectomy with transmural endoscopic drainage for the treatment of walled-off pancreatic necrosis. Gastrointest Endosc. 2009;69:1085-1094. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 164] [Cited by in F6Publishing: 174] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 8. | Seewald S, Groth S, Omar S, Imazu H, Seitz U, de Weerth A, Soetikno R, Zhong Y, Sriram PV, Ponnudurai R. Aggressive endoscopic therapy for pancreatic necrosis and pancreatic abscess: a new safe and effective treatment algorithm (videos). Gastrointest Endosc. 2005;62:92-100. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 238] [Cited by in F6Publishing: 249] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 9. | Voermans RP, Veldkamp MC, Rauws EA, Bruno MJ, Fockens P. Endoscopic transmural debridement of symptomatic organized pancreatic necrosis (with videos). Gastrointest Endosc. 2007;66:909-916. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 110] [Cited by in F6Publishing: 114] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 10. | Raczynski S, Teich N, Borte G, Wittenburg H, Mössner J, Caca K. Percutaneous transgastric irrigation drainage in combination with endoscopic necrosectomy in necrotizing pancreatitis (with videos). Gastrointest Endosc. 2006;64:420-424. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 44] [Cited by in F6Publishing: 47] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 11. | Seifert H, Biermer M, Schmitt W, Jürgensen C, Will U, Gerlach R, Kreitmair C, Meining A, Wehrmann T, Rösch T. Transluminal endoscopic necrosectomy after acute pancreatitis: a multicentre study with long-term follow-up (the GEPARD Study). Gut. 2009;58:1260-1266. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 333] [Cited by in F6Publishing: 288] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 12. | Gardner TB, Coelho-Prabhu N, Gordon SR, Gelrud A, Maple JT, Papachristou GI, Freeman ML, Topazian MD, Attam R, Mackenzie TA. Direct endoscopic necrosectomy for the treatment of walled-off pancreatic necrosis: results from a multicenter U.S. series. Gastrointest Endosc. 2011;73:718-726. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 218] [Cited by in F6Publishing: 195] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 13. | Bhatia M, Brady M, Shokuhi S, Christmas S, Neoptolemos JP, Slavin J. Inflammatory mediators in acute pancreatitis. J Pathol. 2000;190:117-125. [PubMed] [Cited in This Article: ] |
| 14. | Mofidi R, Duff MD, Wigmore SJ, Madhavan KK, Garden OJ, Parks RW. Association between early systemic inflammatory response, severity of multiorgan dysfunction and death in acute pancreatitis. Br J Surg. 2006;93:738-744. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 363] [Cited by in F6Publishing: 336] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 15. | Ammori BJ, Leeder PC, King RF, Barclay GR, Martin IG, Larvin M, McMahon MJ. Early increase in intestinal permeability in patients with severe acute pancreatitis: correlation with endotoxemia, organ failure, and mortality. J Gastrointest Surg. 1999;3:252-262. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 10] [Reference Citation Analysis (0)] |
| 16. | Deitch EA. The role of intestinal barrier failure and bacterial translocation in the development of systemic infection and multiple organ failure. Arch Surg. 1990;125:403-404. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 354] [Cited by in F6Publishing: 320] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 17. | Beger HG, Büchler M, Bittner R, Oettinger W, Block S, Nevalainen T. Necrosectomy and postoperative local lavage in patients with necrotizing pancreatitis: results of a prospective clinical trial. World J Surg. 1988;12:255-262. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 85] [Cited by in F6Publishing: 90] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 18. | Traverso LW, Kozarek RA. Pancreatic necrosectomy: definitions and technique. J Gastrointest Surg. 2005;9:436-439. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 69] [Cited by in F6Publishing: 73] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 19. | van Santvoort HC, Besselink MG, Bakker OJ, Hofker HS, Boermeester MA, Dejong CH, van Goor H, Schaapherder AF, van Eijck CH, Bollen TL. A step-up approach or open necrosectomy for necrotizing pancreatitis. N Engl J Med. 2010;362:1491-1502. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1038] [Cited by in F6Publishing: 924] [Article Influence: 66.0] [Reference Citation Analysis (0)] |
| 20. | Freeny PC, Hauptmann E, Althaus SJ, Traverso LW, Sinanan M. Percutaneous CT-guided catheter drainage of infected acute necrotizing pancreatitis: techniques and results. AJR Am J Roentgenol. 1998;170:969-975. [PubMed] [Cited in This Article: ] |
| 21. | Papachristou GI, Takahashi N, Chahal P, Sarr MG, Baron TH. Peroral endoscopic drainage/debridement of walled-off pancreatic necrosis. Ann Surg. 2007;245:943-951. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 205] [Cited by in F6Publishing: 190] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 22. | Charnley RM, Lochan R, Gray H, O’Sullivan CB, Scott J, Oppong KE. Endoscopic necrosectomy as primary therapy in the management of infected pancreatic necrosis. Endoscopy. 2006;38:925-928. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 137] [Cited by in F6Publishing: 147] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 23. | Belle S, Collet P, Post S, Kaehler G. Temporary cystogastrostomy with self-expanding metallic stents for pancreatic necrosis. Endoscopy. 2010;42:493-495. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 63] [Cited by in F6Publishing: 72] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 24. | Antillon MR, Bechtold ML, Bartalos CR, Marshall JB. Transgastric endoscopic necrosectomy with temporary metallic esophageal stent placement for the treatment of infected pancreatic necrosis (with video). Gastrointest Endosc. 2009;69:178-180. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 52] [Article Influence: 3.5] [Reference Citation Analysis (0)] |