Published online Mar 24, 2020. doi: 10.5306/wjco.v11.i3.162
Peer-review started: October 21, 2019
First decision: November 5, 2019
Revised: December 9, 2019
Accepted: December 13, 2019
Article in press: December 13, 2019
Published online: March 24, 2020
Histiocytic sarcoma (HS) of the spleen is reported to be a rare and lethal disease. The clinicopathological features of splenic HS have not been well described. The objective of this paper is to describe the diagnosis and treatment of a case of this rare disease and provide a review of the literature.
In this article, we discuss the case of a 40-year-old Hispanic female who presented with progressive thrombocytopenia and multiple hypoechoic lesions in the spleen without splenomegaly. Positron emission tomography-computed tomography showed increased activity in cervical lymph nodes, as well as multiple bone and splenic lesions with positive uptake. Two bone marrow biopsies and fine-needle aspiration of the cervical lymph node were inconclusive. Laparoscopic splenectomy was performed, and gross examination showed a 110.1 g spleen with multiple rubbery, nodular lesions within the subcapsular sinus and splenic parenchyma. The microscopic findings showed multinodular histiocyte proliferation with atypia and multilobulated nuclei, which were positive for CD163, CD4, and CD68 by immunohistochemical analysis. The final pathologic diagnosis was difficult and was found to be low-grade HS of the spleen, after consultations with two renowned hematopathology institutions. At the patient’s five-month follow-up visit, her bone marrow metastasis had progressed. She is waiting to be enrolled in a clinical trial.
Pathologic diagnosis of splenic HS can be challenging. Low-grade differentiation may be associated with a slow progressive disease.
Core tip: Histiocytic sarcoma (HS) of the spleen is a rare and potentially lethal condition. We report a patient with thrombocytopenia of unknown etiology, multiple splenic lesions, and disseminated bone metastasis treated by laparoscopic splenectomy. The final pathologic diagnosis showed low-grade HS of the spleen. It is very rare that HS occurs without splenomegaly.
- Citation: Huang K, Columbie AF, Allan RW, Misra S. Thrombocytopenia with multiple splenic lesions - histiocytic sarcoma of the spleen without splenomegaly: A case report. World J Clin Oncol 2020; 11(3): 162-168
- URL: https://www.wjgnet.com/2218-4333/full/v11/i3/162.htm
- DOI: https://dx.doi.org/10.5306/wjco.v11.i3.162
Histiocytic sarcoma (HS) is a rare malignant neoplasm that occurs in lymph nodes, skin, and the gastrointestinal tract. It is a malignant proliferation of cells showing morphologic and immunophenotypic characteristics of mature tissue histiocytes[1], and represents less than 0.5% of all non-Hodgkin’s lymphoma. HS of the spleen is a rare and potentially lethal condition that can remain asymptomatic or only mildly symptomatic for an extended period[2]. The diagnosis depends on verification of histiocytic lineage and distinguishing HS from other benign or malignant diseases, such as benign histiocytic proliferation, hemophagocytic syndrome, malignant histiocytosis, and acute monocytic leukemia using immunohistochemical techniques and molecular genetic tools[3-5].
Due to its rarity, the number of “true” cases reported as primary splenic HS in the English literature are few (up to eight cases reported), with most recent references from 2012. The clinicopathological features of HS have not been well described. Primary splenic HS cases often show a multi-nodular lesion in an enlarged spleen, with no specific findings in imaging studies[6,7]. Patients with splenic HS have a poor prognosis due to the aggressive behavior of this entity, even though a splenectomy might induce temporary remission. Liver or bone marrow infiltration constitutes a common finding that partially obscures the prognostic evaluation. Early evaluation and diagnosis, before dissemination of the disease, may improve the prognosis and prospects of survival. We report a case of thrombocytopenia of unknown etiology with multiple splenic lesions and disseminated bone metastasis treated by laparoscopic splenectomy. The final diagnosis was low-grade HS of the spleen.
Left upper quadrant pain, drenching sweats.
A 40-year-old Hispanic female was referred to our clinic due to persistent thrombocytopenia and multiple splenic lesions. She initially presented with drenching sweats and left upper abdominal pain that was associated with progressive thrombocytopenia for four months. She denied recent weight loss, fever, or chills.
The patient denied previous medical or surgical history.
Not contributory.
On physical examination, no hepatomegaly, splenomegaly, or superficial lymphadenopathy was observed.
Initial assessment revealed thrombocytopenia with a platelet count of 37000/mm3. Her white blood cell count was 10300/μL, and hemoglobin was 14.0 g/dL. The biochemistry profile was unremarkable. Further study of serum protein electrophoresis, peripheral blood flow cytometry, and fine-needle aspiration (FNA) of the right cervical level II lymph node was unremarkable. She had two bone marrow biopsies, which did not show evidence of lymphoproliferative disorder.
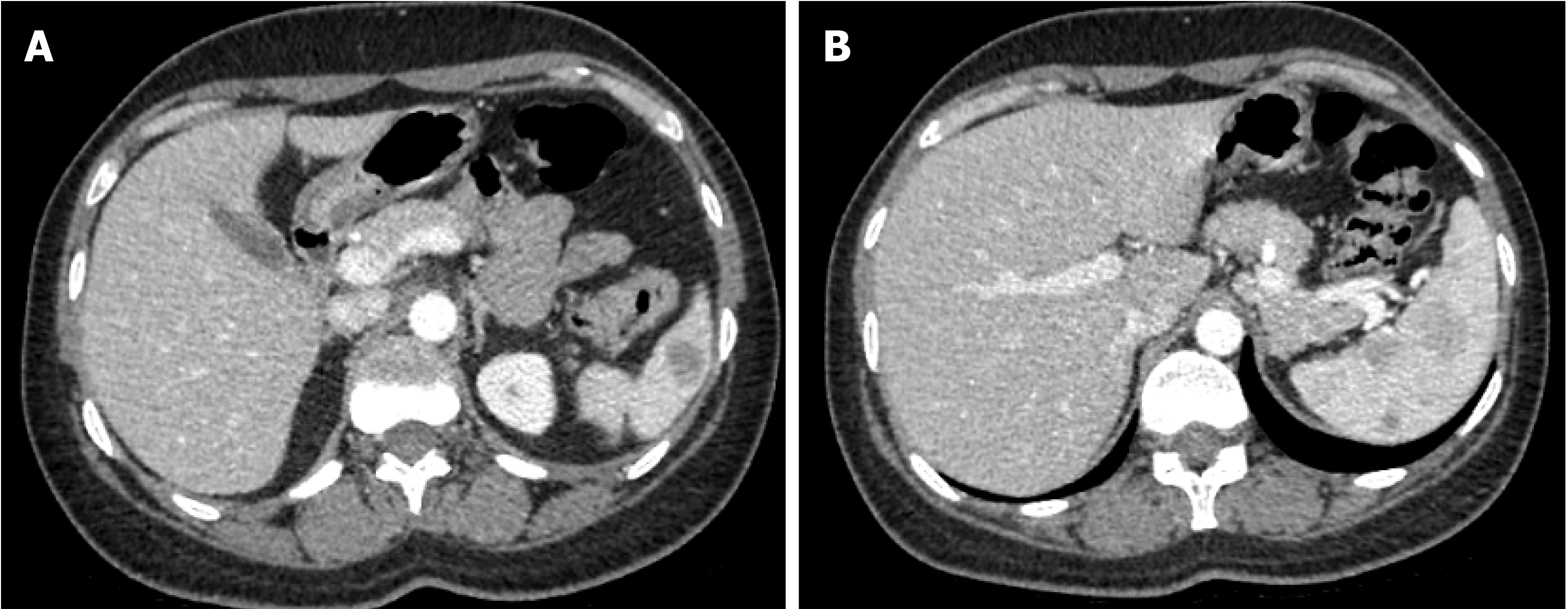
Magnetic resonance imaging (MRI) of the spleen demonstrated a normal-sized spleen (greatest dimension 10 cm) with multiple T1 hypointense/T2 hyperintense lesions, suggestive of possible extramedullary hematopoiesis or lymphoma. Positron emission tomography-computed tomography (PET/CT) showed mild uptake (standardized uptake value (SUV) 3.5) in one of the lesions along the splenic hilum. All other splenic nodules were non-avid. A small cervical node (SUV 3.3), left breast nodule (SUV 2.1), and several bone sites (brightest at T3, SUV 4.3) were also noted.
Low-grade HS of spleen.
The patient was started on 40 mg of prednisone. This was progressively increased to 80 mg daily, with some improvement in her platelet count, which had been stable at 75000/mm3. She was otherwise doing well. She had no complaints of fever or weight loss, and no enlarged lymph nodes or neurological symptoms were noted on physical examination. The case was discussed at a tumor board. A decision was made to perform laparoscopic splenectomy. The patient was placed on a steroid taper with prednisone, 20 mg daily, preoperatively. Subsequently, her platelet count was hovering around 41000/mm3.
During surgery, a 10 mm trocar was inserted at the mid-clavicular line between the umbilicus and the left costal margin, and three trocars (10 mm, 5 mm, 5 mm) were placed at the anterior axillary line and midline during the procedure. A laparoscopic view showed a normal-sized liver and spleen, with no accessory splenules visualized. In addition, no metastatic or disseminated lesions were detected. The spleen was extracted from a 4 cm midline incision. The operation time was 2 h, and blood loss was 150 mL. To address her thrombocytopenia, 20 U of platelets were transfused after splenic vessels were transected.
Gross specimen: The weight of the resected spleen was 110.1 g and measured 12 cm × 8.0 cm × 3.6 cm. There were numerous nodular, tan, rubbery, well-circumscribed lesions with smooth surfaces. The lesions were located within the subcapsular sinus and splenic parenchyma (greater than ten lesions).
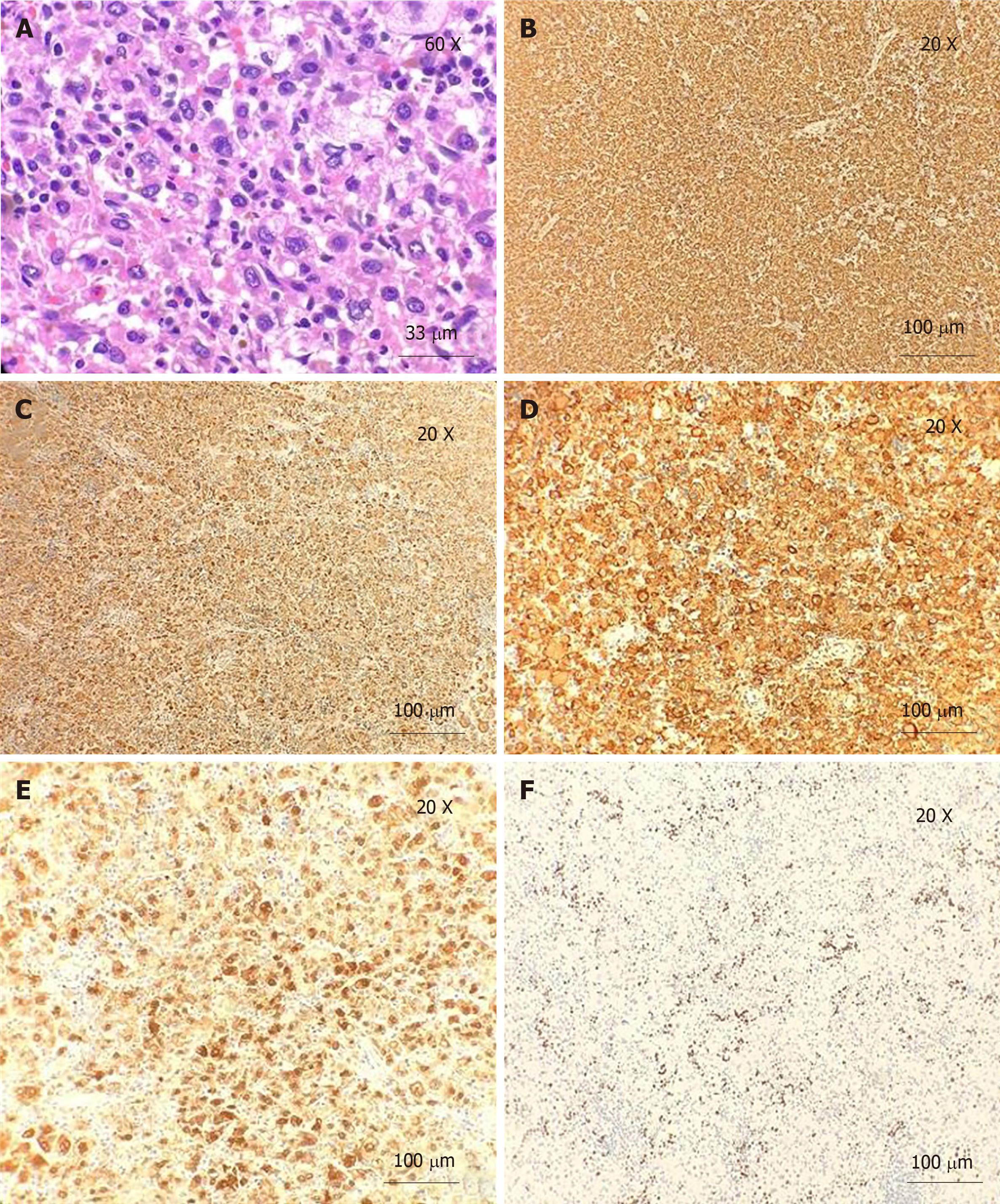
Microscopically, the H and E-stained sections showed a spleen with multinodular proliferation of histiocytoid cells. There was an accompanying infiltrate of scattered small lymphoid cells, neutrophils, and eosinophils. The histiocytoid cells had abundant eosinophilic cytoplasm and showed variable nuclear atypia (Figures 1 and 2). Immunohistochemistry showed immunoreactivity for CD163, CD4, and CD68, factor XIIIa, and CD14. The Ki-67 showed a proliferative index of 10%. Only rare mitotic figures, with rare atypical mitoses, were noted. The patient’s pathologic diagnosis was difficult. After consulting two renowned pathology institutions, the final pathologic diagnosis was HS of the spleen.
The postoperative course was uneventful. The patient’s platelet count normalized after surgical intervention: 13900/mm3 on postoperative day one (POD1) and 16300/mm3 on POD2. The patient was discharged on POD2 with instructions to follow-up one month after surgery. She has been feeling well and had marked improvement in pain and night sweats, with platelet values remaining stable. She has not noticed any enlarged lymph nodes, fever, weight loss, or neurological deficits. The patient was seen at a renowned cancer center after a final diagnosis of low-grade HS. Her repeat PET, one month after surgery, showed low avidity of bone marrow without interval changes. She remains in observation without adjuvant treatment. However, her five-month PET scan showed progressed bone marrow metastasis. She is waiting to be enrolled in a clinical trial.
HS is a rare malignant proliferation of cells showing morphologic and immun-ophenotypic features, similar to mature tissue histiocytes. Clinically, it is generally accepted that most patients with HS have a poor prognosis due to early disseminated disease and limited response to chemotherapy[8]. Previous cases, including extra-nodal HS of non-splenic origin, showed that the stage of the disease and possibly tumor size can be important prognostic indicators[9,10]. HS of the spleen is a rare condition, and its clinicopathological features have not been well described[7]. According to the nine cases reported so far (Table 1), the majority of patients are female (6/9), with an age range of 29-82 years (median 67), and presented with thrombocytopenia and multinodular lesions in the setting of splenomegaly. The most common distant metastases are liver and bone marrow. Survival time ranges from 1 month to 5 years (median six months). Early diagnosis, before dissemination of this disease, may improve the prognosis. Extra-nodal extension and bone marrow involvement with consequent cytopenia (due to hemophagocytosis) obscure the prognosis.
| Case [Ref.] | Age (yr)/Sex | Chief complaint | Imaging | Weight of spleen | Treatment | Survival | Metastasis |
| 1[6] | 38/male | Weakness | Multinodular | 264 g | S | - | - |
| 2[2] | 29/male | Edema | Nodular | 735 g | S (R+C) | 5 Yr 1 Mo | Liver |
| 3[2] | 60/male | Thrombocytopenia | Nodular | 610 g | S+C | 1 Yr 6 Mo | Liver, BM |
| 4[2] | 66/female | Edema, anemia | Multinodular | 750 g | R+S+C | 2 Yr 6 Mo | Liver, BM |
| 5[15] | 71/female | Thrombocytopenia | Multinodular | 470 g | C | 6 Mo | Hilum LN |
| 6[16] | 82/female | Evans syn | Multinodular | 110 g | R | 1 Mo | Liver |
| 7[17] | 58/female | Anemia | Nodular | 800 g | R+S+C | 3 Mo | - |
| 8[18] | 67/female | Thrombocytopenia | Non-nodular | 760 g | S | 6 Mo | BM |
| 9[19] | 81/female | Anemia, thrombocytopenia | Multinodular | 236 g | S | 5 Mo | Liver |
| 10 | 40/female | Thrombocytopenia | Multinodular | 110.1 g | S | - | BM |
The diagnosis of HS was based on histology and cytomorphology examinations, along with an immunohistochemical profile that pointed toward histiocytic lineage. Histologically, the lesion was composed of non-cohesive large cells with polygonal to cuboidal shape. These large neoplastic cells have abundant eosinophilic cytoplasm with vacuoles and a large, oval, eccentrically-located nucleus with vesicular chromatin and prominent, irregular nucleoli. Rarely, hemophagocytosis and giant cells may also be seen.
Cytomorphologic findings are not specific for HS; hence, immunohistochemical stains were utilized to prove histiocytic differentiation. The neoplastic cells were positive for CD68, CD163, CD14, CD4, CD11c, lysozyme, and alpha-1-antitrypsin and negative for epithelial and hematolymphoid differentiation markers. The proliferation index was highly variable, demonstrated by Ki-67 (range from 1 to 99%). Some genetic aberrations have been variably detected in cases of HS, among which BRAF (present in up to 70% of cases), HRAS, and BRAF gene fusion are the most frequently linked.
The conclusive diagnosis of HS is based on not only histological and immunohistochemical examination of histiocytic differentiation but also exclusion of other immunophenotypes, including lymphoid, epithelial, and melanocytic differentiation. Among the differential diagnoses, Langerhans cell histiocytosis (LCH), hemophagocytic lymphohistiocytosis, follicular cell sarcoma, and interdigitating cell sarcoma are among the most emphasized. HS and LCH are both determined by histiocytic proliferation. However, these entities have structural, enzymatic, and immunohistochemical differences that allow for proper differentiation in the majority of cases. The presence of positive immunohistochemistry for alpha-1-antitrypsin and lysozymes in this case effectively ruled out LCH. Follicular dendritic cell sarcoma was also discarded using the same criteria for LCH. Interdigitating dendritic cell sarcoma shares an immunohistochemical profile closely related to HS, while still being negative for alpha-1-antitrypsin. BRAF mutation has not been detected in these cases. The presence of positive staining for lysozymes can obscure the differential diagnosis; however, the ultrastructural examination demonstrated the scattered distribution of these instead of abundant and evenly distributed lysozymes in HS. Hemophagocytic lymphohistiocytosis is also a neoplastic proliferation of histiocytes by definition. Hematophagocytosis, while found in both entities, is more common and prominent in the former and constitutes a rare finding in the latter.
Imaging findings are essential for the early diagnosis of HS. However, satisfactory diagnostic imaging characteristics are still lacking. Contrast-enhanced CT scans demonstrated multiple, partially confluent, hypoattenuating masses in the enlarged spleen with multiple liver infiltration, and ultrasonography revealed multiple, ill-defined hypoechoic lesions. HS commonly presents as multiple hypointense T1 and hyperintense T2 lesions on MRI, which can also be found in cases of splenic neoplasm such as splenic lymphoma, hemagiomatosis, or angiosarcoma. Case reports have indicated the possible role of PET scan in histiocytic lineage disorders, such as LCH[12], and PET scans can be very valuable in the evaluation of disease dissemination and tailoring treatment in HS[13]. In our patient, initially, among those multiple splenic lesions on MRI, only one of the lesions along the splenic hilum showed a mild SUV of 3.5 on PET/CT, and all other splenic nodules showed non-avidity on the PET scan. No significant interval avidity changes were seen at previous active bone sites in the repeated PET scan three months later. However, progression of bone marrow metastasis was seen five months later, which is consistent with the characteristics of low-grade disease.
Splenectomy is useful for definitive diagnosis of the disease. At the early stage of the disease, splenectomy would be expected to be beneficial for those with HS of the spleen to prevent continuous dissemination from the primary tumor site. Our patient presented with multiple splenic lesions with a disseminated bone lesion. Two bone marrow biopsies and FNA of lymph nodes were inconclusive concerning the diagnosis. Moreover, our patient suffered from progressive thrombocytopenia. Laparoscopic splenectomy was performed for both therapeutic and diagnostic purposes. There is no standard treatment regimen for patients with HS, and patients should be encouraged to enroll in a clinical trial if one is available[14]. Adjuvant treatments of HS include radiotherapy, chemotherapy, and combinations thereof, depending on the stage of the disease. Lymphoma-type systemic chemotherapy has been recommended for multiple systemic diseases. Nine previously reported cases of HS of the spleen recovered asymptomatically after splenectomy for a definitive diagnosis (including five patients who underwent subsequent chemotherapy[2]). All but one patient (who was alive 13 mo after splenectomy and whose outcome is not yet known) died from liver infiltration[6,15].
Our patient presented with disseminated bone metastasis and multiple splenic lesions, without initial splenomegaly. Her bone marrow metastases were stable and then progressed at eight months after diagnosis without adjuvant treatment. She remains in good performance status without other distant organ metastases. Her low-grade differentiated tumor may be associated with a slow progressive disease.
HS of the spleen is a rare, lethal disease. The pathologic diagnosis can be difficult due to its differentiation with malignant LCH or other benign histiocytic proliferation, as in our case. PET scans can be very valuable in the evaluation of disease dissemination. Low-grade differentiation may be associated with a slow progressive disease.
The authors thank Nguyen Quoc-Han, MD, for language and grammar revision and LeAnn Garza of English Pro, LLC for substantive editing of this manuscript. This research was supported (in whole or part) by HCA Healthcare and/or an HCA Healthcare affiliated entity. The views expressed in this publication represent those of the authors and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.
Manuscript source: Unsolicited manuscript
Specialty type: Oncology
Country of origin: United States
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Sun WB, Viswanath YKS S-Editor: Dou Y L-Editor: Webster JR E-Editor: Liu MY
| 1. | Grogan TM, Pileri SA, Chan JKC, Weiss LM, Fletcher CDM. Histiocytic sarcoma. In: Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Vardiman JW. World Health Organization Classification of Tumours: Pathology and Genetics of Tumours of Haematopoietic and Lymphoid Tissues. Lyon: ARC Press, 2008; 356-357. [Cited in This Article: ] |
| 2. | Kimura H, Nasu K, Sakai C, Shiga Y, Miyamoto E, Shintaku M, Wakatsuki S, Tominaga K, Abe M, Maruyama Y. Histiocytic sarcoma of the spleen associated with hypoalbuminemia, hypo gamma-globulinemia and thrombocytopenia as a possibly unique clinical entity--report of three cases. Leuk Lymphoma. 1998;31:217-224. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 20] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 3. | Lauritzen AF, Delsol G, Hansen NE, Horn T, Ersbøll J, Hou-Jensen K, Ralfkiaer E. Histiocytic sarcomas and monoblastic leukemias. A clinical, histologic, and immunophenotypical study. Am J Clin Pathol. 1994;102:45-54. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 58] [Cited by in F6Publishing: 60] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 4. | Risdall RJ, McKenna RW, Nesbit ME, Krivit W, Balfour HH, Simmons RL, Brunning RD. Virus-associated hemophagocytic syndrome: a benign histiocytic proliferation distinct from malignant histiocytosis. Cancer. 1979;44:993-1002. [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 3] [Reference Citation Analysis (0)] |
| 5. | Cattoretti G, Villa A, Vezzoni P, Giardini R, Lombardi L, Rilke F. Malignant histiocytosis. A phenotypic and genotypic investigation. Am J Pathol. 1990;136:1009-1019. [PubMed] [Cited in This Article: ] |
| 6. | Franchino C, Reich C, Distenfeld A, Ubriaco A, Knowles DM. A clinicopathologically distinctive primary splenic histiocytic neoplasm. Demonstration of its histiocyte derivation by immunophenotypic and molecular genetic analysis. Am J Surg Pathol. 1988;12:398-404. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 22] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 7. | Jaffe E. Histiocytic and dendritic cell neoplasms. In: Jaffe ESHN, Stein H, Vardiman JW, editors. World health organization classification of tumours: pathology and genetics of tumours of haematopoietic and lymphoid tissues. Lyon: IARC, 2001; 273–277. [Cited in This Article: ] |
| 8. | Copie-Bergman C, Wotherspoon AC, Norton AJ, Diss TC, Isaacson PG. True histiocytic lymphoma: a morphologic, immunohistochemical, and molecular genetic study of 13 cases. Am J Surg Pathol. 1998;22:1386-1392. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 81] [Cited by in F6Publishing: 83] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 9. | Vos JA, Abbondanzo SL, Barekman CL, Andriko JW, Miettinen M, Aguilera NS. Histiocytic sarcoma: a study of five cases including the histiocyte marker CD163. Mod Pathol. 2005;18:693-704. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 182] [Cited by in F6Publishing: 156] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 10. | Hornick JL, Jaffe ES, Fletcher CD. Extranodal histiocytic sarcoma: clinicopathologic analysis of 14 cases of a rare epithelioid malignancy. Am J Surg Pathol. 2004;28:1133-1144. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 218] [Cited by in F6Publishing: 202] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 11. | Blum R, Seymour JF, Hicks RJ. Role of 18FDG-positron emission tomography scanning in the management of histiocytosis. Leuk Lymphoma. 2002;43:2155-2157. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 28] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 12. | Kaste SC, Rodriguez-Galindo C, McCarville ME, Shulkin BL. PET-CT in pediatric Langerhans cell histiocytosis. Pediatr Radiol. 2007;37:615-622. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 76] [Cited by in F6Publishing: 60] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 13. | Yaman E, Ozturk B, Erdem O, Gokcora N, Coskun U, Uluoglu O, Benekli M. Histiocytic sarcoma: PET-CT evaluation of a rare entity. Ann Nucl Med. 2008;22:715-717. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Jaobsen E. Histioytic sarcoma. Available from: https://www.uptodate.com/contents/histiocytic-sarcoma. [Cited in This Article: ] |
| 15. | Audouin J, Vercelli-Retta J, Le Tourneau A, Adida C, Camilleri-Broët S, Molina T, Diebold J. Primary histiocytic sarcoma of the spleen associated with erythrophagocytic histiocytosis. Pathol Res Pract. 2003;199:107-112. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 27] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 16. | Kobayashi S, Kimura F, Hama Y, Ogura K, Torikai H, Kobayashi A, Ikeda T, Sato K, Aida S, Kosuda S, Motoyoshi K. Histiocytic sarcoma of the spleen: case report of asymptomatic onset of thrombocytopenia and complex imaging features. Int J Hematol. 2008;87:83-87. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 17. | Oka K, Nakamine H, Maeda K, Yamakawa M, Imai H, Tada K, Ito M, Watanabe Y, Suzuki H, Iwasa M, Tanaka I. Primary histiocytic sarcoma of the spleen associated with hemophagocytosis. Int J Hematol. 2008;87:405-409. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 18. | Yamada S, Tasaki T, Satoh N, Nabeshima A, