Revised: December 13, 2012
Accepted: January 23, 2013
Published online: March 26, 2013
Brugada syndrome (BrS) is a rare and inherited primary arrhythmic syndrome characterized by ST-segment elevations in the right precordial leads (V1-V3) with an increased risk of sudden cardiac death (SCD). Arrhythmias in BrS are often nocturne, and brady-arrhythmias are often seen in patients with loss-of-function mutations in SCN5A. In this case-report we present a 75-year old woman referred to our outpatient clinic for inherited cardiac diseases for a familial clinical work-up. Since childhood she had suffered from dizziness, absence seizures, and countless Syncope’s. In 2004 sick sinus syndrome was suspected and she was treated with implantation of a pacemaker (PM) at another institution. An inherited cardiac disease was one day suddenly suspected, as the patient had a 61-year old brother who was diagnosed with symptomatic BrS, and treated with an implantable cardioverter defibrillator (ICD) after aborted SCD. A mutation screening revealed a SCN5A [S231CfsX251 (c.692-693delCA)] loss-of-function mutation not previously reported, and as a part of the cascade screening in relatives she was therefore referred to our clinic. In the 7 year period after PM implantation she had experienced no cardiac symptoms, although her electrocardiogram changes now were consistent with a BrS type 1 pattern. A genetic test confirmed that she had the same mutation in SCN5A as her brother. In this case-report we present a loss-of function mutation in SCN5A not previously associated with BrS nor presented in healthy controls. Sinus node dysfunction has previously been documented in patients with symptomatic BrS, which suggests it is not a rare concomitant. The only accepted treatment of BrS is today implantation of an ICD. In the future studies should evaluate if PM in some cases of symptomatic BrS can be used instead of ICDs in patients with a loss-of-function SCN5A mutations
- Citation: Risgaard B, Bundgaard H, Jabbari R, Haunsø S, Winkel BG, Tfelt-Hansen J. Pacemaker implantation in a patient with brugada and sick sinus syndrome. World J Cardiol 2013; 5(3): 65-67
- URL: https://www.wjgnet.com/1949-8462/full/v5/i3/65.htm
- DOI: https://dx.doi.org/10.4330/wjc.v5.i3.65
Brugada syndrome (BrS) is a rare and inherited primary arrhythmic syndrome characterized by ST-segment elevations in the right precordial leads (V1-V3) with an increased risk of sudden cardiac death (SCD) due to malignant ventricular arrhythmias in the absence of a structural heart disease[1]. Arrhythmias in BrS are often nocturne, and brady-arrhythmias are often seen in patients with loss-of-function mutations in the Sodium channel gene (SCN5A)[2]. SCN5A encodes the alpha subunit (Nav1.5) of the cardiac sodium channel complex and it is the only recommended gene for targeted screening in BrS[3]. Mutations in SCN5A have been reported to be associated with several other types of disease entities such as lone atrial fibrillation and the Long QT Syndrome[4]. It is suggested that the loss-of-function mutations in SCN5A create the substrate for a re-entry circuit in the ventricular myocardium, but may also increase vagal activity, thus facilitating development of arrhythmias[1].
A 75-year old woman was in 2011 referred to our outpatient clinic for inherited cardiac diseases for a familial clinical work-up. Since childhood she had suffered from dizziness, absence seizures, and countless syncope’s. In 2004 sick sinus syndrome was suspected as a 24 h Holter monitoring revealed 67 sinus arrests [electrocardiogram (ECG) not available] of more than 2.5 s with the longest being 6.85 s. The asystole were followed by dizziness but no syncope’s appeared under surveillance. The SSS diagnosis seemed obvious, and she was treated with implantation of a pacemaker (PM) at another institution.
An inherited cardiac disease was one day suddenly suspected, as the patient had a 61-year old brother who was diagnosed with symptomatic BrS, and treated with an implantable cardioverter defibrillator (ICD) after aborted SCD. A mutation screening revealed a SCN5A [S231CfsX251 (c.692-693delCA)] loss-of-function mutation not previously reported, and as a part of the cascade screening in relatives she was therefore referred to our clinic.
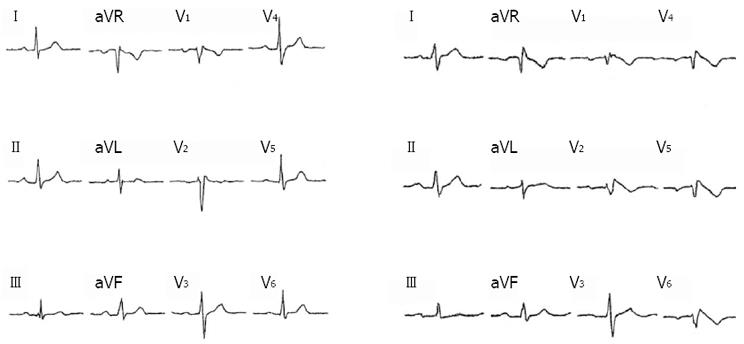
In the 7 year period after PM implantation she had experienced no cardiac symptoms. An ECG and elevated electrode placement ECG (EEP-ECG) showed no BrS pattern. However, BrS was suspected due to the family history and a Flecanide test (150 mg intravenously) was performed. After infusion, 1 and 2 mm ST elevations appeared in V1 and V2, respectively (Figure 1). Changes were consistent with a BrS type 1 ECG pattern and a genetic test confirmed that she had the same mutation in SCN5A as her brother. As the patient had been free of symptoms after PM implantation, it was decided not to upgrade to an ICD unless syncope would re-appear.
In this case-report we present a loss-of function mutation in SCN5A not previously associated with BrS nor presented in healthy controls[5]. It has been suggested that the loss-of-function mutations in SCN5A create the substrate for a re-entry circuit in the ventricular myocardium, but may also increase vagal activity, thus facilitating development of arrhythmias in BrS[1]. The mutation, that we report, was initially found in a brother diagnosed with BrS after aborted SCD and hereafter in our patient, initially diagnosed with SSS. Sinus node dysfunction has previously been documented in patients with symptomatic BrS, which suggests it is not a rare concomitant[6,7]. However, PM implant in this case kept the patient free of symptoms for several years, and this support the theory that increased vagal tonus may cause bradycardia-related arrhythmias which in other isolated cases have been treated successfully with rapid pacing as well[1]. The only accepted treatment of BrS is today implantation of an ICD. In the future studies should evaluate if PM in some cases of symptomatic BrS can be used instead of ICDs in patients with ICDs in patients with SCN5A loss-of-function mutations.
P- Reviewer Chahine M S- Editor Zhai HH L- Editor A E- Editor Zhang DN
| 1. | Antzelevitch C. Brugada syndrome. Pacing Clin Electrophysiol. 2006;29:1130-1159. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 262] [Cited by in F6Publishing: 273] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 2. | Kasanuki H, Ohnishi S, Ohtuka M, Matsuda N, Nirei T, Isogai R, Shoda M, Toyoshima Y, Hosoda S. Idiopathic ventricular fibrillation induced with vagal activity in patients without obvious heart disease. Circulation. 1997;95:2277-2285. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 258] [Cited by in F6Publishing: 271] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 3. | Ackerman MJ, Priori SG, Willems S, Berul C, Brugada R, Calkins H, Camm AJ, Ellinor PT, Gollob M, Hamilton R. HRS/EHRA expert consensus statement on the state of genetic testing for the channelopathies and cardiomyopathies this document was developed as a partnership between the Heart Rhythm Society (HRS) and the European Heart Rhythm Association (EHRA). Heart Rhythm. 2011;8:1308-1339. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 761] [Cited by in F6Publishing: 724] [Article Influence: 60.3] [Reference Citation Analysis (0)] |
| 4. | Tfelt-Hansen J, Winkel BG, Grunnet M, Jespersen T. Inherited cardiac diseases caused by mutations in the Nav1.5 sodium channel. J Cardiovasc Electrophysiol. 2010;21:107-115. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 63] [Cited by in F6Publishing: 65] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 5. | Exome Variant Server. 2012. Available from: http: //evs.gs.washington.edu/EVS/(Accessed 2012; July 1). [Cited in This Article: ] |
| 6. | Sumiyoshi M, Nakazato Y, Tokano T, Yasuda M, Mineda Y, Nakata Y, Daida H. Sinus node dysfunction concomitant with Brugada syndrome. Circ J. 2005;69:946-950. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 7. | Shimizu N, Iwamoto M, Nakano Y, Sumita S, Ishikawa T, Hokosaki T, Akaike T, Nishizawa T, Takigiku K, Shibata T. Long-term electrocardiographic follow-up from childhood of an adult patient with Brugada syndrome associated with sick sinus syndrome. Circ J. 2009;73:575-579. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7] [Cited by in F6Publishing: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |