Copyright
©The Author(s) 2025.
World J Hepatol. Aug 27, 2025; 17(8): 107456
Published online Aug 27, 2025. doi: 10.4254/wjh.v17.i8.107456
Published online Aug 27, 2025. doi: 10.4254/wjh.v17.i8.107456
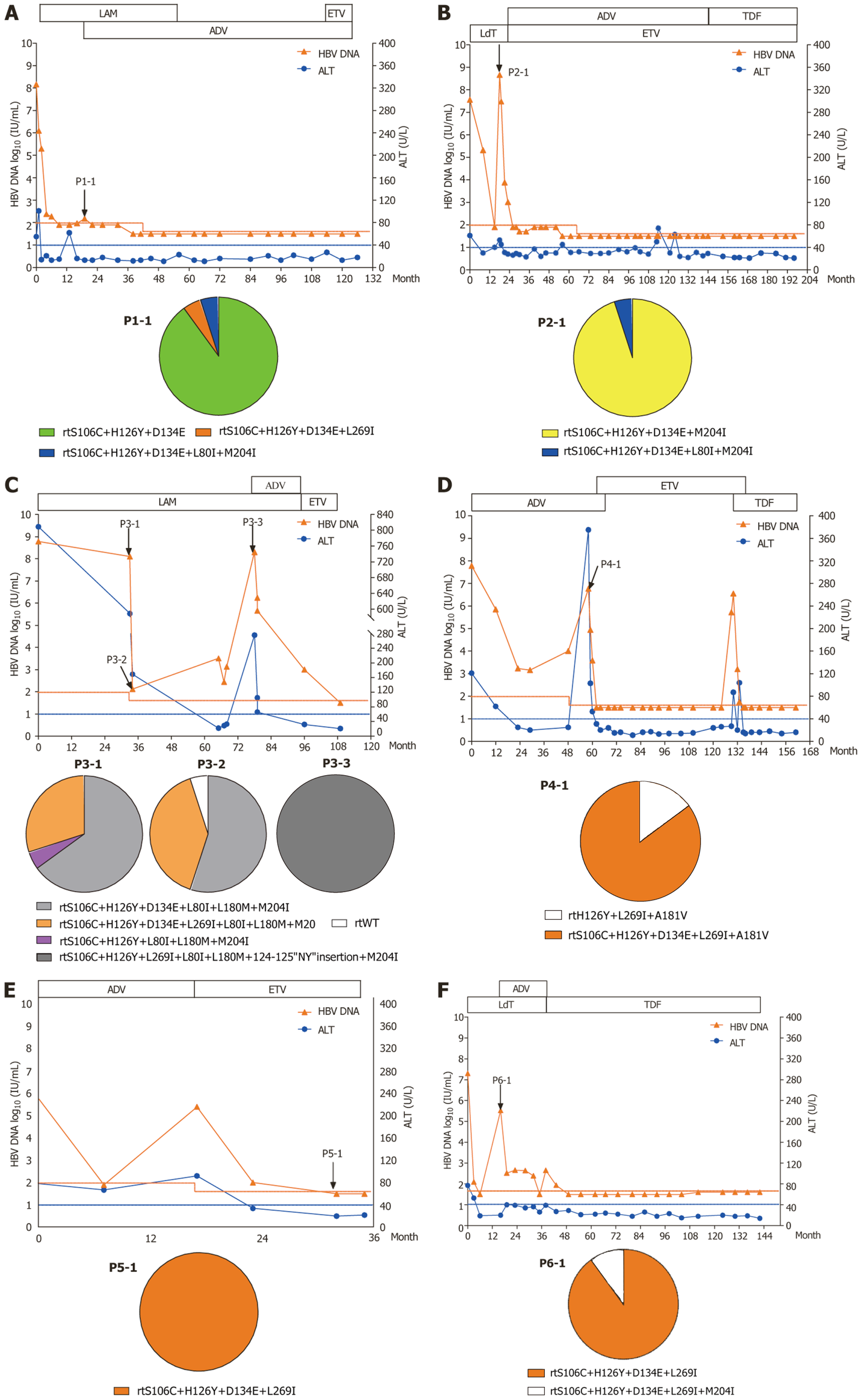
Figure 1 The evolution of drug-resistant hepatitis B virus mutants and clinical responses during the antiviral therapy of six repre
- Citation: Si LL, Fan ZP, Liu WH, Chen RJ, Chen XY, Ji D, Li L, Chen C, Liao H, Wang J, Xu DP, Zhao J, Liu Y. Hepatitis B virus rtCYE/rtCYEI mutations may contribute limited tenofovir resistance: Analysis of a large sample of Chinese patients. World J Hepatol 2025; 17(8): 107456
- URL: https://www.wjgnet.com/1948-5182/full/v17/i8/107456.htm
- DOI: https://dx.doi.org/10.4254/wjh.v17.i8.107456