Copyright
©The Author(s) 2025.
World J Psychiatry. Sep 19, 2025; 15(9): 108382
Published online Sep 19, 2025. doi: 10.5498/wjp.v15.i9.108382
Published online Sep 19, 2025. doi: 10.5498/wjp.v15.i9.108382
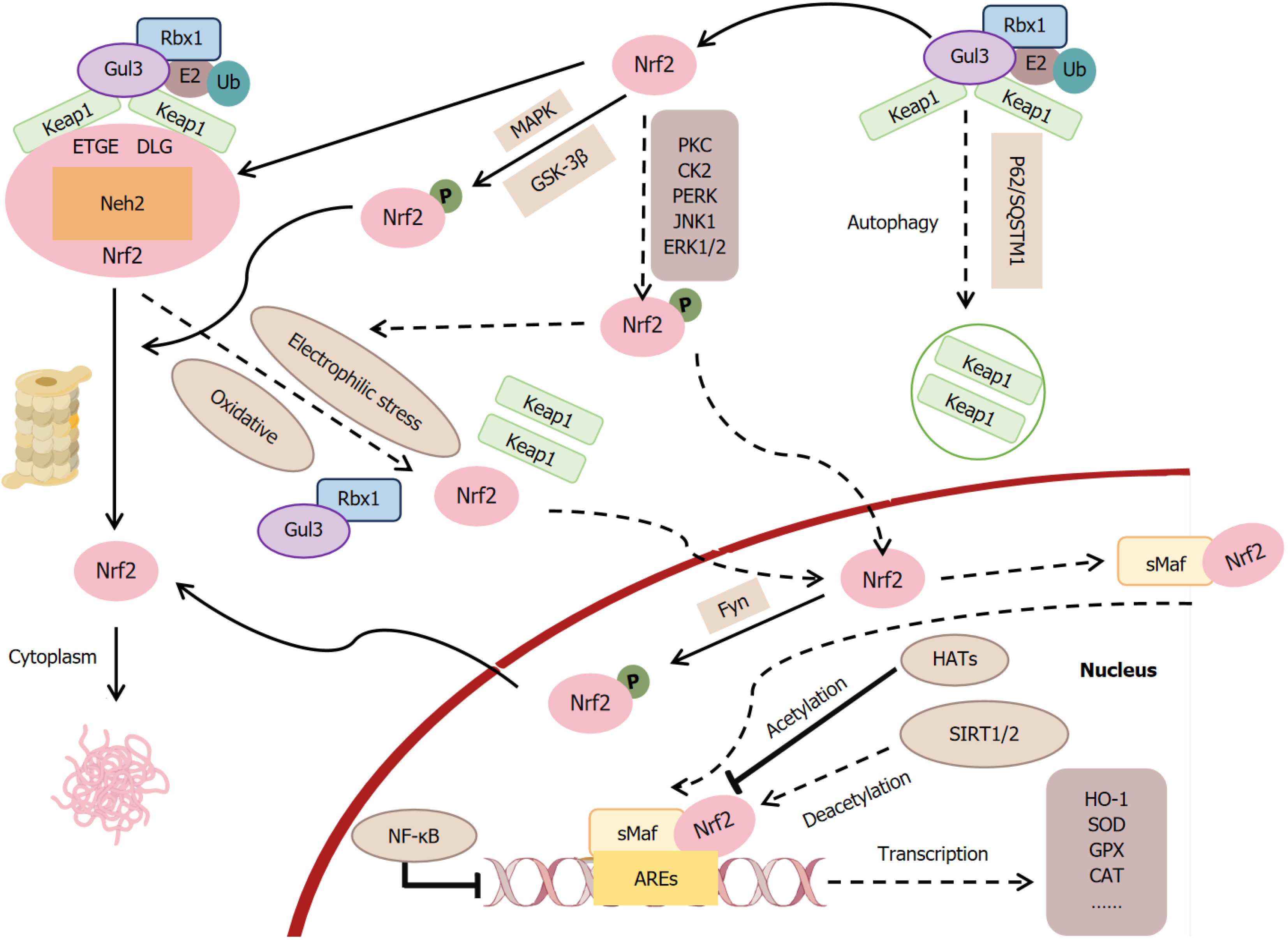
Figure 3 Activation mechanism of the nuclear factor erythroid 2-related factor 2/heme oxygenase-1 pathway: Nuclear factor erythroid 2-related factor 2 is tightly regulated by degradation and stabilization mechanisms.
Kelch-like ECH-associated protein 1 (Keap1)-Cul3 mediates nuclear factor erythroid 2-related factor 2 (Nrf2) ubiquitination and degradation, while p62 sequesters Keap1, enhancing Nrf2 accumulation. Glycogen synthase kinase-3β and mitogen-activated protein kinase phosphorylate Nrf2, promoting degradation, whereas protein kinase C, casein kinase 2, protein kinase RNA–like endoplasmic reticulum kinase, c-Jun N-terminal kinase 1, and extracellular regulated protein kinase 1/2 phosphorylation drive Nrf2 nuclear translocation. In the nucleus, Nrf2-sMaf heterodimers bind antioxidant response elements (AREs) to activate transcription. Conversely, Fyn kinase exports Nrf2, and nuclear factor kappa-B compete for ARE binding, suppressing transcription. Acetylation by histone acetyltransferases enhances Nrf2’s DNA binding and transcriptional activity, whereas deacetylation by silent information regulator 1/2 suppresses its activity. Nrf2: Nuclear factor erythroid 2-related factor 2; Keap1: Kelch-like ECH-associated protein 1; GSK-3β: Glycogen synthase kinase-3β; MAPK: Mitogen-activated protein kinase; PKC: Protein kinase C; CK2: Casein kinase 2; PERK: Protein kinase RNA–like endoplasmic reticulum kinase; JNK1: C-Jun N-terminal kinase 1; ERK: Extracellular regulated protein kinase; NF-κB: Nuclear factor kappa-B; AREs: Antioxidant response elements; HATs: Histone acetyltransferases; SIRT: Silent information regulator; HO-1: Heme oxygenase-1; SOD: Superoxide dismutase; CAT: Catalase; GPX: Glutathione peroxidase.
- Citation: Zhang YM, Zhang ZG. Modulating nuclear factor erythroid 2-related factor 2 and heme oxygenase-1 in liver-brain axis disorders. World J Psychiatry 2025; 15(9): 108382
- URL: https://www.wjgnet.com/2220-3206/full/v15/i9/108382.htm
- DOI: https://dx.doi.org/10.5498/wjp.v15.i9.108382