Copyright
©The Author(s) 2018.
World J Gastroenterol. Sep 7, 2018; 24(33): 3738-3748
Published online Sep 7, 2018. doi: 10.3748/wjg.v24.i33.3738
Published online Sep 7, 2018. doi: 10.3748/wjg.v24.i33.3738
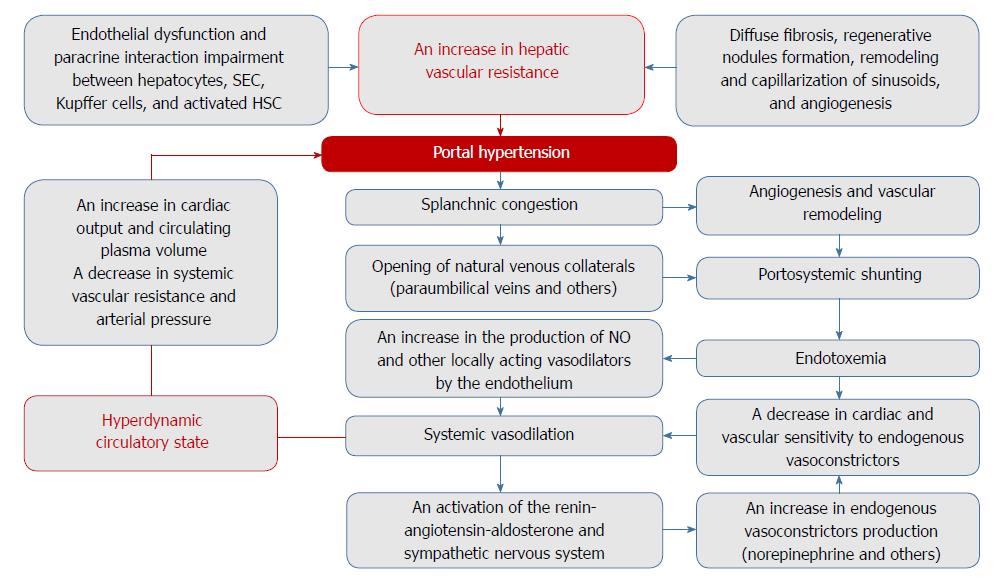
Figure 1 Potential mechanisms of portal hypertension pathogenesis in cirrhosis.
The newly formed blood vessels, which bypass sinusoids in response to the gross morphofunctional rearrangement of the liver in cirrhosis, fail to provide oxygen and nutrients to the tissues. With endothelial dysfunction and impaired paracrine interaction between hepatocytes, sinusoidal endothelial cells (SEC), Kupffer cells, and activated hepatic stellate cells (HSC), this increases hepatic vascular resistance to portal blood flow. Further progression of portal hypertension is a consequence of complex processes including angiogenesis, vascular remodeling, and endothelial dysfunction, which contribute to splanchnic congestion, systemic vasodilation, and portosystemic shunt formation. The subsequent hyperdynamic circulatory state worsens the course of the disease.
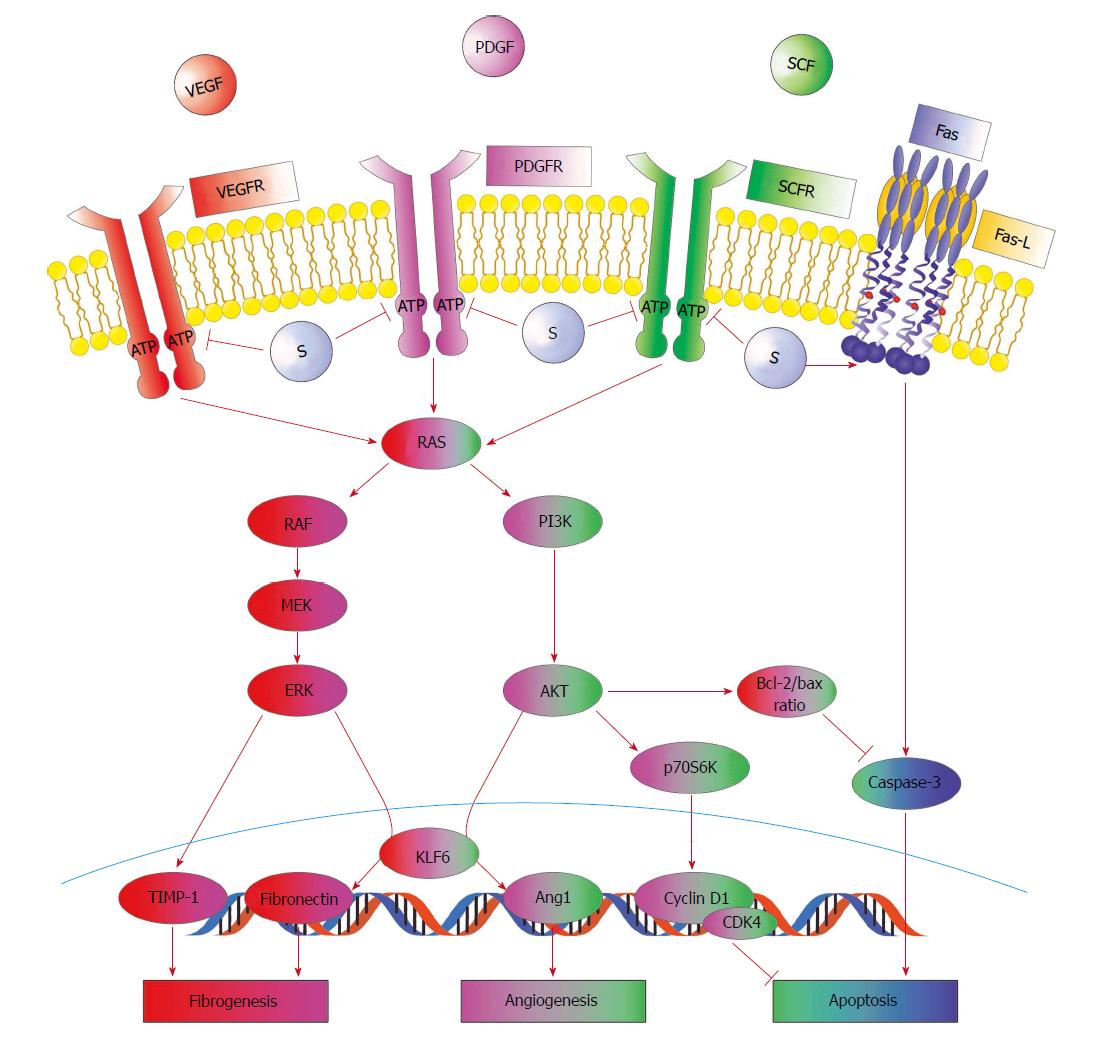
Figure 2 Positive effects of sorafenib on some pathogenetic pathways of fibrogenesis and angiogenesis in the liver.
Sorafenib (S) blocks the ATP-binding site of the vascular endothelial growth factor receptor (VEGFR), platelet-derived growth factor receptor (PDGFR), and stem cell growth factor receptor (SCFR) tyrosine kinases located on the surface of hepatic stellate cells (HSC), inhibiting the two main cellular pathways of the RAS protein. At the same time, sorafenib increases the expression of Fas and its ligand. This decreases the severity of fibrogenesis and angiogenesis and increases apoptosis, leading to a reduction in hepatic vascular resistance to portal blood flow.
- Citation: Garbuzenko DV, Arefyev NO, Kazachkov EL. Antiangiogenic therapy for portal hypertension in liver cirrhosis: Current progress and perspectives. World J Gastroenterol 2018; 24(33): 3738-3748
- URL: https://www.wjgnet.com/1007-9327/full/v24/i33/3738.htm
- DOI: https://dx.doi.org/10.3748/wjg.v24.i33.3738