Published online Jul 15, 2015. doi: 10.4251/wjgo.v7.i7.87
Peer-review started: March 21, 2015
First decision: April 10, 2015
Revised: May 14, 2015
Accepted: June 1, 2015
Article in press: June 2, 2015
Published online: July 15, 2015
AIM: To investigate the role of endothelial nitric oxide synthase -786T > C promoter polymorphism in the etiology of gastric cancer (GC).
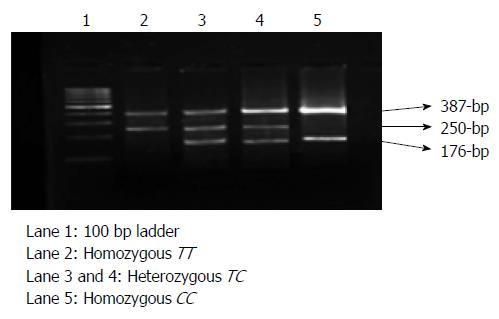
METHODS: A total of 150 GC patients and 150 control subjects were included in the study. The information on demographic features was elicited with an informed consent from all the patients and control subjects using a structured questionnaire. Helicobacter pylori (H. pylori) infectivity status was tested in antral biopsies from all the subjects by rapid urease test following the method of Vaira et al. Genomic DNA was isolated from whole blood samples following the salting out method of Lahiri et al. Genotype analysis of the rs2070744 polymorphism was carried out by allele-specific polymerase chain reaction method. The genotypes were determined based on the appearance of bands on an agarose gel stained with ethidium bromide under ultraviolet gel documentation with the help of 100 bp ladder. Odds ratios and corresponding 95%CIs were determined using java stat online software.
RESULTS: There was a significant difference in the distribution of C allele (C vs T; P = 0.000, OR = 5.038) in patient group compared to the control subjects exhibiting a fivefold increased risk for GC. When the T/T and C/C genotypes were compared, there was an enhanced GC risk for individuals with C/C genotype (T/T vs C/C; P = 0.000). Among the demographic factors, smoking and alcoholism were the common risk factors in patients compared to the control subjects (P < 0.05). Patients with smoking and alcoholism developed cancer even in heterozygous T/C condition (smoking: P = 0.020 and alcoholism: P = 0.005). Individuals with H. pylori infection showed seven fold increased risk for cancer. All the patients with C/C genotype revealed a significant association between H. pylori infection and GC. Among the patients 2.4% of them revealed familial incidence of GC. No significant difference was noticed between cases and controls with regard to consanguinity (P = 0.473).
CONCLUSION: The Present data suggest that eNOS-786 C/C genotype and C allele may be considered as potential risk factors in patients with GC.
Core tip: The present study reveals first molecular epidemiological evidence from south Indian cohort for the association of endothelial nitric oxide synthase -786T > C promoter polymorphism with a risk to develop gastric cancer (GC). The CC genotype and C allele of the -786T > C polymorphism were significantly associated with an elevated risk to GC, probably due to the lowered nitric oxide levels in case of C/C genotype which result in tumour proliferation, angiogenesis and metastasis.
-
Citation: Krishnaveni D, Amar Chand B, Shravan Kumar P, Uma Devi M, Ramanna M, Jyothy A, Pratibha N, Balakrishna N, Venkateshwari A. Association of
endothelial nitric oxide synthase gene T-786C promoter polymorphism with gastric cancer. World J Gastrointest Oncol 2015; 7(7): 87-94 - URL: https://www.wjgnet.com/1948-5204/full/v7/i7/87.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v7.i7.87
Gastric cancer (GC), also known as stomach cancer, is the accumulation of malignant cells that form a tumour in any part of the stomach. In most cases, it is adenocarcinoma, starts off in the mucus producing cells present in the inner most lining of the stomach. On the whole 90% of the stomach tumours are of malignant and 95% of these tumours reported to be adenocarcinomas[1]. GC is defined as a multifactorial disorder resulting from various genetic, epigenetic predisposition and environmental risk factors[2]. The incidence and mortality rates of GC vary by ethnicity and sex. On global scale, GC causes approximately 800000 deaths per year and it is the third leading cause of cancer death worldwide in both males and females[3]. A recent study from Tata Memorial Centre (TMC) on cancer mortality in India has rightly focused GC as the second largest cause of cancer related deaths among Indians[4].
