INTRODUCTION
Ocular trauma can induce cataract formation[1-6]. The methods used to evaluate the visual outcome in eyes managed for traumatic and senile cataracts are similar; however, the damage to other ocular tissues resulting from trauma may additionally compromise the visual gain following surgery. Therefore, the visual outcome may differ depending upon co morbidities. Ocular trauma remains a controversial topic and the debate over management strategies continues. The international classification of ocular trauma proposed almost 15 years ago requires re-evaluation and should be more robust in terms of predicting the outcome of open-globe injury (OGI)[7].
The introduction of the Birmingham Eye Trauma Terminology System (BETTS) has standardized ocular trauma documentation[8]. Consequently, visual outcomes following traumatic cataract surgery and the determinants predicting the outcome can be investigated with respect to the BETTS category[8,9]. Although visual outcomes of traumatic cataracts have been reported, most studies involved only small populations or were case studies.
The timing of cataract surgery and intraocular lens (IOL) implantation in trauma continues to be debated worldwide. A number of issues regarding the management of traumatic cataract remain unresolved. The high risk of amblyopia and intraocular inflammation as well as strong vitreoretinal adhesions in the pediatric age group require management based on different principles. Prospective, controlled clinical studies of OGI are not possible. This article reviews pertinent data regarding these management issues and controversies, and provides recommendations for treatment based on the available published data and the authors’ personal experience. Blindness due to injury is a social and economical burden on society and the individual[6,7,9].
This study was performed with the approval of the Hospital Ethical Committee and complies with the tenets of the Declaration of Helsinki. Written consent in the local language was obtained for the use of the information in this article.
EPIDEMIOLOGY AND INCIDENCE
The incidence, based mainly on retrospective studies and eye injury registries, varies in different regions of world. Incidence variability also arises because of different demographic conditions, including age, sex, environment, and socio-economical conditions[4]. The incidence of traumatic cataract also varies within the pediatric age group[4-6,10,11].
It is imperative that surgeons are aware that traumatic cataract is not a senile cataract. The injury is rarely limited to the lens, but may also be associated with the zonules, posterior capsule, and posterior segment. A patient with traumatic cataract should be informed of the potential visual outcome and the high risk of intraoperative complications[12-19].
It is important to be able to predict visual outcome during the pre-treatment examination as prognosis is to be known to clinician as well as patient. The influence of different variables has been investigated, which we review here with respect to the final visual outcome[20-26].
SPECIAL CONSIDERATION OF TRAUMATIC CATARACT IN THE PEDIATRIC AGE GROUP
Incidence: The incidence varied among studies, most of which were retrospective and involved small databases. Studies on traumatic cataracts in children reported incidences of 25% in southern India, and 12% and 46% in Western India[4-6].
Challenges in children with traumatic cataract include amblyopia, a tendency for inflammation, synechiae and secondary cataract[20]. Children account for approximately one-third of all serious eye injuries[22]. Despite this, the classification and scoring systems in pediatric trauma are based on those developed for adults[22]. The debate over the position of zones II and III is even more pronounced in pediatric trauma. During the first 5 years the length of the pars plana changes rapidly from approximately 1.8 mm in neonates to 3 mm by 1 year of age, and attains 5 mm at 5 years of age[23,24]. Therefore, pediatric trauma assessment for research purposes is liable to inaccuracy, depending on how the injury is classified.
Children and younger patients exhibit stronger adherence between the posterior capsule and the anterior vitreous centrally; furthermore, the central vitreous is anatomically connected to the peripheral retina at the vitreous base[17]. Any traction on the anterior vitreous face is transmitted to the retina, and the younger the patient, the greater the risk. Additionally, children are at risk of amblyopia. In younger children, a surgeon may resort to a single-step procedure and perform lens extraction, IOL implantation and, if required, anterior vitrectomy for an optimal outcome[25-30]. Primary IOL implantation not only prevents amblyopia but also synechiae formation, which can close the lens capsule by the time secondary IOL implantation is due to be performed[31-33].
Although the management of trauma in children has many similarities to that in adults, striking differences also exist. Foremost is that adults have reached visual maturity, whereas amblyopia is a major contributor to poor outcomes in children, especially in under 5 year olds. The worst outcomes reported were that < 50% of the children with an OGI achieved good vision and amblyopia was a major confounding factor[31-33]. To achieve a clear visual axis following globe repair when treating children with OGI, this must be accompanied by accurate refraction and aggressive patching therapy to improve the visual outcome[34]. A further difference is that a simple examination can prove difficult in a child, especially during the early stages after trauma, resulting in the requirement for general anesthesia or sedation for proper assessment. The involvement of a pediatric ophthalmologist and access to pediatric facilities is therefore essential for ultimate management.
Optical rehabilitation is important because an IOL is not implanted in all cases[21]. Orthoptic treatment following optical correction, including patching and monitoring of visual regain, is vital because amblyopia is an important factor[21].
In conclusion, children with an OGI cannot simply be treated as small adults. Patient age and the effects of amblyopia should be included as additional negative prognostic factors.
Sex
Generally, males are more affected by injuries because of greater participation in outdoor activities; however, no significant difference was found in visual outcome according to sex.
Hospital attendance
Patient hospital attendance has had a significant effect on visual outcome (Table 1)[2,4,34-40]. Patients attend a hospital either by self reporting or via outreach activities, including mobile diagnostic camps and school screening. Patients who have attended an outreach program usually present late; thus morphological changes and visual outcome differ.
Habitat
Ocular injuries are more common in rural areas because of the type of related domestic and professional activities[2,4,39,40]. However, the visual outcome did not differ significantly in relation to the habitat.
Previous treatment
The number of patients who received previous treatment depended upon awareness and the availability of services. Past treatment did not have a significant effect on the final visual outcome[39,40].
Object causing the injury
The object responsible for an injury varies according to geographical and socio-economic status, with a marked variation in the United States Eye Injury Register and reports from other sources[2,4,39]. However, there was no significant difference with respect to the visual outcome.
Activity during injury
The type of activity during injury also varies according to geographical and socio-economic status, as indicated by the United States Eye Injury Registry and reports from other sources[2,4], although no significant difference was found regarding visual outcome[39,40].
Preoperative visual acuity of no light perception and poor visual prognosis
Visual acuity can be profoundly impaired, even to the extent of no light perception (NLP), in the presence of significant media opacity (e.g., corneal edema, hyphema, cataract, dense vitreous hemorrhage), retinal detachment, associated subretinal or subhyaloid hemorrhage, hemorrhagic choroids, and even psychological factors (e.g., hysteria). The assessment of light perception is a subjective measurement and not a fool-proof test in the presence of severe media opacity secondary to dense vitreous hemorrhage, traumatic cataract, dense hyphema, or corneal edema. Even with the bright light of an indirect ophthalmoscope, the assessment of light perception can give a false impression of NLP[38]. Ultrasonography is useful in assessing the posterior segment in eyes with media opacity, and to differentiate between retinal detachment and vitreous hemorrhage[38]. However, when using this methodology it is sometimes difficult to differentiate a detached retina from blood clots in the vitreous cavity and membranes[38]. Before deciding on enucleation in NLP patients, reversible causes of vision loss should be excluded, including psychological factors[38]. Even when enucleation appears inevitable, the ophthalmologist should still discuss the possible options with the patient before making a final decision. Primary enucleation of severely traumatized eyes with NLP is controversial due to the risk of sympathetic ophthalmia. Sympathetic ophthalmia with the potential for bilateral blindness is a relative indication for enucleation of an injured eye. There fore, primary surgical repair should not necessarily be discounted because of the risk of sympathetic ophthalmia in eyes with NLP. Currently, most surgeons recommend a globe-salvaging procedure for eyes with severe trauma with NLP vision at initial presentation.
The age in children at which IOL insertion and rehabilitation of aphakia can be undertaken remains under debate.
Morphology of traumatic cataract
There is no standard morphological classification. Attempts at grading have been made, but these are arbitrary[12]. The morphology of traumatic cataract depends mainly upon the type of injury and the time interval between the injury and intervention.
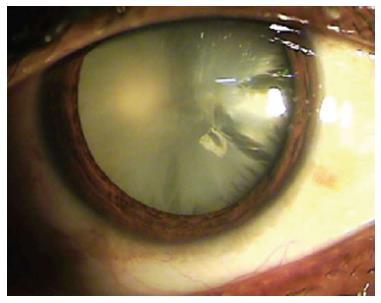
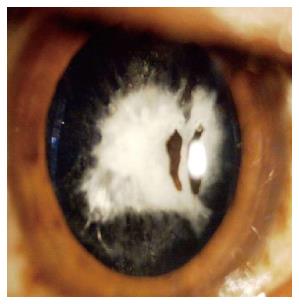
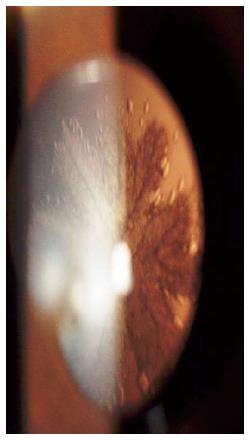
Based on lenticular opacity, cataracts were classified as: total (Figure 1); white soft with soft material floating in the anterior chamber with broken anterior capsule (Figure 2); membranous, in which both capsules were fused with little or no cortical material (Figure 3); and rosette (Figure 4). A cataract was defined as total when no clear lens matter was observed between the capsule and nucleus. For a membranous cataract the capsule and organized matter were fused and formed a membrane of varying density. A cataract was considered to be white soft when loose cortical material was found in the anterior chamber together with a ruptured lens capsule. A lens with a rosette pattern of opacity was classified as a rosette cataract. One study proposed that all cataract cases could be assigned to these groups[13].
Figure 1 When no clear lens matter was visible between the capsule and the nucleus, the cataract was defined as a total cataract.
Figure 2 When loose cortical material was found in the anterior chamber together with a ruptured lens capsule, the cataract was defined as a white soft cataract.
Figure 3 When the capsule and organised matter were fused and formed a membrane of varying density, the cataract was defined as a membranous cataract.
Figure 4 A lens with a rosette pattern of opacity was classified as a rosette type cataract.
Management and surgical approaches to traumatic cataract
The choice of surgery in adults in whom amblyopia is not an issue is governed by the surgeon’s own preferences and to a certain extent by the status of the cataractous lens. If the anterior capsule is significantly disrupted and there is free floating lens matter in the anterior chamber, the surgeon may be justified in primary cataract extraction with or without IOL implantation (Figure 3).
Eyes with a lens vitreous admixture should be considered for combined cataract extraction with limited anterior vitrectomy. Judicious use of a vitrector and not an aspirator should be made while removing vitreous admixture in the ruptured lens matter (Figure 4). Any traction on the vitreous may result in inadvertent retinal breaks[14]. When there is additional injury to the posterior segment, early pars plana lensectomy and vitrectomy by a posterior segment specialist is warranted[14]. In eyes with an intact anterior capsule and total traumatic cataract, a second-sitting cataract extraction with IOL implantation should be the best and safest approach for optimal visual outcome[17,18].
Wherever possible, a multistep procedure after control of inflammation, with adequate corneal clarity and an appropriate IOL power calculation, should be adopted[18,19]. It remains debated whether an anterior or posterior surgical approach via the pars plana route should be performed.
The following forms of surgical approach may be used, according to morphology: (1) unimanual or bimanual aspiration; (2) lensectomy/membranectomy using the limbal or pars plana approach and insertion of the lens in the sulcus; and (3) phacoemulcification or small-incision cataract surgery when the nucleus is harder[13].
Primary posterior capsulotomy
Determination of the correct IOL power prior to surgery may be difficult, if not impossible, for a variety of reasons. Often the other eye serves as a guide, and inflammatory debris can settle on the IOL surface, cleansing of which may require postoperative yttrium-aluminum-garnet laser treatment or even surgery. In addition, the edge of the IOL will interfere with the surgeon’s visualization of the peripheral retina should subsequent development of proliferative vitreoretinopathy necessitate vitrectomy[35]. There has been considerable debate over the use of primary posterior capsulotomy[39-41].
Primary posterior capsulotomy and vitrectomy in traumatic cataract have proved to be positive outcome predictors[39,40]. It was found that condensation of the anterior vitreous in severe inflammation was a positive outcome predictor in nonrandomized studies, while a randomized controlled trial suggested that this was a positive predictor.
Timing of intervention
The timing of cataract extraction and IOL implantation has been discussed extensively. Because evidence supports both primary and secondary cataract extractions, a number of crucial factors should be considered before making a decision. Cataract extraction together with primary wound repair may have distinct advantages, including controlling inflammation, however, this may raise intraocular pressure because of soft lens matter in the anterior chamber[13]. Secondary advantages include the direct visualization of the posterior segment and optic nerve[14]. Similarly, in pediatric patients the removal of a media opacity may be crucial to prevent vision-deprivation amblyopia. In patients with a lens vitreous admixture, this is a potent stimulator for further proliferative vitreoretinopathy and may also result in traction on the retina; hence primary extraction of the lens and vitreous is imperative in such patients [35,36]. Minor advantages of primary lens removal are the patient’s convenience and possibly cost effectiveness[14].
Proponents of second-sitting cataract extraction recommend effective control of intraocular inflammation, good media clarity, and a stable wound before considering traumatic cataract extraction[14,39]. On adequate control of inflammation, IOL implantation at second-stage cataract extraction may be associated with a better outcome[14,39]. An IOL power calculation is appropriate when IOL implantation is planned for a second sitting[18,35].
Current data suggest that improved visual outcome results from intervention at 2-30 d[36]. Awareness of several details is necessary before embarking on primary or delayed cataract extraction with IOL implantation. These include the age of the patient, the expertise of the surgeon and assisting staff, the infrastructure available, and the status of the cataractous lens and the lens vitreous admixture, which will act as a guide to the surgeon in planning the surgery.
The ophthalmologist must not subject a patient to half-completed or compromised surgery because of a lack of expertise or proper infrastructure. If the facility is not equipped to provide the surgeon after hours with the full range of equipment, instruments, and material together with a full and knowledgeable staff, it is preferable not to contemplate primary lens removal. In most hospitals globally when an OGI is seen after hours, the state-of-the-art facilities necessary to attempt cataract extraction and IOL implantation are not normally available.
Lens implant
Lens implantation is an important stage in traumatic cataract surgery. A primary implant at the time of cataract extraction or a secondary implant during a second sitting is important for optical rehabilitation[39,40]. The lens implant may be placed in the capsule or sulcus, be iris supported, or involve scleral fixation depending upon the extent of the damage. Lens implants help in optical rehabilitation and the prevention of amblyopia in children. Lens insertion makes a significant difference to visual outcome.
Number of surgeries
Ocular injuries may cause damage to multiple ocular structures. It may therefore be impossible to treat all of the tissues in one session; thus a number of surgeries may be required to complete surgical management. Corneal/scleral wound repair, cataract extraction, lens implant, membranectomy, or vitrectomy may be performed at different stages[39,40]. Various studies reported that the number of surgeries did not have a significant effect on the final visual outcome.
Type of injury
The type of injury may influence visual outcome. Reports have suggested that OGI has a better outcome, whereas the opposite was true for penetrative injury[38-40]. Influences open globe of BETTS as a predictive factor (Table 1)[39,40]. When compared to the open-globe subgroups, cataract caused by penetrating injury had a better outcome whereas globe rupture had a poorer outcome[39-41].
CONCLUSION
Despite the advances in state-of-the-art surgery and understanding ocular trauma, a range of unresolved, controversial issues remain in the management and treatment of OGIs. During the last two decades all of these issues have been addressed and real progress has been made in many aspects of the definitive management of traumatic cataracts.
The timing of the intervention in traumatic cataract appears to be a never-ending debate. “Sooner the better” was the traditional view; however, an alternative view is that a better outcome results from intervention between 3-30 d. Although a controlled, prospective clinical trial would be the ideal, no two ocular trauma cases are alike, and confounding factors can affect the final outcome.
A morphological assessment may be used to guide management. The age at intervention and laterality play important roles as predictors of visual outcome[21,40,49]. The accuracy of predictive models varies between adult and pediatric traumatic-cataract cases.
Controlling for the significant differences that occur among individual injuries is difficult. This makes the independent assessment of potential risk factors and treatment variances for visual and anatomical outcome difficult. Current management is based on the surgeon’s experience, and will continue to be based on a retrospective review of accumulated data and the personal preferences of the treating ophthalmologist in OGI. These types of management problems are dealt with on a case-by-case basis, and even the most experienced ophthalmologists will at some time find themselves in a dilemma regarding the strategic planning of ocular trauma management of OGI. This review has attempted to provide a comprehensive overview of most of the controversies related to the management of OGI, and has presented our preferred guidelines. This should aid ophthalmologists in patient counseling and the process of making decisions regarding the management of OGIs involving the anterior or posterior segment.
In summary, the vast majority of ophthalmologists who encounter a traumatic cataract have sufficient experience in lens extraction and IOL implantation in the non-traumatic setting. What every ophthalmologist must accept is that an injured lens requires many individualized, conscious decisions regarding what to do, when to do it, and how to achieve the best possible outcome.