Published online Dec 14, 2007. doi: 10.3748/wjg.v13.i46.6277
Revised: August 24, 2007
Accepted: August 31, 2007
Published online: December 14, 2007
There are various well described forms of chronic cholestatic jaundice in adults, such as autoimmune cholangitis, drug-induced cholangitis and intrahepatic cholestasis of pregnancy. We present two cases of prolonged cholestasis following removal of gallstones at endoscopic retrograde cholangiopancreatography (ERCP) and subsequent clear cholangiography. Both patients were taking oral estrogens at the time of presentation, which were subsequently withdrawn. The first case responded rapidly to corticosteroid treatment, and the second case had a much slower resolution with ursodeoxycholic acid. Both cases highlighted the significance of estrogen-induced cholestasis in female patients with protracted jaundice following ERCP and removal of intra-ductal stones. After oral estrogens are discontinued, a short course of steroids needs to be considered.
- Citation: Dunn J, McNair A. Prolonged cholestasis following successful removal of common bile duct stones: Beware patients on estrogen therapy. World J Gastroenterol 2007; 13(46): 6277-6280
- URL: https://www.wjgnet.com/1007-9327/full/v13/i46/6277.htm
- DOI: https://dx.doi.org/10.3748/wjg.v13.i46.6277
Estrogens have been a well-recognized cause of cholestatic jaundice since 1962[1], and are commonly used in an experimental model of hepatocellular cholestasis. The similarity between such drug reactions and the syndrome of intrahepatic cholestasis of pregnancy has been reported[2]. Withdrawal of the estrogenic effect by delivery of the fetus or drug withdrawal leads to improvement of liver function. This tends to occur over several weeks or months[3]. It has been shown in observational studies that estrogen therapy is an important risk factor for gallbladder disease[4]. A recent randomized double-blind placebo-controlled trial of otherwise healthy postmenopausal women has demonstrated that the risk of adverse biliary tract outcomes, such as cholecystitis, is substantially increased by exogenous estrogen therapy[5]. Estrogens are thought to promote gallstone formation by significantly elevating the biliary cholesterol saturation index and a reduction of the nucleation time[6]. We present two cases of prolonged cholestasis after removal of obstructing common bile duct (CBD) stones at endoscopic retrograde cholangiopancreatography (ERCP).
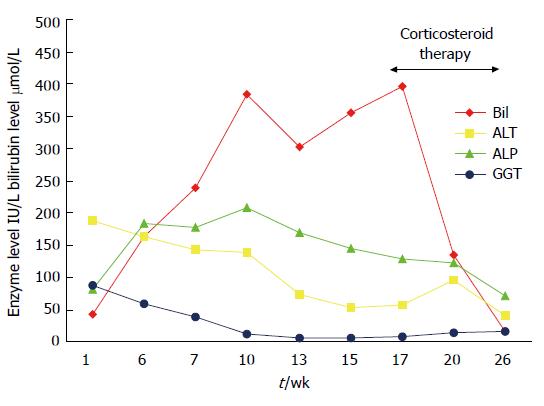
A 25-year-old Caucasian woman presented at 6 wk post partum with pruritus and right upper quadrant pain. There was no jaundice evident, and she had been asymptomatic throughout her pregnancy. She had stopped taking Microgynon (ethinlyestradiol 30 μg and levonorgestrel 150 μg) 1 year previously. Her mother had undergone cholecystectomy aged 50 years. Physical examination was unremarkable, and liver function tests showed a total serum bilirubin level of 43 μmol/L, alanine aminotransferase (ALT) 187 IU/L, alkaline phosphatase (ALP) 81 IU/L and gamma-glutamyltranspeptidase (γ-GT) 88 IU/L (Figure 1). Liver ultrasound scan showed multiple calculi in the gall bladder and a CBD of 6 mm, with no evidence of intraductal stones.
She presented again 6 wk later with worsening pruritus, biliary colic and jaundice. The patient had restarted Microgynon in the interim period. She denied exposure to alcohol or illegal drugs. There were no risk factors for viral hepatitis. Her weight had dropped by 8 kg and she had icterus. Abdominal palpation revealed a tender right upper quadrant but no hepatosplenomegaly. Retesting of liver biochemistry showed a total serum bilirubin level of 163 μmol/L and a twofold increase in serum ALP. ALT remained elevated at 163 IU/L. Abdominal ultrasound scanning showed a thick-walled edematous gallbladder with multiple small calculi, one impacted in the neck of the gallbladder. The CBD measured 3.5 mm.
Endoscopic retrograde cholangiopancreatography (ERCP) was performed and revealed two calculi in the CBD, the largest measuring 5 mm. A 12-mm sphincterotomy was made and the CBD dredged with a balloon. Both stones were removed and an occlusion cholangiogram confirmed clearance of the duct. Her total serum bilirubin level continued to rise over the next 10 wk. A second ERCP performed 3 wk after the first showed a dilated CBD, but no obvious filling defects. The sphincterotomy was extended. A balloon dredge and occlusion cholangiogram confirmed that the duct was clear. Microgynon was stopped at this point. Autoantibody profile and hepatitis A, B and C serology were negative. Ceruloplasmin and ferritin levels were normal. The hemoglobin level was stable and the haptoglobin level normal. Because of persisting jaundice, a third ERCP was performed 3 wk later and revealed a normal condition.
Seventeen weeks following the initial presentation (7 wk after stopping the oral contraceptive pill), total serum bilirubin level continued to rise and reached 386 μmol/L. The patient was treated with prednisolone 40 mg/d, which was reduced by 5 mg/d at weekly intervals over a period of 8 wk. Response to treatment is shown in Figure 1. Her symptoms improved dramatically during treatment with the corticosteroid, with a concomitant restoration of normal liver biochemistry. She has now been followed up for more than 12 mo and is asymptomatic with weight gain of 10 kg.
A 66-year-old Caucasian woman presented with a 4-wk history of jaundice, right upper quadrant pain and pruritus. She had a past history of hysterectomy and bilateral salpingo-oopherectomy, carried out when she was 40 years old, complicated by post-operative deep vein thrombosis. She started Premarin (conjugated estrogens) 625 μg for menopausal symptoms when she was 50 years old, and had been taking this continuously up to the time of her presentation. The patient had no history of jaundice, hepatitis, blood transfusion or travel outside Western Europe. She had no risk factors for viral hepatitis and did not drink alcohol. On examination she was jaundiced, with no signs of chronic liver disease, and was afebrile. Tenderness on palpation of the right hypochondrium was evident, and the liver was palpable 2 cm below the costal margin.
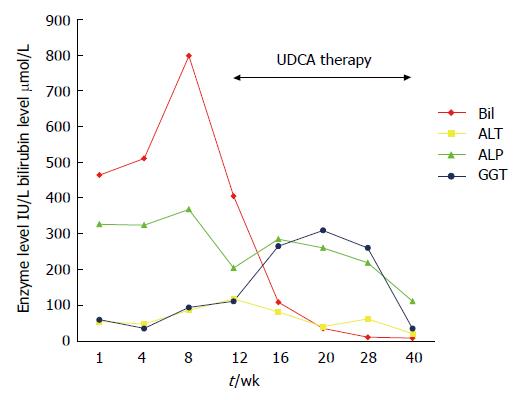
Laboratory investigation revealed a total serum bilirubin of 464 μmol/L, ALT 56 IU/L, ALP 328 IU/L and γ-GT 60 IU/L. Serological tests for hepatitis A, B and C were negative. Anti-nuclear factor, and anti-mitochondrial and smooth muscle antibodies were also negative. Ultrasound examination of the liver revealed a dilatated CBD measuring 10 mm and multiple small radio-opaque calculi in the duct.
The patient underwent ERCP and sphincterotomy; all intraductal stones were removed with a balloon. However, 8 wk after her initial presentation, the patient remained markedly jaundiced with worsening pruritus, malaise, anorexia and weight loss of 6 kg. Liver function tests had worsened: total serum bilirubin 798 μmol/L, ALT 87 IU/L, ALP 370 IU/L and γ-GT 96 IU/L (Figure 2). Premarin was stopped.
A CT scan showed a normal gallbladder, with no evidence of malignancy and no biliary tree dilatation. A repeat ERCP showed a normal biliary system with no evidence of retained CBD stones. Ursodeoxycholic acid (UDCA) 1200 mg/d was commenced following ERCP. Four weeks later, her symptoms had begun to improve, and her bilirubin level had fallen to around half its peak value. After a further month, her symptoms had resolved completely, although her liver biochemistry did not completely return to normal until 40 wk after her initial presentation.
In both cases, prolonged cholestasis after removal of CBD stones at ERCP was thought to have been caused by oral estrogen therapy. The association between oral estrogen-containing preparations and several cholestatic syndromes has been well recognised since 1962[1]; and of the canalicular type with little portal inflammation. It is thought that these reactions are because of the estrogen component; hence it is widely used as an experimental model of hepatocellular cholestasis. This effect is mediated by its glucuronidated metabolites, which inhibit canalicular bile salt and glutathione excretion, which results in inhibition of bile salt transport[7].
Clinical features include malaise, pruritus, jaundice and anorexia with subsequent weight loss, were seen in both our patients. These features are also seen in the clinical syndrome of intrahepatic cholestais of pregnancy (ICP). Indeed a Chilean group has found that, in 42 patients with cholestatic jaundice following use of oral estrogen compounds, 27 had previously jaundice and pruritus during pregnancy, which suggests a link between the two disorders[2]. This must be considered in the context of the strong geographical predilection of this disease to Chile. Crucially, withdrawal of the estrogenic effect by delivery of the fetus or drug withdrawal leads to improvement in liver function. This tends to take place over several weeks or months. Neither of the patients described here had a history of ICP, although liver function tests had not previously been performed.
Case 1 had proven gallstones at 6 wk post partum and liver function tests were consistent with stones in the CBD. The rapid deterioration in liver function both clinically and biochemically after commencing oral estrogens, and the resolution on discontinuation of the treatment support the hypothesis that the cholestasis was a drug-induced liver injury. Other potential causes were excluded. In particular, the absence of antimitochondrial antibodies, normal intrahepatic bile ducts on ERCP, and the spontaneous normalization of liver biochemistry excluded both primary biliary cirrhosis and primary sclerosing cholangitis.
Case 2 had been on estrogen replacement therapy for 16 year at the time of her presentation with obstructive jaundice. However, the only factor that could account for the prolonged cholestasis was the estrogen therapy, and the temporal relationship between stopping estrogens and improvement in liver function further supports the conjecture that hormone replacement therapy was responsible (although UDCA therapy may have played a role in the resolution of the cholestasis).
Since estrogen therapy alone did not cause jaundice in this patient, there must have been an interaction between the effects of mechanical obstruction to bile flow and the cholestatic effects of estrogens. The patient has remained off estrogens since the procedure.
The cause of the differences in time for resolution of jaundice in cases 1 and 2 is unclear. The jaundice in both cases was likely to have resolved following the withdrawal of estrogens, and it is possible that the longer duration of jaundice in case 2 was simply a reflection of the variable length of time for estrogen-induced cholestasis to resolve following treatment withdrawal. Typically, this takes several weeks to months, but it has been reported to take up to 10 years[3]. It is also possible that the difference in time for the jaundice to resolve reflected differences in the modes of action of the drugs used to treat the cholestasis. The anti-inflammatory effects of corticosteroids may be of benefit for individuals with a pre-existing mechanical obstruction. It is known that when biliary obstruction occurs, an inflammatory response is mounted with the release of pro-inflammatory cytokines (e.g. tumor necrosis factor-α and interleukin-1), a reduction in the expression of nuclear bile acid receptor, and infiltration of neutrophils, all of which are thought to aggravate cholestatic injury[8].
Corticosteroid use has previously been described in two cases reports of prolonged cholestasis following ERCP and successful removal of gallstones[9]. The patients were not taking estrogens (both were male) and the authors hypothesized that the canalicular function had been directly compromised by a mechanical obstruction. They also postulated that the radiocontrast medium infused under high pressure during ERCP may have had a toxic effect, with disruption of the canalicular membrane, but no evidence was given to support this theory. The two cases presented differed from both our patients, who were taking oral estrogens. Whilst the onset of jaundice appears to have been precipitated by the presence of obstructing intraductal calculi, the estrogen therapy appears to have been responsible for the persistence of cholestasis. Presumably, the biliary obstruction in some way sensitized the biliary canaliculi to the cholestatic effects of estrogens, perhaps through an associated inflammatory response (discussed above) that led to the prolonged cholestasis. The interaction between these two factors may in part be idiosyncratic and/or dependent on genetic factors, since this has not previously been reported.
UDCA was used in the second case because of concerns about side effects, particularly steroid-induced osteoporosis in this postmenopausal woman. Its use resulted in a less impressive response, but it may not have influenced the course of the cholestasis. However, evidence from a number of large studies has confirmed the efficacy of UDCA in ICP, and the drug is now used routinely for treating this condition[10]. The use of UDCA in treating cholestasis secondary to oral estrogen therapy in humans has not been reported, although ethinylestradiol-induced cholestasis in rats has been shown to respond to UDCA[11]. The authors of that study concluded that UDCA increased bile flow by increasing bile acid secretion, through the normalization of the expression of the canalicular bile salt export pump. UDCA has also been noted to decrease the glucuronidation of estrogens, thereby decreasing the production of cholestatic metabolites[12].
Although corticosteroid treatment appeared more effective than UDCA, the two agents have been compared in a randomized study in ICP. Treatment with UDCA led to a significant reduction in ALT and bilirubin and an improvement in pruritus, while dexamethasone had no such effects[13].
Endoscopists who carry out ERCPs should be aware of the potential causes of persistent jaundice following the removal of intraductal stones. In female patients with protracted jaundice, the possibility of estrogen-induced cholestasis should always be considered, and oral estrogens discontinued. In such cases, a short course of corticosteroid treatment should be considered.
S- Editor Zhu LH L- Editor Kerr C E- Editor Ma WH
| 1. | PEREZ-MERA RA, SHIELDS CE. Jaundice associated with norethindrone acetate therapy. N Engl J Med. 1962;267:1137-1138. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 17] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 2. | Reyes H, Simon FR. Intrahepatic cholestasis of pregnancy: an estrogen-related disease. Semin Liver Dis. 1993;13:289-301. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 122] [Cited by in F6Publishing: 120] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 3. | Wedén M, Glaumann H, Einarsson K. Protracted cholestasis probably induced by oral contraceptive. J Intern Med. 1992;231:561-565. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7] [Cited by in F6Publishing: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 4. | Donovan JM. Physical and metabolic factors in gallstone pathogenesis. Gastroenterol Clin North Am. 1999;28:75-97. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 45] [Cited by in F6Publishing: 49] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 5. | Cirillo DJ, Wallace RB, Rodabough RJ, Greenland P, LaCroix AZ, Limacher MC, Larson JC. Effect of estrogen therapy on gallbladder disease. JAMA. 2005;293:330-339. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 174] [Cited by in F6Publishing: 159] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 6. | Uhler ML, Marks JW, Voigt BJ, Judd HL. Comparison of the impact of transdermal versus oral estrogens on biliary markers of gallstone formation in postmenopausal women. J Clin Endocrinol Metab. 1998;83:410-414. [PubMed] [Cited in This Article: ] |
| 7. | Meyers M, Slikker W, Pascoe G, Vore M. Characterization of cholestasis induced by estradiol-17 beta-D-glucuronide in the rat. J Pharmacol Exp Ther. 1980;214:87-93. [PubMed] [Cited in This Article: ] |
| 8. | Trauner M, Boyer JL. Cholestatic syndromes. Curr Opin Gastroenterol. 2004;20:220-230. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 26] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 9. | Dourakis SP, Mayroyannis C, Alexopoulou A, Hadziyannis SJ. Prolonged cholestatic jaundice after endoscopic retrograde cholangiography. Hepatogastroenterology. 1997;44:677-680. [PubMed] [Cited in This Article: ] |
| 10. | Kondrackiene J, Beuers U, Kupcinskas L. Efficacy and safety of ursodeoxycholic acid versus cholestyramine in intrahepatic cholestasis of pregnancy. Gastroenterology. 2005;129:894-901. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 174] [Cited by in F6Publishing: 179] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 11. | Micheline D, Emmanuel J, Serge E. Effect of Ursodeoxycholic Acid on the Expression of the Hepatocellular Bile Acid Transporters (Ntcp and bsep) in Rats With Estrogen-Induced Cholestasis. J Pediatr Gastroenterol Nutr. 2002;35:185-191. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 31] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 12. | Sánchez Pozzi EJ, Crocenzi FA, Pellegrino JM, Catania VA, Luquita MG, Roma MG, Rodríguez Garay EA, Mottino AD. Ursodeoxycholate reduces ethinylestradiol glucuronidation in the rat: role in prevention of estrogen-induced cholestasis. J Pharmacol Exp Ther. 2003;306:279-286. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 25] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 13. | Glantz A, Marschall HU, Lammert F, Mattsson LA. Intrahepatic cholestasis of pregnancy: a randomized controlled trial comparing dexamethasone and ursodeoxycholic acid. Hepatology. 2005;42:1399-1405. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 184] [Cited by in F6Publishing: 192] [Article Influence: 10.1] [Reference Citation Analysis (0)] |