Nitric oxide (NO) is a short lived vasoactive substance of prime importance constitutively produced from L - arginine by the enzyme nitric oxide synthase (NOS)[5]. Four isoforms of NOS have been identified and described as inducible nitric oxide synthase (iNOS), neuronal nitric oxide synthase (nNOS), endothelial nitric oxide synthase (eNOS) and mitochondrial nitric oxide synthase (mNOS)[6]. The eNOS gene has a pivotal role in the maintenance of stress balance because of its ability to generate nitric oxide (NO) and this feature of the gene makes it a logical candidate gene for various cancers[7]. Human eNOS is encoded by NOS3 gene comprised of 26 exons with a total size of 21 kb and mapped on to chromosome 7q35 to 36[8]. The NOS3 gene transcribes an mRNA of 4052 nucleotides which translates 135-KD protein containing 1203 amino acids[9].
NO is a hydrophobic, highly diffusive and small pleiotropic free radical, acting as a signaling molecule in various inflammatory diseases and cancers[10]. It is reported to cause DNA damage in the course of nitration, deamination, nitrosation, and inhibit sealing activity of DNA ligase, facilitating the accumulation of breaks in DNA and promote tumor proliferation, angiogenesis and metastasis[11,12].
A single nucleotide polymorphism (T > C) rs2070744 due to transition of a thymine to a cytosine at -786 in the promoter region of eNOS was found to reduce the rate of mRNA transcription by 50%, resulting in decreased serum NO levels which can inhibit apoptosis or stimulate tumour proliferation, angiogenesis and metastasis[13]. These effects might depend upon the fact that a mutant allele can bind the replication protein A1, which acts as a gene repressor protein[14]. The -786T > C promoter polymorphism has been reported to play very important role in various cardiovascular diseases, hypertension and diabetic neuropathy[15-17]. The genetic-epidemiological studies examining the association of T > C promoter polymorphism with gastric cancer (GC) from Indian population were meagre. Hence, the present case-control study was aimed to investigate the association between the eNOS (-786T > C) promoter polymorphism rs2070744 and the risk of GC in south Indian population.
The study included 150 endoscopically and histopathologically confirmed GC patients referred to the Department of Gastroenterology, Gandhi Hospital and Indo-American cancer Hospital and Research Centre, Hyderabad over a period of 3 years. A total of 150 healthy control subjects with no family history of gastric ulcer or cancer were selected randomly among the persons having normal upper gastro intestinal endoscopy report. The information on demographic features such as age, gender, dietary habits, weight, consanguinity, familial incidence of cancer, addiction to smoking and alcohol was elicited with an informed consent from all the patients and control subjects using a structured questionnaire. H. pylori infectivity status was tested in antral biopsies from all the patients by rapid urease test following the method of Vaira et al[18] (1988).
The study was reviewed and approved by Ethics Committee of the Institute in order to conduct the experiments on human subjects and the procedures followed were in accordance with the ethical standards of the committee.
Five millilitres of blood was collected from both patients and control subjects in EDTA coated vacutainers.
DNA Isolation: Genomic DNA was isolated from whole blood samples following the salting out method of Lahiri et al[19] (1991).
Analysis of the eNOS-786T > C promoter polymorphism was carried out by allele -specific polymerase chain reaction method. The oligonucleotide primers used in the reaction were C0: 5’ TTT CTC CAG CCC CTC AGA TG 3’; 2684C: 5’ GGC AGA GGC AGG GTC AGA CG 3’; 2684 T: 5’ CAT CAA GCT CTT CCC TGT CT 3’ and T0: 5’ AGG CCC AGC AAG GAT GTA GT 3’. Amplification was performed in a total volume of 20 μL containing 50 ng genomic DNA, 0.25 μmol/L 2684T and 2684C primers, 0.063 μmol/L T0 and C0 primers, 62.5 μmol/L dNTPs, 1.5 μmol/L MgCl2, and 0.4 U Taq polymerase (Genei Bangalore). After a hot start at 96 °C, amplification was achieved by 35 cycles at 94 °C for 30 s, 60 °C for 30 s, and 72 °C for 20 s.
After performing polymerase chain reaction, the amplicons were resolved on a 3% agarose gel stained with ethidium bromide and visualized under ultraviolet gel documentation (Figure 1). The C and T alleles gave a 176 bp and a 250 bp product, respectively, with a 387 bp common product. The genotypes were determined based on the appearance of bands with the help of 100 bp ladder. Ten percent of the samples were taken randomly, subjected to sequencing and no bias was observed in the genotyping. The study revealed similar findings with 100% concordant results.
The evaluation of case and control differences in the distribution of alleles and genotypes was carried out by Pearson’s χ2 test of association. Odds ratios (ORs) and corresponding 95%CIs were determined using Javastat 2-way Contingency analysis to measure the strength of association between eNOS-786T > C promoter polymorphism and GC[20]. All statistical tests were two-tailed and P-values < 0.05 were considered to be statistically significant. Statistical review of the study was performed by a biomedical statistician.
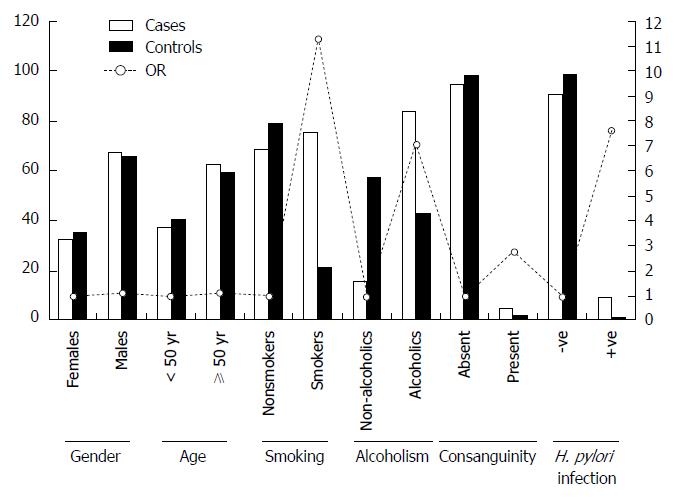
The present case-control study was conducted on a total of 150 patients and 150 control subjects. Mean age of the patients in the study group was 53 years (Mean ± SD = 53 ± 14 years) and that of controls was 50 years (Mean ± SD = 50 ± 11 years). The demographic characteristics of the study population have been represented in Figure 2. All the GC patients and control subjects were of South Indian origin. The study subjects were classified based on demographic factors such as age, gender, addiction to smoking and alcoholism, consanguinity, H. pylori infection, etc. We found no significant difference between cases and controls with regard to gender and age. The risk factor profile exhibited that addiction to smoking and alcohol were the most common risk factors in patients compared to the control subjects (P < 0.05). Individuals with smoking exhibited eleven fold increased risk, where as those with alcoholism revealed seven fold increased risk of developing GC. Individuals with H. pylori infection showed 7.6 fold increased risk for cancer. Among the patients 2.4% of them revealed familial incidence of GC. No significant difference was noticed between cases and controls with regard to consanguinity (P = 0.473).
The distribution of genotype and allele frequencies of eNOS-786T > C promoter polymorphism in patients and controls were given in Table 1. The frequency of T/T, T/C and C/C genotypes in patients were 2%, 8% and 90%, where as in controls the distribution was 14.67%, 19.33% and 66%, respectively. The allelic frequencies were found to be 6% of T and 94% of C in patient group, where as 24.33% and 75.67% in controls, respectively.
| Genotype | Patients (n = 150) | Controls (n = 150) | Odds ratio | P value | ||
| n | % | n | % | (95%CI) | ||
| T/T | 3 | 2.00 | 22 | 14.67 | Reference | |
| T/C | 12 | 8.00 | 29 | 19.33 | 3.034 (0.67-15.53) | 0.136 |
| C/C | 135 | 90.00 | 99 | 66.00 | 10.00 (2.73-43.24) | 0.000a |
| T/C + C/C | 147 | 98.00 | 128 | 85.33 | 8.42 (2.32-36.2) | 0.000a |
| T/T + T/C vs C/C | 15 | 10.00 | 51 | 34.00 | 4.636 (2.37-9.17) | 0.000a |
| Alleles | ||||||
| T | 18 | 6.00 | 73 | 24.33 | Reference | |
| C | 282 | 94.00 | 227 | 75.67 | 5.038 (2.841-9.026) | 0.000a |
The allelic frequencies were compared in order to assess the risk ratio based on the type of allele present in the individuals. There was a significant difference in the distribution of C allele (C vs T; P = 0.000, OR = 5.038) in patient group compared to the control subjects (Table 1) exhibiting a five fold increased risk of GC. Further, we compared T/T, T/C and C/C genotypes against each other in various combinations. When the T/T and C/C genotypes were compared, there was ten fold enhanced GC risk for individuals with C/C genotype (T/T vs C/C; P = 0.000). In addition, we compared T/T vs T/C + C/C genotypes as well as T/T + T/C vs C/C genotypes and found a significant association with the disease, exhibiting 8.4 fold and 4.6 fold increased risk, respectively, in patients compared to the controls (T/T + T/C vs C/C; P = 0.000, OR = 4.636, and T/T vs T/C + C/C; P = 0.000, OR = 8.42).
Table 2 exhibits the genotype distribution based on H. pylori infection. The H. pylori +ve and -ve individuals in cases and controls were compared separately and observed no association of T/T and T/C genotypes, but all the patients with C/C genotype revealed a significant association between H. pylori infection and GC.
| Characteristic | Controls n (%) | Cases n (%) | χ2 | OR (95%CI) | P value |
| H. pylori infection | |||||
| T/T genotype | |||||
| +ve | 1 (33.33) | 0 (0.00) | |||
| -ve | 2 (66.67) | 22 (100.00) | 1.424 | - | 0.120 |
| T/C genotype | |||||
| +ve | 3 (25.00) | 1 (3.45) | |||
| -ve | 9 (75.00) | 28 (96.55) | 2.364 | 9.333 (0.699-267.06) | 0.068 |
| C/C genotype | |||||
| +ve | 10 (7.41) | 1 (1.01) | |||
| -ve | 125 (92.59) | 98 (98.99) | 3.887 | 7.840 (1.008-166.50) | 0.027a |
Table 3 stratifies the genotype distribution based on addictions like smoking and alcoholism. Individuals having the habit of smoking developed cancer with T/C and C/C genotypes showing six fold and eleven fold augmented risk of GC, respectively. Similarly, those with alcoholism developed GC with T/C and C/C genotypes exhibiting eleven fold and five fold enhanced risk of cancer, respectively. Both smoking and alcoholism did not show any association with T/T genotype. It is very clear from the Table 3 that association of heterozygous condition with disease indicates single C allele in association with addictions is enough to act as a risk allele for developing the disease (smoking: P = 0.020 and alcoholism: P = 0.005).
| Characteristic | Cases n (%) | Controls n (%) | χ2 | OR (95%CI) | P value |
| Smoking status | |||||
| T/T genotype | |||||
| Smokers | 2 (66.67) | 4 (18.18) | |||
| Nonsmokers | 1 (33.33) | 18 (81.82) | 1.263 | 9.000 (0.44-339.56) | 0.133 |
| T/C genotype | |||||
| Smokers | 7 (58.33) | 5 (17.24) | |||
| Nonsmokers | 5 (41.67) | 24 (82.76) | 5.080 | 6.720 (1.21-40.80) | 0.020a |
| C/C genotype | |||||
| Smokers | 104 (77.04) | 23 (23.23) | |||
| Nonsmokers | 31 (22.96) | 76 (76.77) | 64.475 | 11.086 (5.74-21.58) | 0.000b |
| Alcoholism | |||||
| T/T genotype | |||||
| Alcoholics | 2 (66.67) | 7 (31.82) | |||
| Non-alcoholics | 1 (33.33) | 15 (68.18) | 0.290 | 4.286 (0.23-145.14) | 0.530 |
| T/C genotype | |||||
| Alcoholics | 10 (83.33) | 9 (31.03) | |||
| Non-alcoholics | 2 (16.67) | 20 (68.97) | 7.351 | 11.11 (1.68-93.07) | 0.005b |
| C/C genotype | |||||
| Alcoholics | 113 (83.70) | 48 (48.48) | |||
| Non-alcoholics | 22 (16.30) | 51 (51.52) | 31.385 | 5.457 (2.86-10.46) | 0.000b |
Gastric cancer, the fifth most frequent cancer in the world and third most in India, is defined as a multifactorial disorder resulting from interaction among distinctive genetic, epigenetic and environmental risk factors[21]. A single nucleotide polymorphism (-786T > C) in eNOS gene promoter rs2070744 demonstrated to play very important role in various cardiovascular diseases, hypertension and diabetic neuropathy and some cancers[7,9].
The polymorphism was found to reduce the transcription rate resulting in decreased serum NO levels which can inhibit apoptosis or stimulate tumour proliferation, angiogenesis and metastasis. The C allele of T > C polymorphism may influence the expression and activity of the NOS enzyme and shown to increase the risk for the development of various diseases[15-17]. There is much more contradiction among the association studies on eNOS T > C promoter polymorphism from divergent ethnic groups. A study by Paradossi et al[22] (2004) revealed no significant association of T > C promoter polymorphism with atherosclerosis in Italian population. An association of C allele and C/C genotype with coronary artery disease (CAD) was demonstrated in the Iranian population by Khaki-Khatibi et al[23] (2013). A hospital-based case-control study by Lu et al[12] (2006) on non-Hispanic white women exhibited a significant association of T > C promoter polymorphism with sporadic breast cancer. In a study by Ghilardi et al[24] (2003), the -786T > C polymorphism was found to be associated with vascular invasion in breast cancer[24]. Another study by Jang et al[25] (2013) on South Korean population highlighted that TC+CC genotype of the -786T > C polymorphism was significantly associated with an increased risk of colorectal cancer. However, the molecular basis for the significant association of C/C genotype of eNOS-786T > C promoter polymorphism remains to be explored.
The present study revealed a statistically significant difference in the distribution of C/C genotype and C allele in GC patients compared to control individuals and is in accordance with the report of Ciftçi et al[26] (2008). A significantly augmented risk of GC was observed in individuals with C/C genotype than those with T/C and T/T genotypes and it is evident from the literature that C/C genotype showed down regulation of eNOS transcription yielding decreased NO levels and ultimately inhibit apoptosis or stimulate tumor proliferation, invasion, angiogenesis and metastasis[13]. It was reported that NO can act as both a pro- and anti-tumorigenic factor. The pro tumorigenic vs anti-tumorigenic effect of NO mainly depends on the genetic profile of the individual, cellular microenvironment, the localization and activity of NOS isoforms, and overall NO levels[27]. In some tumor tissues, NO has been found to enhance tumor angiogenesis and induce vasodilatation, thus accelerating tumor growth[28]. In other tumors, including gastric and colon cancer, a decreased amount of NOS protein was demonstrated by immunohistochemistry, and there was a possible relationship between lowered levels of NO and carcinogenesis[29,30]. Previous studies on GC have demonstrated that the expression of the endothelial NOS, neuronal NOS and inducible NOS in the tumor tissue was significantly lower than in normal gastric mucosa and indicates a marked reduction of all three NOS isoforms expression[29,30]. The function of lowered NO levels in tumor development, promotion, progression and metastasis is still obscure. But it is postulated that reduced NO production in tumors help the tumor cells escape programmed cell death and retain the ability to multiply, ultimately contribute to progression and metastatic potential of the tumor[29].
It should be emphasized that from the present study, the demographic factors revealed a significant association of smoking, alcoholism with GC and may be assigned to environmental risk triggering factors present in alcohol and smoke. The present findings can be correlated with earlier studies which had shown that smoking and alcohol were co - operating in increasing GC risk and interpreted based on the fact that cigarette smoke may enhance the risk to develop GC via the formation of nitroso amine, a potent carcinogen, where as consuming alcohol had impact on tumor volume doubling time (TVDT) invigorating tumor growth by promoting angiogenesis[31,32].
An important finding from the current study is that there is an interplay between addictions such as smoking, alcoholism and the eNOS genotype which could play a critical role in the etiology of GC. Patients having the habit of smoking or alcoholism develop cancer even with a single C allele showing that only one C allele is sufficient to act as a risk allele for developing the disease. This is in accordance with studies of Wang et al[33] (1996) in cardiovascular disorders demonstrating the effect of the eNOS genotype on the risk for the development of disease.
In conclusion, we clearly observed that C allele and CC genotype of the -786T > C polymorphism rs2070744 were significantly associated with etiology of GC and probably due to the lowered NO levels in C/C genotype which may ultimately result in tumor proliferation, angiogenesis and metastasis. To the best of our knowledge, present data provides the first molecular epidemiological evidence from south Indian population for the association of T > C polymorphism with a risk to develop GC. However, a large confirmatory study involving other populations is warranted to understand the population-specificity and the relative contribution of this polymorphism in the disease phenotype.
Gastric cancer is a major cause of cancer mortality worldwide. The etiology of a specific cancer may be associated with a set of genetic variants and their interaction with environmental factors. A single nucleotide polymorphism (SNP) occurs when a normally expected nucleotide (Adenine, Guanine, Thymine or Cytosine) in the genome is replaced by other nucleotide. Analysis of SNPs in cancer research has pleiotropic medical implications for health issues, as well as cancer biology. New findings can help in developing targeted therapies for early intervention and management of cancer.
There has been an increasing focus on the role of SNPs in the development and progression of various cancers and also to assess risk prediction and diagnostics. These SNPs may contribute to differences in disease susceptibility. Analysis and characterization of SNPs as biomarkers in cancer has become the hotspot of current research. The -786T > C polymorphism of the endothelial nitric oxide synthase (eNOS) gene is one among such important polymorphisms in the etiology of gastric cancer (GC).
The eNOS-786T > C promoter polymorphism was reported to play a role in various cardiovascular diseases. Very limited studies were available on the association of T > C promoter polymorphism with cancer and no studies were done from south Indian population. The results obtained from divergent ethnic groups were contradictory for the association of eNOS T > C promoter polymorphism. A study on atherosclerosis revealed no significant association of T > C promoter polymorphism with the disease in Italian population where as another study on colorectal cancer in the South Korean population highlighted that TC + CC genotype of the -786T > C polymorphism was significantly associated with an increased risk of cancer. Present data clearly revealed an association of C allele and C/C genotype with a five fold increased risk of development of GC. Patients with habit of smoking or alcoholism had cancer even with a single C allele showing that only one C allele is able to act as a risk allele for developing the disease. Thus, the present study suggests a strong role of eNOS gene rs2070744 promoter polymorphism in modifying cellular micro environment which in turn facilitate tumour development.
The present study showed that the eNOS -786T > C promoter polymorphism influenced the risk of GC in patients with C allele and CC genotype. The identification of patients with high-risk of GC could help in development of novel treatment strategies and personalized medicine for effective management of the disease.
SNP refer to a DNA sequence variation occurs commonly within a population (approximately 1%) in which a normal nucleotide (Adenine, Guanine, Thymine or Cytosine) in the genome is replaced by another nucleotide that differs between members of a biological species or paired chromosomes. Polymerase chain reaction: The polymerase chain reaction is a technique used in molecular biology to amplify a single copy or a few copies of a piece of DNA, generating thousands to millions of copies of a particular DNA sequence and useful in functional analysis of genes, detection and diagnosis of hereditary and infectious diseases and the identification of genetic fingerprints; Agarose gel electrophoresis is a method used in biochemistry, molecular biology, and clinical chemistry to separate a mixed population of DNA or RNA in a matrix of agarose, based on the size of the DNA/RNA fragments.
The authors have written an interesting paper regarding the role of eNOS-786T > C polymorphism in developing GC in south Indian population and found a significant difference in the distribution of C allele in patient group compared to the control subjects exhibiting a fivefold increased risk for GC. Overall the paper is quite educative giving new information in this issue.
P- Reviewer: Koukourakis GV, Mocellin S, Reeh M, Surlin VM S- Editor: Ji FF L- Editor: A E- Editor: Wang CH
| 1. | Schwartz GK. Invasion and metastases in gastric cancer: in vitro and in vivo models with clinical correlations. Semin Oncol. 1996;23:316-324. [PubMed] [Cited in This Article: ] |
| 2. | Koo SH, Jeong TE, Kang J, Kwon KC, Park JW, Noh SM. Prognostic implications for gastric carcinoma based on loss of heterozygosity genotypes correlation with clinicopathologic variables. Cancer Genet Cytogenet. 2004;153:26-31. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 19] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 3. | Cancer Factsheet. World Health Organization. Accessed 2014 Dec 08. Available from: http://globocan.iarc.fr/Pages/fact sheets_cancer.aspx?cancer=stomach. [Cited in This Article: ] |
| 4. | Stomach Cancer incidence in India. Available from: http://www.dnaindia.com/health/reportstomachcancersecondbiggestkillerinindia1670749. [Cited in This Article: ] |
| 5. | Moncada S, Higgs A. The L-arginine-nitric oxide pathway. N Engl J Med. 1993;329:2002-2012. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 4289] [Cited by in F6Publishing: 4089] [Article Influence: 131.9] [Reference Citation Analysis (0)] |
| 6. | Ying L, Hofseth LJ. An emerging role for endothelial nitric oxide synthase in chronic inflammation and cancer. Cancer Res. 2007;67:1407-1410. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 189] [Cited by in F6Publishing: 201] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 7. | Wu X, Wang ZF, Xu Y, Ren R, Heng BL, Su ZX. Association between three eNOS polymorphisms and cancer risk: a meta-analysis. Asian Pac J Cancer Prev. 2014;15:5317-5324. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 16] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 8. | Marsden PA, Heng HH, Scherer SW, Stewart RJ, Hall AV, Shi XM, Tsui LC, Schappert KT. Structure and chromosomal localization of the human constitutive endothelial nitric oxide synthase gene. J Biol Chem. 1993;268:17478-17488. [PubMed] [Cited in This Article: ] |
| 9. | Syed R, Biyabani MU, Prasad S, Deeba F, Jamil K. Correlation and Identification of Variable number of Tandem repeats of eNOS Gene in Coronary artery disease (CAD). Saudi J Biol Sci. 2010;17:209-213. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 10. | Ignarro LJ. Nitric oxide as a unique signaling molecule in the vascular system: a historical overview. J Physiol Pharmacol. 2002;53:503-514. [PubMed] [Cited in This Article: ] |
| 11. | Koh E, Noh SH, Lee YD, Lee HY, Han JW, Lee HW, Hong S. Differential expression of nitric oxide synthase in human stomach cancer. Cancer Lett. 1999;146:173-180. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 31] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 12. | Lu J, Wei Q, Bondy ML, Yu TK, Li D, Brewster A, Shete S, Sahin A, Meric-Bernstam F, Wang LE. Promoter polymorphism (-786t& gt; C) in the endothelial nitric oxide synthase gene is associated with risk of sporadic breast cancer in non-Hispanic white women age younger than 55 years. Cancer. 2006;107:2245-2253. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 43] [Cited by in F6Publishing: 46] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 13. | Nakayama M, Yasue H, Yoshimura M, Shimasaki Y, Kugiyama K, Ogawa H, Motoyama T, Saito Y, Ogawa Y, Miyamoto Y. T-786--& gt; C mutation in the 5’-flanking region of the endothelial nitric oxide synthase gene is associated with coronary spasm. Circulation. 1999;99:2864-2870. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 544] [Cited by in F6Publishing: 572] [Article Influence: 22.9] [Reference Citation Analysis (0)] |
| 14. | Miyamoto Y, Saito Y, Nakayama M, Shimasaki Y, Yoshimura T, Yoshimura M, Harada M, Kajiyama N, Kishimoto I, Kuwahara K. Replication protein A1 reduces transcription of the endothelial nitric oxide synthase gene containing a -786T--& gt; C mutation associated with coronary spastic angina. Hum Mol Genet. 2000;9:2629-2637. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 170] [Cited by in F6Publishing: 176] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 15. | Casas JP, Cavalleri GL, Bautista LE, Smeeth L, Humphries SE, Hingorani AD. Endothelial nitric oxide synthase gene polymorphisms and cardiovascular disease: a HuGE review. Am J Epidemiol. 2006;164:921-935. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 152] [Cited by in F6Publishing: 160] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 16. | Shankarishan P, Borah PK, Ahmed G, Mahanta J. Endothelial nitric oxide synthase gene polymorphisms and the risk of hypertension in an Indian population. Biomed Res Int. 2014;2014:793040. [PubMed] [Cited in This Article: ] |
| 17. | Shah VN, Cheema BS, Kohli HS, Sharma R, Khullar M, Bhansali A. Endothelial Nitric Oxide Synthase Gene Polymorphism and the Risk of Diabetic Neuropathy in Asian Indian Patients with Type 2 Diabetes. J Diabetes Metab. 2013;4:2. [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 18. | Vaira D, Holton J, Cairns S, Polydorou A, Falzon M, Dowsett J, Salmon PR. Urease tests for Campylobacter pylori: care in interpretation. J Clin Pathol. 1988;41:812-813. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 36] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 19. | Lahiri DK, Nurnberger JI. A rapid non-enzymatic method for the preparation of HMW DNA from blood for RFLP studies. Nucleic Acids Res. 1991;19:5444. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1594] [Cited by in F6Publishing: 1680] [Article Influence: 50.9] [Reference Citation Analysis (0)] |
| 20. | Java stat 2-way Contingency Table Analysis. Accessed 2015 Jan 17. Available from: http://statpages.org/ctab2×2.html. [Cited in This Article: ] |
| 21. | Devulapalli K, Bhayal AC, Porike SK, Macherla R, Akka J, Nallari P, Ananthapur V. Role of interstitial collagenase gene promoter polymorphism in the etiology of gastric cancer. Saudi J Gastroenterol. 2014;20:309-314. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7] [Cited by in F6Publishing: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 22. | Paradossi U, Ciofini E, Clerico A, Botto N, Biagini A, Colombo MG. Endothelial function and carotid intima-media thickness in young healthy subjects among endothelial nitric oxide synthase Glu298--& gt; Asp and T-786--& gt; C polymorphisms. Stroke. 2004;35:1305-1309. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 64] [Cited by in F6Publishing: 71] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 23. | Khaki-Khatibi F, Samadi N, Rahnema B, Zade MA, Nobar MR, Vatankhah A. Association between nucleotide mutation of eNOS gene and serum level of vessel expansion factor (VEF) in non-smoker patients with vascular heart disease. Afr J Biotechnol. 2013;12:3023-3028. [DOI] [Cited in This Article: ] |
| 24. | Ghilardi G, Biondi ML, Cecchini F, DeMonti M, Guagnellini E, Scorza R. Vascular invasion in human breast cancer is correlated to T--& gt; 786C polymorphism of NOS3 gene. Nitric Oxide. 2003;9:118-122. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 39] [Cited by in F6Publishing: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 25. | Jang MJ, Jeon YJ, Kim JW, Chong SY, Hong SP, Oh D, Cho YK, Chung KW, Kim NK. Association of eNOS polymorphisms (-786T& gt; C, 4a4b, 894G& gt; T) with colorectal cancer susceptibility in the Korean population. Gene. 2013;512:275-281. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 26. | Ciftçi C, Melil S, Cebi Y, Ersöz M, Cağatay P, Kiliçgedik M, Duman BS. Association of endothelial nitric oxide synthase promoter region (T-786C) gene polymorphism with acute coronary syndrome and coronary heart disease. Lipids Health Dis. 2008;7:5. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 27. | Lancaster JR, Xie K. Tumors face NO problems? Cancer Res. 2006;66:6459-6462. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 75] [Cited by in F6Publishing: 77] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 28. | Thomsen LL, Miles DW. Role of nitric oxide in tumour progression: lessons from human tumours. Cancer Metastasis Rev. 1998;17:107-118. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 204] [Cited by in F6Publishing: 204] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 29. | Rajnakova A, Goh PM, Chan ST, Ngoi SS, Alponat A, Moochhala S. Expression of differential nitric oxide synthase isoforms in human normal gastric mucosa and gastric cancer tissue. Carcinogenesis. 1997;18:1841-1845. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 45] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 30. | Ambs S, Merriam WG, Bennett WP, Felley-Bosco E, Ogunfusika MO, Oser SM, Klein S, Shields PG, Billiar TR, Harris CC. Frequent nitric oxide synthase-2 expression in human colon adenomas: implication for tumor angiogenesis and colon cancer progression. Cancer Res. 1998;58:334-341. [PubMed] [Cited in This Article: ] |
| 31. | Krishnaveni D, Amar CB, Manjari KS, Vidyasagar A, Uma Devi M, Ramanna M, Jyothy A, Pratibha N, Venkateshwari A. MMP 9 Gene Promoter Polymorphism in Gastric Cancer. Ind J Clin Biochem. 2012;27:259-264. [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 32. | Sung NY, Choi KS, Park EC, Park K, Lee SY, Lee AK, Choi IJ, Jung KW, Won YJ, Shin HR. Smoking, alcohol and gastric cancer risk in Korean men: the National Health Insurance Corporation Study. Br J Cancer. 2007;97:700-704. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 77] [Cited by in F6Publishing: 76] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 33. | Wang XL, Sim AS, Badenhop RF, McCredie RM, Wilcken DE. A smoking-dependent risk of coronary artery disease associated with a polymorphism of the endothelial nitric oxide synthase gene. Nat Med. 1996;2:41-45. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 398] [Cited by in F6Publishing: 409] [Article Influence: 14.6] [Reference Citation Analysis (0)] |