Published online Dec 14, 2011. doi: 10.3748/wjg.v17.i46.5105
Revised: March 28, 2011
Accepted: April 18, 2011
Published online: December 14, 2011
AIM: To investigate the in vitro effects of pantoprazole on rat lower esophageal sphincter (LES) tone.
METHODS: Rats weighing 250-300 g, provided by the Yeditepe University Experimental Research Center (YÜDETAM), were used throughout the study. They were anesthetized before decapitation. LES tissues whose mucosal lining were removed were placed in a standard 30-mL organ bath with a modified Krebs solution and continuously aerated with 95% oxygen-5% carbon dioxide gas mixture and kept at room temperature. The tissues were allowed to stabilize for 60 min. Subsequently, the contractile response to 10-6 mol/L carbachol was obtained. Different concentrations of freshly prepared pantoprazole were added directly to the tissue bath to generate cumulative concentrations of 5 × 10-6 mol/L, 5 × 10-5 mol/L, and 1.5 × 10-4 mol/L. Activities were recorded on an online computer via a 4-channel transducer data acquisition system using the software BSL PRO v 3.7, which also analyzed the data.
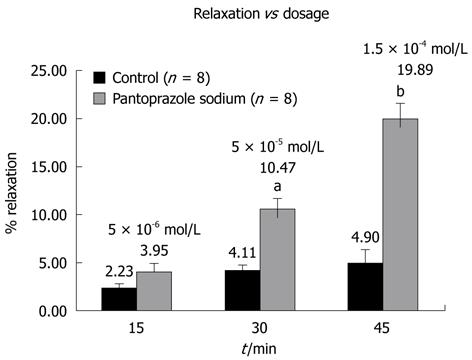
RESULTS: Pantoprazole at 5 × 10-6 mol/L caused a small, but statistically insignificant, relaxation in the carbachol-contracted LES (2.23% vs 3.95%). The 5 × 10-5 mol/L concentration, however, caused a significant relaxation of 10.47% compared with the control. 1.5 × 10-4 mol/L concentration of pantoprazol caused a 19.89% relaxation in the carbachol contracted LES (P < 0.001).
CONCLUSION: This is the first study to demonstrate that pantoprazole has a relaxing effect in isolated LESs. These results might have significant clinical implications for the subset of patients using proton pump inhibitors who do not receive full symptomatic alleviation from gastroesophageal reflux disease.
-
Citation: Duman M, Özer M, Reyhan E, Demirci Y, Atıcı AE, Dalgıç T, Bostancı EB, Genç E.
In vitro effect of pantoprazole on lower esophageal sphincter tone in rats. World J Gastroenterol 2011; 17(46): 5105-5109 - URL: https://www.wjgnet.com/1007-9327/full/v17/i46/5105.htm
- DOI: https://dx.doi.org/10.3748/wjg.v17.i46.5105
The esophagogastric junction is located between the esophagus and the stomach. The high-pressure zone at the junction between the esophagus and the stomach is composed of the lower esophageal sphincter (LES) and the crural diaphragm[1,2]. Circular smooth muscle from the esophageal body generates little if any tone at rest, whereas the circular smooth muscle of LES is characterized by a spontaneously generated basal tone that prevents the reflux of gastric contents into the esophagus[3,4]. The basal tone of the LES is primarily myogenic in origin, but can be modulated by both neural and hormonal factors[5]. In response to esophageal distension and swallowing, the LES relaxes[6]. The abnormal dynamics of LES function are considered to be the most important factors in the pathogenesis of gastroesophageal reflux disease (GERD)[7-10]. GERD is described as the reflux of gastric contents into the esophagus leading to reflux symptoms and esophagitis sufficient to affect patient wellbeing and/or induce complications. These complications range from esophagitis to adenocarcinoma of the distal esophagus. Furthermore it may cause extra esophageal symptoms, such as cough, laryngitis and asthma[11,12]. GERD is a highly prevalent in the general population, affecting up to 10%-30% of the adult population in western countries[13].
Pharmacological therapy is necessary in the majority of patients. GERD is currently treated with acid suppressing drugs, such as proton pump inhibitors (PPIs); however, for those refractory to pharmacological treatment, surgery is often recommended[13,14]. PPIs are the mainstay of medical management for GERD[11]. They have been widely used since the 1980s and have been considered as ideal drugs because of their highly specific pharmacologic actions[15,16]. Although PPIs have been used as a common treatment modality in GERD, there is a lack of experimental studies of their effects on isolated LES preparations.
The aim of this study was to investigate the effect of a PPI, pantoprazole, on the tone of the isolated rat LES preparations contracted by carbachol. This study provides a significant contribution to this somewhat ignored area of research.
The experimental protocol was approved by the Ethical Committee of Yeditepe University Experimental Medicine Research Institute and the use of animals was in compliance with US National Institutes of Health Guide for Care and Use of Laboratory Animals.
Sixteen rats weighing 250-300 g, provided by the Yeditepe University Experimental Research Center (YÜDETAM), were used throughout the study. They were kept in plexiglass cages in a room whose temperature and humidity were controlled with 12-h light/dark cycle, and had free excess to food and water.
Rats were anesthetized with a combination of 10 mg/kg xylazine HCl (Rompun® 2%, Bayer HealthCare AG, Leverkusen-Germany) and 100 mg/kg ketamine HCl (Ketasol® 10%, Richter Pharma AG, Weis-Austria) before decapitation.
A midline incision was performed to open up the abdominal cavity and the LES was carefully dissected out and placed in a petri dish containing Krebs solution at room temperature. Thereafter, the mucosal lining was removed and the sphincteric muscle was set up, as a ring segment 2 mm in width, in Krebs solution contained in a standard 30-mL organ bath. The modified Krebs solution comprised NaCl, 118.07 mmol/L; KCl, 4.69 mmol/L; CaCl2, 2.52 mmol/L; MgSO4, 1.16 mmol/L; KH2PO4, 1.2 mmol/L; NaHCO3, 25 mmol/L, and glucose, 11.10 mmol/L. Krebs solution was continuously aerated with 95% oxygen-5% carbon dioxide gas mixture and kept at 37 ± 0.5 °C throughout the experimental period. The tissues were tied to stainless steel hooks at one end of the organ bath; the other end was connected to a force transducer (FDT 05, May, COMMAT Iletisim Co, Ankara-Turkey) under a resting tension of around 1 g. LES ring activities were recorded on an online computer via a 4-channel transducer data acquisition system (MP35, BIOPAC Systems Inc. Goleta, CA, United States) using the software BSL PRO v 3.7 (BIOPAC Systems Inc. Goleta, CA, United States), which also analyzed the data.
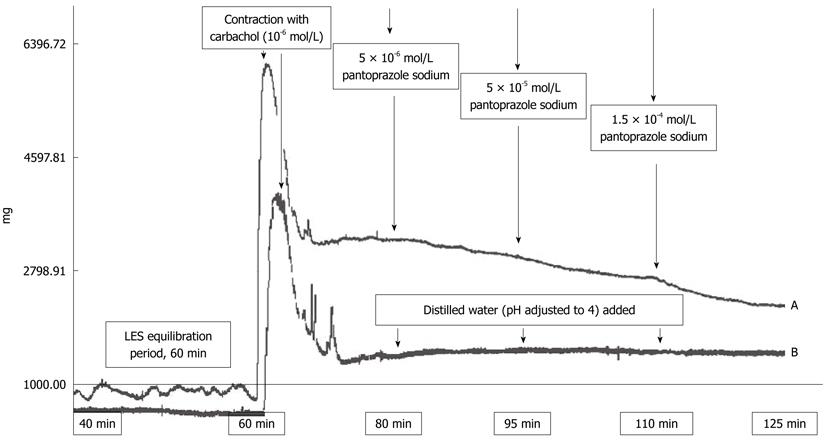
The following compounds were used: carbachol chloride (Carbamylcholine chloride, Sigma-Aldrich Chemical Co. St. Louis, MO, United States) and pantoprazole (Pantoprazole sodium, Dr. Reddy’s Laboratories Ltd. Hyderabad-India). Solutions were prepared daily in distilled water and kept at 4 °C during the experiments. Pantoprazole was treated with 1 mol/L HCl and its pH was adjusted to 4.0 before application to the organ bath. Following a 60-min equilibration period for stabilization, the contractile response to carbachol was obtained by application of a single dose of charbachol to a final concentration of 10-6 mol/L in the organ bath. After the contractions reached a plateau, concentration-response relationships for pantoprazole (final organ bath concentrations of 5 × 10-6 mol/L, 5 × 10-5 mol/L and 1.5 × 10-4 mol/L, with 15 min allotted between each dose) were obtained in a cumulative manner. (These doses were calculated to be the equivalent of Human doses for the rats). Control experiments were also run with only acidified distilled water added to the organ bath. The relaxations were quantified by integrating the area under the curve for each concentration and control group. At the end of the each experiment, tissues were weighed and the final pH of the Krebs solution was measured.
For statistical evaluation, analysis of variance (One way ANOVA) was performed with the program SPSS for windows version 18 (SPSS Inc. Chicago, Illinois). Values of P < 0.05 were considered as statistically significant.
The experiment design is outlined in Figure 1. Pantoprazole caused dose dependent relaxation of the carbachol-contracted LES preparations. No such effect was observed in the control group (Figure 1B). The relaxations were quantified by integrating the area under the curve for each concentration.
The mean of integral values and percent relaxations of eight preparations were compared for statistical evaluation. As shown in Figure 2, application of pantoprazole sodium in a cumulative manner resulted in significant relaxations of LES preparations at 5 × 10-5 mol/L and 1.5 × 10-4 mol/L concentrations.
In the carbachol-contracted LES preparations 5 × 10-6 mol/L pantoprazole caused a 4% relaxation, while higher doses caused significant relaxations. Mean integral relaxation values were 4.11% ± 0.58% (SE) and 10.47% ± 1.2% (SE) for control and 5 × 10-5 mol/L pantoprazole, respectively (P < 0.05). Moreover, these values were 4.90% ± 1.4% (SE) and 19.89% ± 1.7% (SE) for control and 1.5 × 10-4 mol/L concentrations, respectively (P < 0.001) (Figure 2).
The aim of the present work was to assess the in vitro effects of pantoprazole on LES tone in rats. The reason why pantoprazole was chosen was the drug’s frequent use in our Clinic. The major finding of our study was that pantoprazole caused a dose-dependent decrease in LES tone. This is the first study to demonstrate that pantoprazole has such an effect on isolated LES.
LES is an important specialized smooth muscle in the gastrointestinal tract and has been the subject of investigation by many authors[17-20]. GERD is a highly prevalent condition and is a major burden to society as well as the afflicted individual. Although numerous clinical studies have been conducted to clarify the mechanism of GERD, a clear consensus has not been reached. Regarding the pathophysiologies of GERD, decrease of LES basal tone and transient relaxations of the LES (TLOSRs) as a response to gastric distension[21], and excessive exposure of the esophagus to gastric acid, have been reported to be important[21-24].
GERD is, in most cases, successfully treated with PPIs, which have largely replaced Histamine H2 receptor blockers because of their well documented efficacy and because they are well tolerated, with relatively few serious adverse effects. However, a significant number of patients do not receive full symptomatic relief[25,26]. Thus, a significant question that has to be addressed is why some GERD patients are resistant to the effects of PPIs? In addition to neonates and infants who respond poorly to PPIs[27], some adults do not benefit from them either. In a study conducted by Hemmink et al[28] in 2008, there were fewer acid reflux episodes in patients on PPI therapy; however, weak acidic reflux episodes increased under the influence of PPIs. The total number of reflux episodes, on the other hand, was not affected. In addition to these, there have been recent papers regarding the adverse effects of PPIs[29,30]. Corley et al[31] showed that PPIs are associated with hip fractures among at-risk patients. They can also cause neutropenia in some patients[32]. Acid suppression also causes nosocomial Clostridium difficile infections in a dose-dependent manner[33].
These results point out the necessity of developing novel approaches for GERD. Coman et al[34] demonstrated the significance of adding prokinetic drugs to the treatment of GERD, in a study conducted on 1118 patients. The effects of specific GABA B receptor agonists have also been studied[35]. Drugs that reduce TLOSRs have also been suggested as pharmacological agents for GERD[36].
At present, the mechanism of the pantoprazole-induced relaxation of LESs can only be speculated. However, there are 2 types of muscles in the LES, circular muscle and sling muscle. Circular smooth muscle is tonically contracted with cholinergic stimulation. In response to swallowing, a peristaltic contraction travels down the length of the esophagus and the LES relaxes.
Nitric oxide (NO)[37,38] and vasoactive intestinal polypeptide (VIP)[39,40] are proposed as neurotransmitters that control relaxation. Both VIP and NO can be released from esophageal nerves with an appropriate stimulus, and NO synthase and VIP are found in myenteric neurons that innervate the circular smooth muscle of the esophagus. Sarioglu et al[41], showed the relaxant effect of omeprazole in rabbit corpus cavernosum in vitro. They concluded that the relaxant effect is probably due to the L-type Ca2+ channel blockage by omeprazole. We can speculate that a similar mechanism is responsible for the effect of pantoprazole on LESs.
The present study is the first to demonstrate a dose-dependent decrease in the carbachol-induced contraction of the LES by pantoprazole. Although this finding has been observed in an isolated tissue, it might have some clinical correlates and might help to understand why the treatment of GERD requires additional pharmacological interventions.
The present study was supported by Yeditepe University. The authors are indebted to Ahmet Ayar Dr. Professor for his valuable methodological advice.
Gastroesophageal reflux disease (GERD) is a highly prevalent condition in the general population, affecting up to 10%-30% of the adult population in Western countries[13]. The incidence of GERD is rising very rapidly due to the stressful lives. New approaches are necessary for its treatment.
Not all patients benefit from the proton pump inhibitors (PPIs) that are frequently used for the treatment of GERD. The authors conducted an experiment to investigate the effects of these drugs on isolated rat lower esophageal sphincters (LESs). There was a dose dependent decrease in LES tone.
The study conducted is the first to demonstrate the effects of pantoprazole on the isolated LESs of rat, including the dose dependent decrease in the tone of LESs under the effect of the drug.
The study suggests that doctors should be cautious about long-term use of PPIs for the treatment of GERD.
This paper should be of interest to a broad readership including gastroenterologists, pharmacologists, and physicians of internal medicine. It is also of interest to gastrointestinal surgeons. This paper is very interesting and is an important study to publish.
Peer reviewer: Lygia Stewart, MD, Professor of Clinical Surgery, University of California San Francisco, 4150 Clement Street, San Francisco, CA 94121, United States
S- Editor Tian L L- Editor Stewart GJ E- Editor Zheng XM
| 1. | Farré R, Sifrim D. Regulation of basal tone, relaxation and contraction of the lower oesophageal sphincter. Relevance to drug discovery for oesophageal disorders. Br J Pharmacol. 2008;153:858-869. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 44] [Cited by in F6Publishing: 47] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 2. | Cuomo R, Grasso R, Sarnelli G, Bruzzese D, Bottiglieri ME, Alfieri M, Sifrim D, Budillon G. Role of diaphragmatic crura and lower esophageal sphincter in gastroesophageal reflux disease: manometric and pH-metric study of small hiatal hernia. Dig Dis Sci. 2001;46:2687-2694. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 72] [Reference Citation Analysis (1)] |
| 3. | Zhang Y, Paterson WG. Role of sarcoplasmic reticulum in control of membrane potential and nitrergic response in opossum lower esophageal sphincter. Br J Pharmacol. 2003;140:1097-1107. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 25] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 4. | Pandolfino JE, Shi G, Curry J, Joehl RJ, Brasseur JG, Kahrilas PJ. Esophagogastric junction distensibility: a factor contributing to sphincter incompetence. Am J Physiol Gastrointest Liver Physiol. 2002;282:G1052-G1058. [PubMed] [Cited in This Article: ] |
| 5. | Zhang Y, Miller DV, Paterson WG. Opposing roles of K(+) and Cl(-) channels in maintenance of opossum lower esophageal sphincter tone. Am J Physiol Gastrointest Liver Physiol. 2000;279:G1226-G1234. [PubMed] [Cited in This Article: ] |
| 6. | Dogan I, Bhargava V, Liu J, Mittal RK. Axial stretch: A novel mechanism of the lower esophageal sphincter relaxation. Am J Physiol Gastrointest Liver Physiol. 2007;292:G329-G334. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 36] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 7. | Sanmiguel CP, Hagiike M, Mintchev MP, Cruz RD, Phillips EH, Cunneen SA, Conklin JL, Soffer EE. Effect of electrical stimulation of the LES on LES pressure in a canine model. Am J Physiol Gastrointest Liver Physiol. 2008;295:G389-G394. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 32] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 8. | Beaumont H, Jensen J, Carlsson A, Ruth M, Lehmann A, Boeckxstaens G. Effect of delta9-tetrahydrocannabinol, a cannabinoid receptor agonist, on the triggering of transient lower oesophageal sphincter relaxations in dogs and humans. Br J Pharmacol. 2009;156:153-162. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 57] [Cited by in F6Publishing: 61] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 9. | Staunton E, Smid SD, Dent J, Blackshaw LA. Triggering of transient LES relaxations in ferrets: role of sympathetic pathways and effects of baclofen. Am J Physiol Gastrointest Liver Physiol. 2000;279:G157-G162. [PubMed] [Cited in This Article: ] |
| 10. | McMahon BP, Drewes AM, Gregersen H. Functional oesophago-gastric junction imaging. World J Gastroenterol. 2006;12:2818-2824. [PubMed] [Cited in This Article: ] |
| 11. | Kalaitzakis E, Björnsson E. A review of esomeprazole in the treatment of gastroesophageal reflux disease (GERD). Ther Clin Risk Manag. 2007;3:653-663. [PubMed] [Cited in This Article: ] |
| 12. | Vakil N. The prevention of gastropathy and upper abdominal symptoms caused by nonsteroidal anti-inflammatory drugs. Rev Gastroenterol Disord. 2006;6:221-226. [PubMed] [Cited in This Article: ] |
| 13. | Warrington S, Baisley K, Lee D, Lomax K, Delemos B, Boyce M, Morocutti A. Pharmacodynamic effects of single doses of rabeprazole 20 mg and pantoprazole 40 mg in patients with GERD and nocturnal heartburn. Aliment Pharmacol Ther. 2007;25:511-517. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 14. | Scholten T. Long-term management of gastroesophageal reflux disease with pantoprazole. Ther Clin Risk Manag. 2007;3:231-243. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 15. | Calabrese C, Fabbri A, Di Febo G. Long-term management of GERD in the elderly with pantoprazole. Clin Interv Aging. 2007;2:85-92. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 12] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 16. | Tamhankar AP, Peters JH, Portale G, Hsieh CC, Hagen JA, Bremner CG, DeMeester TR. Omeprazole does not reduce gastroesophageal reflux: new insights using multichannel intraluminal impedance technology. J Gastrointest Surg. 2004;8:890-87; discussion 890-87;. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 118] [Cited by in F6Publishing: 108] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 17. | Kohjitani A, Miyawaki T, Funahashi M, Higuchi H, Matsuo R, Shimada M. Ketamine and midazolam differentially inhibit nonadrenergic noncholinergic lower esophageal sphincter relaxation in rabbits: role of superoxide anion and nitric oxide synthase. Anesthesiology. 2003;98:449-458. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 15] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 18. | Zhang Q, Horowitz M, Rigda R, Rayner C, Worynski A, Holloway RH. Effect of hyperglycemia on triggering of transient lower esophageal sphincter relaxations. Am J Physiol Gastrointest Liver Physiol. 2004;286:G797-G803. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 21] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 19. | Yildiz F, Tugay M, Utkan T, Yazir Y. Effect of chronic renal failure on foregut smooth muscle reactivity: an experimental study. J Pediatr Surg. 2007;42:647-652. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1] [Cited by in F6Publishing: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 20. | Kim N, Cao W, Song IS, Kim CY, Sohn UD, Harnett KM, Biancani P. Leukotriene D4-induced contraction of cat esophageal and lower esophageal sphincter circular smooth muscle. Gastroenterology. 1998;115:919-928. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 22] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 21. | Dent J. Pathogenesis of gastro-oesophageal reflux disease and novel options for its therapy. Neurogastroenterol Motil. 2008;20 Suppl 1:91-102. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 35] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 22. | Bredenoord AJ. Impedance-pH monitoring: new standard for measuring gastro-oesophageal reflux. Neurogastroenterol Motil. 2008;20:434-439. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 35] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 23. | Ozin Y, Dagli U, Kuran S, Sahin B. Manometric findings in patients with isolated distal gastroesophageal reflux. World J Gastroenterol. 2009;15:5461-5464. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 6] [Cited by in F6Publishing: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 24. | Young RL, Page AJ, Cooper NJ, Frisby CL, Blackshaw LA. Sensory and motor innervation of the crural diaphragm by the vagus nerves. Gastroenterology. 2010;138:1091-1101. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 43] [Cited by in F6Publishing: 48] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 25. | Klinkenberg-Knol EC, Meuwissen SG. Combined gastric and oesophageal 24-hour pH monitoring and oesophageal manometry in patients with reflux disease, resistant to treatment with omeprazole. Aliment Pharmacol Ther. 1990;4:485-495. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 56] [Cited by in F6Publishing: 56] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 26. | Vakil N. Treatment of gastroesophageal reflux disease: defining endpoints that are important to patients. Rev Gastroenterol Disord. 2004;4 Suppl 4:S3-S7. [PubMed] [Cited in This Article: ] |
| 27. | Orenstein SR, Hassall E. Pantoprazole for symptoms of infant GERD: the emperor has no clothes! J Pediatr Gastroenterol Nutr. 2010;51:537; author reply 537-539. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5] [Cited by in F6Publishing: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 28. | Hemmink GJ, Bredenoord AJ, Weusten BL, Monkelbaan JF, Timmer R, Smout AJ. Esophageal pH-impedance monitoring in patients with therapy-resistant reflux symptoms: 'on' or 'off' proton pump inhibitor? Am J Gastroenterol. 2008;103:2446-2453. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 156] [Cited by in F6Publishing: 147] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 29. | Nealis TB, Howden CW. Is there a dark side to long-term proton pump inhibitor therapy? Am J Ther. 2008;15:536-542. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 30] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 30. | Thomson AB, Sauve MD, Kassam N, Kamitakahara H. Safety of the long-term use of proton pump inhibitors. World J Gastroenterol. 2010;16:2323-2330. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 185] [Cited by in F6Publishing: 183] [Article Influence: 13.1] [Reference Citation Analysis (1)] |
| 31. | Corley DA, Kubo A, Zhao W, Quesenberry C. Proton pump inhibitors and histamine-2 receptor antagonists are associated with hip fractures among at-risk patients. Gastroenterology. 2010;139:93-101. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 208] [Cited by in F6Publishing: 213] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 32. | Gouraud A, Vochelle V, Descotes J, Vial T. Proton pump inhibitor-induced neutropenia: possible cross-reactivity between omeprazole and pantoprazole. Clin Drug Investig. 2010;30:559-563. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 22] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 33. | Howell MD, Novack V, Grgurich P, Soulliard D, Novack L, Pencina M, Talmor D. Iatrogenic gastric acid suppression and the risk of nosocomial Clostridium difficile infection. Arch Intern Med. 2010;170:784-790. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 312] [Cited by in F6Publishing: 304] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 34. | Coman AE, Popa E, Grigore C, Maidaniuc M, Petrovanu R. [Causes of functional digestive disorders and therapeutic approach in primary care medicine]. Rev Med Chir Soc Med Nat Iasi. 2010;114:75-79. [PubMed] [Cited in This Article: ] |
| 35. | Lehmann A, Antonsson M, Holmberg AA, Blackshaw LA, Brändén L, Bräuner-Osborne H, Christiansen B, Dent J, Elebring T, Jacobson BM. (R)-(3-amino-2-fluoropropyl) phosphinic acid (AZD3355), a novel GABAB receptor agonist, inhibits transient lower esophageal sphincter relaxation through a peripheral mode of action. J Pharmacol Exp Ther. 2009;331:504-512. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 39] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 36. | Hirsch DP, Tytgat GN, Boeckxstaens GE. Transient lower oesophageal sphincter relaxations--a pharmacological target for gastro-oesophageal reflux disease? Aliment Pharmacol Ther. 2002;16:17-26. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 64] [Cited by in F6Publishing: 65] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 37. | Murray J, Du C, Ledlow A, Bates JN, Conklin JL. Nitric oxide: mediator of nonadrenergic noncholinergic responses of opossum esophageal muscle. Am J Physiol. 1991;261:G401-G406. [PubMed] [Cited in This Article: ] |
| 38. | Tøttrup A, Knudsen MA, Gregersen H. The role of the L-arginine-nitric oxide pathway in relaxation of the opossum lower oesophageal sphincter. Br J Pharmacol. 1991;104:113-116. [PubMed] [Cited in This Article: ] |
| 39. | Behar J, Guenard V, Walsh JH, Biancani P. VIP and acetylcholine: neurotransmitters in esophageal circular smooth muscle. Am J Physiol. 1989;257:G380-G385. [PubMed] [Cited in This Article: ] |
| 40. | Biancani P, Walsh JH, Behar J. Vasoactive intestinal polypeptide. A neurotransmitter for lower esophageal sphincter relaxation. J Clin Invest. 1984;73:963-967. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 114] [Cited by in F6Publishing: 102] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 41. | Sarioglu Y, Yildirim S, Utkan T, Yildirim MK, Uma S. Evidence of relaxant effect of omeprazole in rabbit corpus cavernosum in vitro. Life Sci. 2000;66:1411-1421. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |