Published online Jan 7, 2007. doi: 10.3748/wjg.v13.i1.39
Revised: September 25, 2006
Accepted: November 21, 2006
Published online: January 7, 2007
For genome multiplication hepadnaviruses use the transcriptional machinery of the cell that is found within the nucleus. Thus the viral genome has to be transported through the cytoplasm and nuclear pore. The intracytosolic translocation is facilitated by the viral capsid that surrounds the genome and that interacts with cellular microtubules. The subsequent passage through the nuclear pore complexes (NPC) is mediated by the nuclear transport receptors importin α and β. Importin α binds to the C-terminus of the capsid protein that comprises a nuclear localization signal (NLS). The exposure of the NLS is regulated and depends upon genome maturation and/or phosphorylation of the capsid protein. As for other karyophilic cargos using this pathway importin α interacts with importin β that facilitates docking of the import complex to the NPC and the passage through the pore. Being a unique strategy, the import of the viral capsid is incomplete in that it becomes arrested inside the nuclear basket, which is a cage-like structure on the karyoplasmic face of the NPC. Presumably only this compartment provides the factors that are required for capsid disassembly and genome release that is restricted to those capsids comprising a mature viral DNA genome.
- Citation: Kann M, Schmitz A, Rabe B. Intracellular transport of hepatitis B virus. World J Gastroenterol 2007; 13(1): 39-47
- URL: https://www.wjgnet.com/1007-9327/full/v13/i1/39.htm
- DOI: https://dx.doi.org/10.3748/wjg.v13.i1.39
Eukaryotic cells are divided into different compartments and viruses have to get access to the compartment that provides the cellular machinery for replication. Unlike bacteriophages that “just” have to pass the bacterial wall and membrane all viruses that infect eukaryotic cells have to travel through the cell to reach the place of replication. Dependent upon the virus and the type of genome, the machinery of DNA replication, transcription and RNA processing may be required. Gaining access to the nucleus where these factors are found requires active transport since passive diffusion is ineffective.
Although summarized by the term “intracellular transport” one has to differentiate between the intracytoplasmic transport towards the nucleus and the passage through the nuclear envelope into the karyoplasm. Nucleic acids are not karyophilic per se. Thus proteins attached to the viral genome must interact with cellular factors that facilitate the different transport processes. As intracytosolic and nuclear transport are based on different mechanisms it is evident that the interacting domains on the viral proteins have to be exposed in a coordinated manner. Moreover an effective virus, meaning a virus with a good particle-infectious unit ratio, has to release its genome from surrounding proteins only after termination of the various transport steps preventing abortion of the infection process. It has to be considered that the analysis of the underlying principles is not only important for analysis of potential drug targets for treatment of individual viral infections but also for creation of efficient vectors in gene therapy.
The need for active and directed intracytosolic transport results from the high viscosity of the cytoplasm. Protein concentrations of 170-350 mg/mL, RNA concentrations of 30 μg/mL[1] and micro compartmentalization evoke a viscosity 10 to 100 fold higher than the viscosity of water[2]. In consequence diffusion processes are enormously reduced so that only particles with diameters below 50 nm significantly diffuse[3]. Obviously such a passive movement is incompatible with efficient trafficking of organelles. Eukaryotic cells thus provide different active transport machineries that are not only used by large structures but even by small macromolecules as it was shown for the heat shock protein 90 (Hsp90)/glucocorticoid receptor β (GRβ) complex[4] and the human tumour suppressor protein p53[5]. It is thus likely that structures as the HBV capsid with a diameter of 36 nm (80 % of the capsids that show a T = 4 symmetry, 20 % exhibit a T = 3 symmetry and a diameter of 32 nm[6,7]) use the same active cellular transport pathways towards the nucleus.
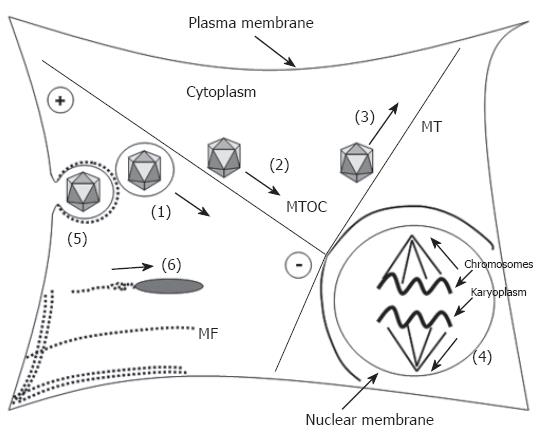
Most investigations on intracytosolic transport of viruses are done by adding inhibitors to cells during infection. To prevent misinterpretations it is important to realize the variety of cellular processes that are affected. Eukaryotic cells provide two active cytoplasmic transport systems based on microfilaments (MF) and microtubules (MT). Microfilaments have diameters of 5-9 nm and are double-stranded helical polymers of the ATPase actin. They form linear bundles, 2D networks and 3D gels and are most highly concentrated underneath the plasma membrane. Microfilaments are dynamic polar structures with a fast-growing plus-end and a relatively inert, slow-growing minus-end. They stabilize the cell structure and are involved in cell movement e.g. by filopodia and lamellopodia. With a velocity of 2-6 mm/d transport via MF is slow[8,9] and generally thought to serve as the transport pathway for short distances.
Beside its role in transport actin is also involved in the internalization and/or formation of endocytic vesicles. In clathrin-mediated endocytosis a functional actin cytoskeleton enhances the internalization of the clathrin-coated vesicles but without being obligatory[10-12]. In phagocytosis and macropinocytosis local actin polymerization at the cytosolic site of the plasma membrane is required for vesicle formation as well as actin depolymerization[13-16]. Similarly, releasing of caveolin-coated vesicles into the cytoplasm depends on actin polymerization and depolymerization in caveolae-mediated endocytosis[17,18].
Microtubules are hollow cylinders of 11-13 protofila-ments made of the GTPase α- and β-tubulin. MT are 25 nm in diameter and like MF they have a highly dynamic, fast-growing plus-end and a less dynamic minus-end, which is typically attached to a microtubule-organizing centre (MTOC). MT surround the nucleus and extend from the perinuclear MTOC to the cell periphery. They are thought to be the major long-range transport system[9] allowing a velocity of 3-5 mm per hour (HSV 1 capsids, retrograde)[19].
For both MT and MF two different mechanisms of transport exist (Figure 1) using polymerization (filament growth)/depolymerization (filament shrinkage) or motor proteins[20]. In the first mechanism the cargo binds dynamically via adapter proteins to one end of a growing or shortening filament and can be pushed or pulled in the given direction. Mitotic chromosomes for example bind at its kinetochor via a dynein/dynactin-complex to the plus-end of the kinetochor-MT[21-23]. Depolymerization at the plus-end as well as depolymerization at the minus-end directs the chromosomes to the minus-end that is fixed at the spindle pole. Phagosomes[24,25], macropinosomes[26,27] endosomes and lysosomes[27,28] are able to cross the cytosol by the help of a polymerizing actin tail comparable to the movement of the bacteria listeria monocytogenes[29-31] and the nuclear polyhedrosis virus (NPV) capsid[32-34].
However the most commonly used transport strategy for macromolecules, mRNA, RNPs (ribonucleoprotein complexes), cellular organelles and vesicles along polar MF or MT involves motor protein complexes, namely myosins, kinesins and dyneins[20,35-38]. These cargo specific filament binding proteins exhibit a motor domain (ATPase) and move ATP-dependent and unidirectional along the stable filaments. Dependent upon the involved filament and transport direction different motor protein complexes are used. Class VI myosins are unconventional myosins which move towards the minus-end of MF[39]. They are involved in the transport of clathrin-coated vesicles from the plasma membrane into the inner cell[40,41]. Class I and class V myosins in contrast migrate towards the plus-end of MF[20,35]. Myosin I mediates transport of membranes whereas myosin V is responsible for the transport of organelles, as e.g. recycling endosomes towards the plasma membrane[20,42,43]. In addition myosin V can transport cargos along MT[44] thus linking both transport systems.
The MT specific kinesins can be distinguished upon the localization of the conserved motor domain into conventional kinesins, which have an N-terminal motor domain and that direct cargos in the anterograde direction towards the plus-end of the MT (i.e. towards the cell periphery)[20,36]. They participate in the intracellular transport and localization of different cellular membrane organelles, as the extension of the ER (endoplasmatic reticulum) from the nucleus towards the cell periphery[45,46]. In addition, conventional kinesins are used in directing progeny capsids of herpes simplex virus 1 (HSV 1) in the anterograde direction[47].
Unconventional kinesins, which have a C-terminal motor domain move in contrast towards the minus-end of MT. They mediate axonal MT transport of vesicles and organelles[36].
The cytoplasmic dynein is commonly associated with the cofactor dynactin (dynactin-complex)[48] transporting cellular vesicles (endosomes and caveolin-negative vesicles[49-52] and macromolecules (e.g. tumor suppressor protein p53[5]) in the retrograde direction towards MT minus-end (the MTOC). Apparently the dynein-complex is frequently used in transport of viral capsids towards the nucleus as it was shown for adenoviruses[53-55], parvoviruses[56-59] and HSV 1 upon infection[47,60].
With the exception of NPV all viruses being analyzed so far make use of the MT transport system for their transport towards the nucleus[61]. This includes large viruses that have to travel long distances as the herpes simplex virus 1 (HSV 1;[47]) that has to be transported for centimeters between the axon end and the cell body and parvoviruses (18-26 nm)[56,58,62] that are below the diffusion limit inside the cytoplasm.
Evaluating the transport mode of hepatitis B virus is however not trivial as the entry mechanism is not fully understood. This is mainly caused by a lack of an appropriate and effective in vitro infection system that allows study of the early steps of the hepadnaviral life cycle. As described in “Viral and cellular determinants involved in hepadnaviral entry” hepadnaviruses enter the cells in vesicles[63] but do not need acidification for infection[64]. This step that normally occurs upon endocytosis has a major effect on viral structures as described for adeno- and parvoviruses[57,65,66]. The altered structure distinguishes capsids that have passed endocytosis from progeny capsids that are newly synthesized. The different exposed epitopes allow variant interactions so that incoming capsids are targeted to the nucleus while progeny capsids are not.
As such an acid-induced conformational change is missing in the hepadnaviral life cycle the viral capsids released from the endosomal pathway and the newly synthesized progeny capsids have the same structure. Consequently both types of capsid can participate in nuclear transport of the viral genome.
The restrictions of the experimental systems for analysis of hepadnaviral viral infections are most likely caused by an insufficient entry. It was thus a self suggested idea to replace the viral surface proteins by a lipid shell as it is done in protein transfection (lipofection)[67]. The lipids fuse with the plasma membrane and release the capsids into the cytoplasm. For studying the intracellular transport of the hepadnaviral capsid lipofection has the advantage that no cellular transport vesicles are involved.
In fact lipofection of hepatoma cells yielded in a highly productive HBV infection similar to the in vivo efficiency.
As lipofection is independent upon receptors high amounts of capsids could be loaded on the cells. This allows one to follow the fate of the capsids and of the viral genomes by microscopical techniques[67] showing that the capsids accumulated at the nuclear envelope after 15 min. As diffusion can be assumed to take 1 h (for calculation see[9]), these data imply a directed active capsid transport towards the nucleus. Released viral genomes occurred exclusively inside the karyoplasm suggesting site-specific disintegration at the nuclear envelope. The use of the MT depolymerizing drug nocodazole inhibits accumulation of capsids at the nuclear envelope and the release of genomes suggesting that genome liberation requires transport to the nucleus.
The microscopical finding of an active MT-mediated transport towards the nucleus was supported analysing the effect of nocodazole on the hepadnaviral life-cycle. It was demonstrated that MT are essential for formation of nuclear DNA (cccDNA that occurs only after repair of the partially double stranded DNA genome within the nucleus) and for amplification of viral DNA via synthesis of progeny mature capsids.
The use of the MT transport system was confirmed in another experimental system in which capsids were injected into the cytosol of Xenopus laevis oocytes. Due to the longer transport distances 30 min are required for the capsids to reach the nucleus upon injection at the pole of the oocyte opposed the nucleus. Assuming a distance of 0.5 mm that has to be bridged this period is consistent with the cytosolic transport of HSV 1 capsids that traverse the cytoplasm with 3-5 mm per hour (retrograde transport,[9]). As electron microscopy was used as read-out for capsid localization these data could show that the capsids did not bind to undefined sites of the nuclear envelope but to the nuclear pore complexes (NPC)[68]. However, when anti tubulin-antibodies were preinjected the arrival of the capsids at the NPCs was inhibited confirming that even in cells only distantly related to human hepatocytes the same transport system is used.
Although conclusive, it must be considered that all the observations described above were not done in the “authentic primary” cells and that a liver-specific factor may alter e.g. the place of genome release and lead to another transport model that does not involve the capsid. However, the generation of capsids with a translocation motif (TLM) fused to the N-terminus of the capsid protein recently gave further support[69]. These capsids were still capable of encapsidating the polymerase and the pregenome so that mature DNA capsids were generated being able to initiate an HBV infection in primary human hepatocytes. Although the mode of uptake by a TLM remains controversial-directly penetrating the plasma membrane or using transporters[70]-a clear uptake of the capsids could be observed, resulting in accumulation in the perinuclear region. This uptake was not observed when wild-type capsids were used being contradictory to most recent results of others[71]. Irrespectively of this divergence the location of the TLM capsids at the perinuclear region where the MTOC is situated supports that the MT were used for transport towards the nucleus.
An open question not being answered for any cargo that uses MT transport for reaching the nucleus is derived from the polarity and arrangement of the MT. As their minus-end is not directly located at the NPC but attached to the MTOC the cargo must cross the distance between the MTOC and the nucleus. There are observations that even for this short gap passive diffusion is not likely: HSV 1 capsids show an equal distribution around the nucleus after infection and do not accumulate at those NPCs adjacent to the MTOC[47]. However, the mechanism of this translocation remains open.
Viruses that replicate in the nucleus of non-dividing cells have to traverse the nuclear envelope. For this reason nuclear proteins pass the nuclear pore complexes (NPCs). NPCs are large proteinaceous structures consisting of 30 different proteins[72], collectively termed nucleoporins (Nups). Nucleoporins exist in multiples copies, forming a complex of estimated 125 MD[73]. Many nucleoporins contain distinct domains of phenylalanine-glycine (FG) repeats, which mediate the main interaction between nucleoporins and soluble transport receptors. The NPC consists of a central ring-like framework with 8-fold symmetry, representing the part of the complex that is embedded in the nuclear envelope (NE) Attached to a cytoplasmic ring moiety 8 cytoplasmic filaments form an initial docking side for transport complexes. On the karyoplasmic face, 8 fibres form the cage-like structure of the nuclear basket[74]. The central framework is a ring-like assembly built around a central pore through which the exchange of macromolecules occurs. The dimension of the nuclear pore restricts complexes to a diameter of 39 nm including their shell of transport receptors[68]; a size that is exceeded by most viruses or subviral particles.
NPCs regulate the traffic of proteins and nucleic acids into and out of the nucleus[75]. Substrates smaller than roughly 9 nm in size, including ions, metabolites and proteins, travel through the NPC in a diffusion-controlled and energy independent manner[76].
Most nuclear cargos exhibit signals that interact with nuclear transport receptors of the importin β superfamily, comprising importins and transportins. All members of this family exhibit an N-terminal RanGTP-binding domain which is important for dissociating receptor and cargo (reviewed by[77]). There is a variety of different signals that are recognized as exemplified by the M9 domain, bound by transportin, polypeptides of basic amino acids that represent an importin β binding domain (IBB) and “classical” nuclear localization signals (NLSs) that show the consensus sequence K(K/R)X(K/R)[78]. The classical NLS does not directly bind to the transport-mediating receptor importin β (Imp β) but requires an adapter molecule, importin α (Imp α), which connects NLS and - via its IBB-Imp β.
The driving force of nuclear import and export is determined by the different concentrations of RanGTP in the nucleus versus the cytoplasm. RanGTP that is enriched in the karyoplasm, interacts with the transport receptors of the import complex, leading to dissociation of cargo and receptor. While the cargo diffuses deeper into the karyoplasm, the RanGTP-receptor complex becomes exported to the cytoplasm.
Hepadnaviral genomes have to enter the nucleoplasm for replication. As hepatocytes are terminally differentiated cells that do not divide they cannot wait until the cell undergoes mitosis as most retroviruses-excluding HIV-do. As karyophilic proteins the capsids use the nuclear pore complex to get for access to the nucleoplasm.
Nucleic acids are not karyophilic per se. Therefore one or more proteins attached to the genome must interact with cellular nuclear import receptors. In case of the hepadnaviruses three models, each involving a different mediator, may play a crucial role in the nuclear import of the HBV genome: (1) The viral polymerase of the Hepatitis B virus. The enzyme is covalently attached to the viral genome and probably contains a hidden NLS. Expression of the polymerase in eukaryotic cells revealed that the protein stays cytoplasmic[79] but extraction of this complex from mature virions showed that it enters the nucleus[80]. However, the procedure of extraction requires harsh treatment and thus structure altering methods. (2) Some of the heat shock proteins as Hsc70 or Hsp90 activating the polymerase[81-84] and (3) the capsid proteins surrounding the viral replication complex.
The lipofection experiments described above, show that released viral DNA is exclusively present within the nucleus. The release is combined with the accumulation of capsids at the nuclear envelope. It thus has to be concluded that if the polymerase mediates nuclear import the release of the import complex must occur at the nuclear envelope, probably after docking of the capsids to the nuclear pore. Examples for such a pathway are the HSV 1 capsid that becomes opened upon the interaction of a penton with the NPC and adenovirus 2 that releases the complex of DNA and associated proteins at the pore[47,85-88].
The polymerase-associated heat shock proteins may act in a similar manner. For example, the interaction with Hsc70 that exhibits a nuclear transport capacity is established[84]. The capsid-nuclear envelope interaction was more extensively investigated in cell biological assays and in microinjection experiments using oocytes of Xenopus laevis. Another experimental design is based on Digitonin permeabilized cells most commonly used in studying nuclear import reactions in detail. Digitonin permeabilizes only cholesterol containing membranes as e.g. the plasma membrane and membranes of mitochondria. Other membranes as the nuclear and ER membrane remain unaffected[89]. In general adhesive cells are analysed under conditions where they attach to the surface of a glass cover slip thus the Digitonin has to be removed by washing steps. The washing removes the soluble cytosolic proteins (and some small nuclear proteins that rapidly diffuse out of the nucleus) including the cellular nuclear transport factors. Consequently these factors have to be replaced either by addition of selected import factors or in form of a cytosolic extract. However, as the nuclear transport capacity is conserved this process can be transferred to the in vivo situation presupposed that the subjected cargo with its modification is physiological.
Microinjection in the cytoplasm is another established technique. For electron microscopy Xenopus laevis oocytes are frequently used as the huge dimensions of the nucleus allow the analysis of multiple sections. Since nuclear import is phylogenetically well conserved the results are transferable to other cell types as long as no embryogenesis related processes are affected.
These assays and biochemical analyses have been used to analyse the nuclear import of the hepadnaviral genome in more detail. Using permeabilized cells it was shown that the HBV capsid protein contains an NLS within its C-terminal domain. This domain is hidden in the lumen of RNA-containing capsids expressed in E. coli and in eukaryotically expressed capsids devoid of the polymerase[90,91]. These capsids failed to interact with nuclei of permeabilized cells. However, the exposure of the C-terminus on the capsid surface was shown to be linked to genome maturation as in vitro studies revealed[90]. Interestingly, the exposure could be initiated in RNA-containing capsids expressed in E. coli when the C-terminus was in vitro phosphorylated by protein kinase C[92] or protein kinase A[93]. These observations strengthen the idea that phosphorylation and genome maturation are linked. The impact of the exposed C-termini for nuclear pore complex association becomes evident by cleavage experiments in which the C-termini were removed from mature capsids followed by subjection of the capsids to Digitonin-permeabilized cells. According with the hypothesis of a capsid-mediated NPC docking the capsids and the necessity of exposed NLS to the digested capsids failed to interact with the nuclei. Consistently with the identification of an NLS on the capsid protein importin β was found to be the mediator of NPC interaction, requiring importin α as an adapter protein.
Most fascinating is the different import behaviour between capsids that have undergone genome maturation to a different extent. Capsids with an immature DNA genome interacted with the NPCs but remained associated with the pores while subjecting mature capsids to the permeabilized cells resulted in a nuclear capsid stain and released viral genomes within the nucleus.
Based on these data all three import models-polymerase, heat shock proteins or capsid mediation-could be true. The mature capsids could interact with the NPC releasing their genome followed by import of the genome mediated by the polymerase or heat shock proteins. The dissociated capsid subunits enter the nucleus as they apparently do in HBV infected individuals. The immature capsids could have been just more stable thus failing to disintegrate and to release the genome.
Alternatively, only the mature capsids that expose more NLS may have become surrounded by enough nuclear transport receptors to pass a hydrophobic mesh caused by the crosslinking of hydrophobe FxFG repeats of nucleoporins. Only these capsids interact with the factors required for genome release.
To differentiate between the models further investigations were initiated to follow the fate of the capsids at the NPC. Surprisingly, electron microscopy after microinjection into Xenopus laevis oocytes showed that not only the mature capsids passed the pore and entered the nuclear basket but also immature capsids that apparently failed to diffuse deeper into the karyoplasm. Consistent with the experiments in permeabilized cells, RNA containing capsids did not interact with the NPCs.
For the model of nuclear import these findings implied that apparently the capsids mediated the passage through the nuclear pore into the nuclear basket. Here only the mature capsids disintegrated while the immature capsid stay arrested. Hypothetically this arrest can increase efficiency of HBV infection, assuming that genome maturation proceeds in these capsids.
Hepadnaviral polymerases can only successfully synthesize the full length viral DNA when interacting with the capsid proteins. In fact, recent studies on the phosphorylation sites of the HBV capsid protein show that one serine residue (Ser 157) has to be phosphorylated for pregenome packaging while serine 164 is required for allowing DNA synthesis[94,95]. The highly efficient infections by hepadnaviruses thus imply that the genome release is well coordinated in that only capsids with a mature genome disintegrate. The factor however that apparently is only present within the nucleus remains unknown. Incubations of mature capsids with nuclear extracts failed to induce significant genome release while permeabilized hepatoma cells only require minutes to release thousands of HBV genomes per nucleus. Furthermore a liver-specific factor must be assumed as genome release is more efficient in permeabilized hepatoma- than in HeLa cells. In vivo, this conclusion is supported by observations of Untergasser et al[96], who observed that cccDNA generation does not occur upon infection of dendritic cells with chimera of HBV and adenovirus.
Another open question is raised by the high copy number of empty capsids found in the nuclei of HBV infected hepatocytes or in hepatocytes of mice that are transgenic for HBV. Apparently they are not derived from capsids that have transported the genome into the nucleus as the half-life of the genome-as different data in the literature are (ranging from 3 d[97], 55 d[98] to a non-relevant degradation[99])-do not explain their frequent abundance. However, the capsid protein is strongly over expressed with regard to the number of viruses and capsids that participate in nuclear entry of the viral genome. Apparently capsid proteins have a nuclear transport capacity by their NLS, which is not hidden as long as they are not assembled to particles. Based on the results described above one must thus conclude that not capsids but the supernumerous capsid proteins or their assembly intermediates are imported. In accordance with consistent biochemical data of all groups studying the assembly process, it must be proposed that the capsid proteins do not need any other protein for assembly. The only driving force is the affinity to each other being supported by their interaction with other components as e.g. RNA. It is thus likely that after import of high numbers of capsid proteins into the nucleus the threshold concentration is reached resulting in rapid assembly to particles.
Nonetheless, one has to ask why the capsid proteins do not become arrested in the nuclear basket as the immature capsids. One can assume that assembled capsids can interact with at least eight basket proteins, most likely the nucleoporin 153[100], that are arresting the capsid. Although the answer remains experimentally open a protein monomer in contrast is restricted to one interaction that is in competition with the thousands of proteins that pass the nuclear pore every second.
The meaning of the nuclear assembly is unsolved. It might be just a side effect caused by the intrinsic assembly ability of the capsid. However, as the capsid proteins interact preferentially with single stranded nucleic acids the assembly may prevent interference with the cellular transcription and RNA export machinery thus reducing toxicity of the virus and ensuring a long life of the infected cell for persistent virus production.
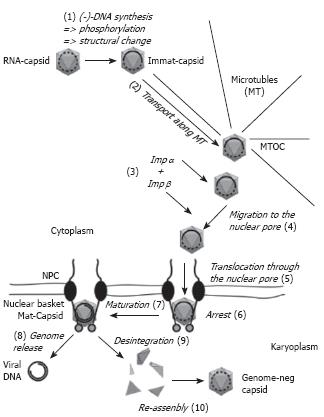
The summary of the current knowledge on the nuclear import of the hepadnaviral genome is depicted in Figure 2. It must be considered however that there are several elements as e.g. the co-ordination of the transport processes that are yet unknown. (1) Upon genome maturation and phosphorylation the hepadnaviral capsids undergo a structural change that leads to exposure of increasing numbers of the C-termini that are part of the capsid protein. (2) The capsids are transported towards the microtubule-organizing centre (MTOC), which is located at the perinuclear region. (3) As the exposed C-terminal domain exhibits a nuclear localization signal the probability of an interaction with the adaptor protein importin α increases. As in the physiological import of karyophilic proteins this complex is bound by importin β. Whether this acquisition already occurs during the MT-mediated transport or (4) during the unknown passage from the MTOC to the nucleus remains open. (5) Importin β next facilitates docking to the cytosolic fibres of the nuclear pore and translocation of the complex into the nuclear basket. (6) After dissociation of the nuclear import receptors from the capsid the capsid most likely interacts with a protein of the basket. (7) While immature capsids stay arrested and may continue with genome maturation, (8) mature capsids that are less stable release the genome with the associated proteins into the nucleus where genome repair takes place. (9) Supernumerous capsid proteins that result from disintegration can diffuse deeper into the karyoplasm where (10) they re-assemble after the capsid protein concentration reaches the threshold concentration for assembly.
S- Editor Liu Y L- Editor Alpini GD E- Editor Ma WH
| 1. | Kehlenbach RH. In vitro analysis of nuclear mRNA export using molecular beacons for target detection. Nucleic Acids Res. 2003;31:e64. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 17] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 2. | Janmey PA, Weitz DA. Dealing with mechanics: mechanisms of force transduction in cells. Trends Biochem Sci. 2004;29:364-370. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 200] [Cited by in F6Publishing: 205] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 3. | Luby-Phelps K. Physical properties of cytoplasm. Curr Opin Cell Biol. 1994;6:3-9. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 158] [Cited by in F6Publishing: 170] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 4. | Zhang X, Clark AF, Yorio T. Heat shock protein 90 is an essential molecular chaperone for nuclear transport of glucocorticoid receptor beta. Invest Ophthalmol Vis Sci. 2006;47:700-708. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 27] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 5. | Giannakakou P, Sackett DL, Ward Y, Webster KR, Blagosklonny MV, Fojo T. p53 is associated with cellular microtubules and is transported to the nucleus by dynein. Nat Cell Biol. 2000;2:709-717. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 282] [Cited by in F6Publishing: 291] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 6. | Crowther RA, Kiselev NA, Böttcher B, Berriman JA, Borisova GP, Ose V, Pumpens P. Three-dimensional structure of hepatitis B virus core particles determined by electron cryomicroscopy. Cell. 1994;77:943-950. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 394] [Cited by in F6Publishing: 400] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 7. | Kenney JM, von Bonsdorff CH, Nassal M, Fuller SD. Evolutionary conservation in the hepatitis B virus core structure: comparison of human and duck cores. Structure. 1995;3:1009-1019. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 107] [Cited by in F6Publishing: 112] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 8. | Brady ST. Molecular motors in the nervous system. Neuron. 1991;7:521-533. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 94] [Cited by in F6Publishing: 111] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 9. | Sodeik B. Mechanisms of viral transport in the cytoplasm. Trends Microbiol. 2000;8:465-472. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 232] [Cited by in F6Publishing: 242] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 10. | Fujimoto LM, Roth R, Heuser JE, Schmid SL. Actin assembly plays a variable, but not obligatory role in receptor-mediated endocytosis in mammalian cells. Traffic. 2000;1:161-171. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 312] [Cited by in F6Publishing: 320] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 11. | Brodsky FM, Chen CY, Knuehl C, Towler MC, Wakeham DE. Biological basket weaving: formation and function of clathrin-coated vesicles. Annu Rev Cell Dev Biol. 2001;17:517-568. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 498] [Cited by in F6Publishing: 481] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 12. | Apodaca G. Endocytic traffic in polarized epithelial cells: role of the actin and microtubule cytoskeleton. Traffic. 2001;2:149-159. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 322] [Cited by in F6Publishing: 339] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 13. | Greenberg S. Signal transduction of phagocytosis. Trends Cell Biol. 1995;5:93-99. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 124] [Cited by in F6Publishing: 129] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 14. | Greenberg S, Grinstein S. Phagocytosis and innate immunity. Curr Opin Immunol. 2002;14:136-145. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 402] [Cited by in F6Publishing: 378] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 15. | Ridley AJ, Paterson HF, Johnston CL, Diekmann D, Hall A. The small GTP-binding protein rac regulates growth factor-induced membrane ruffling. Cell. 1992;70:401-410. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2825] [Cited by in F6Publishing: 2914] [Article Influence: 91.1] [Reference Citation Analysis (0)] |
| 16. | Dowrick P, Kenworthy P, McCann B, Warn R. Circular ruffle formation and closure lead to macropinocytosis in hepatocyte growth factor/scatter factor-treated cells. Eur J Cell Biol. 1993;61:44-53. [PubMed] [Cited in This Article: ] |
| 17. | Pelkmans L, Püntener D, Helenius A. Local actin polymerization and dynamin recruitment in SV40-induced internalization of caveolae. Science. 2002;296:535-539. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 555] [Cited by in F6Publishing: 547] [Article Influence: 24.9] [Reference Citation Analysis (0)] |
| 18. | Pelkmans L, Helenius A. Endocytosis via caveolae. Traffic. 2002;3:311-320. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 559] [Cited by in F6Publishing: 524] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 19. | Lycke E, Kristensson K, Svennerholm B, Vahlne A, Ziegler R. Uptake and transport of herpes simplex virus in neurites of rat dorsal root ganglia cells in culture. J Gen Virol. 1984;65 ( Pt 1):55-64. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 102] [Cited by in F6Publishing: 106] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 20. | Kreis TE, Vale R. Guidebook to the Cytoskeletal and Motor Proteins. 2nd ed. Oxford: Oxford University Press 1999; . [Cited in This Article: ] |
| 21. | Echeverri CJ, Paschal BM, Vaughan KT, Vallee RB. Molecular characterization of the 50-kD subunit of dynactin reveals function for the complex in chromosome alignment and spindle organization during mitosis. J Cell Biol. 1996;132:617-633. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 524] [Cited by in F6Publishing: 546] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 22. | Mitchison TJ, Salmon ED. Poleward kinetochore fiber movement occurs during both metaphase and anaphase-A in newt lung cell mitosis. J Cell Biol. 1992;119:569-582. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 204] [Cited by in F6Publishing: 233] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 23. | Waters JC, Mitchison TJ, Rieder CL, Salmon ED. The kinetochore microtubule minus-end disassembly associated with poleward flux produces a force that can do work. Mol Biol Cell. 1996;7:1547-1558. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 161] [Cited by in F6Publishing: 163] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 24. | Zhang F, Southwick FS, Purich DL. Actin-based phagosome motility. Cell Motil Cytoskeleton. 2002;53:81-88. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 34] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 25. | Desjardins M, Griffiths G. Phagocytosis: latex leads the way. Curr Opin Cell Biol. 2003;15:498-503. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 123] [Cited by in F6Publishing: 128] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 26. | Merrifield CJ, Moss SE, Ballestrem C, Imhof BA, Giese G, Wunderlich I, Almers W. Endocytic vesicles move at the tips of actin tails in cultured mast cells. Nat Cell Biol. 1999;1:72-74. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 250] [Cited by in F6Publishing: 264] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 27. | Taunton J. Actin filament nucleation by endosomes, lysosomes and secretory vesicles. Curr Opin Cell Biol. 2001;13:85-91. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 105] [Cited by in F6Publishing: 108] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 28. | Taunton J, Rowning BA, Coughlin ML, Wu M, Moon RT, Mitchison TJ, Larabell CA. Actin-dependent propulsion of endosomes and lysosomes by recruitment of N-WASP. J Cell Biol. 2000;148:519-530. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 341] [Cited by in F6Publishing: 325] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 29. | Tilney LG, DeRosier DJ, Tilney MS. How Listeria exploits host cell actin to form its own cytoskeleton. I. Formation of a tail and how that tail might be involved in movement. J Cell Biol. 1992;118:71-81. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 92] [Cited by in F6Publishing: 100] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 30. | Tilney LG, DeRosier DJ, Weber A, Tilney MS. How Listeria exploits host cell actin to form its own cytoskeleton. II. Nucleation, actin filament polarity, filament assembly, and evidence for a pointed end capper. J Cell Biol. 1992;118:83-93. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 73] [Cited by in F6Publishing: 86] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 31. | Tilney LG, Portnoy DA. Actin filaments and the growth, movement, and spread of the intracellular bacterial parasite, Listeria monocytogenes. J Cell Biol. 1989;109:1597-1608. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 977] [Cited by in F6Publishing: 1038] [Article Influence: 29.7] [Reference Citation Analysis (0)] |
| 32. | Charlton CA, Volkman LE. Penetration of Autographa californica nuclear polyhedrosis virus nucleocapsids into IPLB Sf 21 cells induces actin cable formation. Virology. 1993;197:245-254. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 95] [Cited by in F6Publishing: 102] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 33. | Lanier LM, Slack JM, Volkman LE. Actin binding and proteolysis by the baculovirus AcMNPV: the role of virion-associated V-CATH. Virology. 1996;216:380-388. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 36] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 34. | Lanier LM, Volkman LE. Actin binding and nucleation by Autographa california M nucleopolyhedrovirus. Virology. 1998;243:167-177. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 86] [Cited by in F6Publishing: 86] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 35. | Mermall V, Post PL, Mooseker MS. Unconventional myosins in cell movement, membrane traffic, and signal transduction. Science. 1998;279:527-533. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 496] [Cited by in F6Publishing: 524] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 36. | Goldstein LS, Philp AV. The road less traveled: emerging principles of kinesin motor utilization. Annu Rev Cell Dev Biol. 1999;15:141-183. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 196] [Cited by in F6Publishing: 211] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 37. | Schroer TA. Motors, clutches and brakes for membrane traffic: a commemorative review in honor of Thomas Kreis. Traffic. 2000;1:3-10. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 27] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 38. | López de Heredia M, Jansen RP. mRNA localization and the cytoskeleton. Curr Opin Cell Biol. 2004;16:80-85. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 99] [Cited by in F6Publishing: 106] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 39. | Wells AL, Lin AW, Chen LQ, Safer D, Cain SM, Hasson T, Carragher BO, Milligan RA, Sweeney HL. Myosin VI is an actin-based motor that moves backwards. Nature. 1999;401:505-508. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 539] [Cited by in F6Publishing: 555] [Article Influence: 22.2] [Reference Citation Analysis (0)] |
| 40. | Buss F, Arden SD, Lindsay M, Luzio JP, Kendrick-Jones J. Myosin VI isoform localized to clathrin-coated vesicles with a role in clathrin-mediated endocytosis. EMBO J. 2001;20:3676-3684. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 236] [Cited by in F6Publishing: 224] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 41. | Buss F, Luzio JP, Kendrick-Jones J. Myosin VI, a new force in clathrin mediated endocytosis. FEBS Lett. 2001;508:295-299. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 54] [Cited by in F6Publishing: 57] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 42. | Lapierre LA, Kumar R, Hales CM, Navarre J, Bhartur SG, Burnette JO, Provance DW, Mercer JA, Bähler M, Goldenring JR. Myosin vb is associated with plasma membrane recycling systems. Mol Biol Cell. 2001;12:1843-1857. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 332] [Cited by in F6Publishing: 325] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 43. | Altschuler Y, Hodson C, Milgram SL. The apical compartment: trafficking pathways, regulators and scaffolding proteins. Curr Opin Cell Biol. 2003;15:423-429. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 48] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 44. | Rogers SL, Gelfand VI. Myosin cooperates with microtubule motors during organelle transport in melanophores. Curr Biol. 1998;8:161-164. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 204] [Cited by in F6Publishing: 206] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 45. | Lee C, Chen LB. Dynamic behavior of endoplasmic reticulum in living cells. Cell. 1988;54:37-46. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 365] [Cited by in F6Publishing: 398] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 46. | Terasaki M. Recent progress on structural interactions of the endoplasmic reticulum. Cell Motil Cytoskeleton. 1990;15:71-75. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 68] [Cited by in F6Publishing: 67] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 47. | Sodeik B, Ebersold MW, Helenius A. Microtubule-mediated transport of incoming herpes simplex virus 1 capsids to the nucleus. J Cell Biol. 1997;136:1007-1021. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 519] [Cited by in F6Publishing: 512] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 48. | Karki S, Holzbaur EL. Cytoplasmic dynein and dynactin in cell division and intracellular transport. Curr Opin Cell Biol. 1999;11:45-53. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 372] [Cited by in F6Publishing: 385] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 49. | Aniento F, Emans N, Griffiths G, Gruenberg J. Cytoplasmic dynein-dependent vesicular transport from early to late endosomes. J Cell Biol. 1993;123:1373-1387. [PubMed] [Cited in This Article: ] |
| 50. | Gruenberg J, Griffiths G, Howell KE. Characterization of the early endosome and putative endocytic carrier vesicles in vivo and with an assay of vesicle fusion in vitro. J Cell Biol. 1989;108:1301-1316. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 421] [Cited by in F6Publishing: 446] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 51. | Oda H, Stockert RJ, Collins C, Wang H, Novikoff PM, Satir P, Wolkoff AW. Interaction of the microtubule cytoskeleton with endocytic vesicles and cytoplasmic dynein in cultured rat hepatocytes. J Biol Chem. 1995;270:15242-15249. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 57] [Cited by in F6Publishing: 61] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 52. | Valetti C, Wetzel DM, Schrader M, Hasbani MJ, Gill SR, Kreis TE, Schroer TA. Role of dynactin in endocytic traffic: effects of dynamitin overexpression and colocalization with CLIP-170. Mol Biol Cell. 1999;10:4107-4120. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 226] [Cited by in F6Publishing: 223] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 53. | Leopold PL, Kreitzer G, Miyazawa N, Rempel S, Pfister KK, Rodriguez-Boulan E, Crystal RG. Dynein- and microtubule-mediated translocation of adenovirus serotype 5 occurs after endosomal lysis. Hum Gene Ther. 2000;11:151-165. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 191] [Cited by in F6Publishing: 200] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 54. | Suomalainen M, Nakano MY, Keller S, Boucke K, Stidwill RP, Greber UF. Microtubule-dependent plus- and minus end-directed motilities are competing processes for nuclear targeting of adenovirus. J Cell Biol. 1999;144:657-672. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 353] [Cited by in F6Publishing: 351] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 55. | Suomalainen M, Nakano MY, Boucke K, Keller S, Greber UF. Adenovirus-activated PKA and p38/MAPK pathways boost microtubule-mediated nuclear targeting of virus. EMBO J. 2001;20:1310-1319. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 183] [Cited by in F6Publishing: 183] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 56. | Suikkanen S, Aaltonen T, Nevalainen M, Välilehto O, Lindholm L, Vuento M, Vihinen-Ranta M. Exploitation of microtubule cytoskeleton and dynein during parvoviral traffic toward the nucleus. J Virol. 2003;77:10270-10279. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 82] [Cited by in F6Publishing: 89] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 57. | Vihinen-Ranta M, Wang D, Weichert WS, Parrish CR. The VP1 N-terminal sequence of canine parvovirus affects nuclear transport of capsids and efficient cell infection. J Virol. 2002;76:1884-1891. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 109] [Cited by in F6Publishing: 110] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 58. | Vihinen-Ranta M, Yuan W, Parrish CR. Cytoplasmic trafficking of the canine parvovirus capsid and its role in infection and nuclear transport. J Virol. 2000;74:4853-4859. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 54] [Cited by in F6Publishing: 55] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 59. | Suikkanen S, Sääjärvi K, Hirsimäki J, Välilehto O, Reunanen H, Vihinen-Ranta M, Vuento M. Role of recycling endosomes and lysosomes in dynein-dependent entry of canine parvovirus. J Virol. 2002;76:4401-4411. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 54] [Cited by in F6Publishing: 59] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 60. | Döhner K, Wolfstein A, Prank U, Echeverri C, Dujardin D, Vallee R, Sodeik B. Function of dynein and dynactin in herpes simplex virus capsid transport. Mol Biol Cell. 2002;13:2795-2809. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 244] [Cited by in F6Publishing: 243] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 61. | van Loo ND, Fortunati E, Ehlert E, Rabelink M, Grosveld F, Scholte BJ. Baculovirus infection of nondividing mammalian cells: mechanisms of entry and nuclear transport of capsids. J Virol. 2001;75:961-970. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 140] [Cited by in F6Publishing: 142] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 62. | Vihinen-Ranta M, Kalela A, Mäkinen P, Kakkola L, Marjomäki V, Vuento M. Intracellular route of canine parvovirus entry. J Virol. 1998;72:802-806. [PubMed] [Cited in This Article: ] |
| 63. | Funk A, Mhamdi M, Lin L, Will H, Sirma H. Itinerary of hepatitis B viruses: delineation of restriction points critical for infectious entry. J Virol. 2004;78:8289-8300. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 38] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 64. | Köck J, Borst EM, Schlicht HJ. Uptake of duck hepatitis B virus into hepatocytes occurs by endocytosis but does not require passage of the virus through an acidic intracellular compartment. J Virol. 1996;70:5827-5831. [PubMed] [Cited in This Article: ] |
| 65. | Christensen J, Cotmore SF, Tattersall P. A novel cellular site-specific DNA-binding protein cooperates with the viral NS1 polypeptide to initiate parvovirus DNA replication. J Virol. 1997;71:1405-1416. [PubMed] [Cited in This Article: ] |
| 66. | Cotmore SF, D'abramo AM, Ticknor CM, Tattersall P. Controlled conformational transitions in the MVM virion expose the VP1 N-terminus and viral genome without particle disassembly. Virology. 1999;254:169-181. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 105] [Cited by in F6Publishing: 105] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 67. | Rabe B, Glebe D, Kann M. Lipid-mediated introduction of hepatitis B virus capsids into nonsusceptible cells allows highly efficient replication and facilitates the study of early infection events. J Virol. 2006;80:5465-5473. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 56] [Cited by in F6Publishing: 66] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 68. | Panté N, Kann M. Nuclear pore complex is able to transport macromolecules with diameters of about 39 nm. Mol Biol Cell. 2002;13:425-434. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 595] [Cited by in F6Publishing: 557] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 69. | Brandenburg B, Stockl L, Gutzeit C, Roos M, Lupberger J, Schwartlander R, Gelderblom H, Sauer IM, Hofschneider PH, Hildt E. A novel system for efficient gene transfer into primary human hepatocytes via cell-permeable hepatitis B virus-like particle. Hepatology. 2005;42:1300-1309. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 30] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 70. | Futaki S. Oligoarginine vectors for intracellular delivery: design and cellular-uptake mechanisms. Biopolymers. 2006;84:241-249. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 156] [Cited by in F6Publishing: 152] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 71. | Cooper A, Shaul Y. Clathrin-mediated endocytosis and lysosomal cleavage of hepatitis B virus capsid-like core particles. J Biol Chem. 2006;281:16563-16569. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 55] [Cited by in F6Publishing: 59] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 72. | Cronshaw JM, Krutchinsky AN, Zhang W, Chait BT, Matunis MJ. Proteomic analysis of the mammalian nuclear pore complex. J Cell Biol. 2002;158:915-927. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 759] [Cited by in F6Publishing: 727] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 73. | Reichelt R, Holzenburg A, Buhle EL, Jarnik M, Engel A, Aebi U. Correlation between structure and mass distribution of the nuclear pore complex and of distinct pore complex components. J Cell Biol. 1990;110:883-894. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 349] [Cited by in F6Publishing: 411] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 74. | Lim RY, Aebi U, Stoffler D. From the trap to the basket: getting to the bottom of the nuclear pore complex. Chromosoma. 2006;115:15-26. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 37] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 75. | Görlich D, Kutay U. Transport between the cell nucleus and the cytoplasm. Annu Rev Cell Dev Biol. 1999;15:607-660. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1581] [Cited by in F6Publishing: 1566] [Article Influence: 62.6] [Reference Citation Analysis (0)] |
| 76. | Paine PL, Moore LC, Horowitz SB. Nuclear envelope permeability. Nature. 1975;254:109-114. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 553] [Cited by in F6Publishing: 608] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 77. | Ström AC, Weis K. Importin-beta-like nuclear transport receptors. Genome Biol. 2001;2:REVIEWS3008. [PubMed] [Cited in This Article: ] |
| 78. | Kalderon D, Richardson WD, Markham AF, Smith AE. Sequence requirements for nuclear location of simian virus 40 large-T antigen. Nature. 1984;311:33-38. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 945] [Cited by in F6Publishing: 1012] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 79. | Yao E, Gong Y, Chen N, Tavis JE. The majority of duck hepatitis B virus reverse transcriptase in cells is nonencapsidated and is bound to a cytoplasmic structure. J Virol. 2000;74:8648-8657. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 31] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 80. | Kann M, Bischof A, Gerlich WH. In vitro model for the nuclear transport of the hepadnavirus genome. J Virol. 1997;71:1310-1316. [PubMed] [Cited in This Article: ] |
| 81. | Beck J, Nassal M. Efficient Hsp90-independent in vitro activation by Hsc70 and Hsp40 of duck hepatitis B virus reverse transcriptase, an assumed Hsp90 client protein. J Biol Chem. 2003;278:36128-36138. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 79] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 82. | Hu J, Anselmo D. In vitro reconstitution of a functional duck hepatitis B virus reverse transcriptase: posttranslational activation by Hsp90. J Virol. 2000;74:11447-11455. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 76] [Cited by in F6Publishing: 83] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 83. | Kose S, Furuta M, Koike M, Yoneda Y, Imamoto N. The 70-kD heat shock cognate protein (hsc70) facilitates the nuclear export of the import receptors. J Cell Biol. 2005;171:19-25. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 84. | Mandell RB, Feldherr CM. The effect of carboxyl-terminal deletions on the nuclear transport rate of rat hsc70. Exp Cell Res. 1992;198:164-169. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 16] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 85. | Ojala PM, Sodeik B, Ebersold MW, Kutay U, Helenius A. Herpes simplex virus type 1 entry into host cells: reconstitution of capsid binding and uncoating at the nuclear pore complex in vitro. Mol Cell Biol. 2000;20:4922-4931. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 203] [Cited by in F6Publishing: 201] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 86. | Greber UF, Suomalainen M, Stidwill RP, Boucke K, Ebersold MW, Helenius A. The role of the nuclear pore complex in adenovirus DNA entry. EMBO J. 1997;16:5998-6007. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 229] [Cited by in F6Publishing: 242] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 87. | Granzow H, Weiland F, Jöns A, Klupp BG, Karger A, Mettenleiter TC. Ultrastructural analysis of the replication cycle of pseudorabies virus in cell culture: a reassessment. J Virol. 1997;71:2072-2082. [PubMed] [Cited in This Article: ] |
| 88. | Saphire AC, Guan T, Schirmer EC, Nemerow GR, Gerace L. Nuclear import of adenovirus DNA in vitro involves the nuclear protein import pathway and hsc70. J Biol Chem. 2000;275:4298-4304. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 106] [Cited by in F6Publishing: 112] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 89. | Adam SA, Marr RS, Gerace L. Nuclear protein import in permeabilized mammalian cells requires soluble cytoplasmic factors. J Cell Biol. 1990;111:807-816. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 695] [Cited by in F6Publishing: 751] [Article Influence: 22.1] [Reference Citation Analysis (0)] |
| 90. | Rabe B, Vlachou A, Panté N, Helenius A, Kann M. Nuclear import of hepatitis B virus capsids and release of the viral genome. Proc Natl Acad Sci USA. 2003;100:9849-9854. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 220] [Cited by in F6Publishing: 194] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 91. | Zlotnick A, Cheng N, Stahl SJ, Conway JF, Steven AC, Wingfield PT. Localization of the C terminus of the assembly domain of hepatitis B virus capsid protein: implications for morphogenesis and organization of encapsidated RNA. Proc Natl Acad Sci USA. 1997;94:9556-9561. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 142] [Cited by in F6Publishing: 148] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 92. | Kann M, Sodeik B, Vlachou A, Gerlich WH, Helenius A. Phosphorylation-dependent binding of hepatitis B virus core particles to the nuclear pore complex. J Cell Biol. 1999;145:45-55. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 182] [Cited by in F6Publishing: 184] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 93. | Vlachou A. Untersuchung von Wechselwirkungen zwischen Hepatitis B Virus Nukleocapsiden und dem Zellkern. Biologie: Giessen 1999; . [Cited in This Article: ] |
| 94. | Gazina EV, Fielding JE, Lin B, Anderson DA. Core protein phosphorylation modulates pregenomic RNA encapsidation to different extents in human and duck hepatitis B viruses. J Virol. 2000;74:4721-4728. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 97] [Cited by in F6Publishing: 98] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 95. | Melegari M, Wolf SK, Schneider RJ. Hepatitis B virus DNA replication is coordinated by core protein serine phosphorylation and HBx expression. J Virol. 2005;79:9810-9820. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 92] [Cited by in F6Publishing: 109] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 96. | Untergasser A, Zedler U, Langenkamp A, Hösel M, Quasdorff M, Esser K, Dienes HP, Tappertzhofen B, Kolanus W, Protzer U. Dendritic cells take up viral antigens but do not support the early steps of hepatitis B virus infection. Hepatology. 2006;43:539-547. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 85] [Cited by in F6Publishing: 94] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 97. | Civitico GM, Locarnini SA. The half-life of duck hepatitis B virus supercoiled DNA in congenitally infected primary hepatocyte cultures. Virology. 1994;203:81-89. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 61] [Cited by in F6Publishing: 62] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 98. | Zhu Y, Yamamoto T, Cullen J, Saputelli J, Aldrich CE, Miller DS, Litwin S, Furman PA, Jilbert AR, Mason WS. Kinetics of hepadnavirus loss from the liver during inhibition of viral DNA synthesis. J Virol. 2001;75:311-322. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 186] [Cited by in F6Publishing: 186] [Article Influence: 8.1] [Reference Citation Analysis (0)] |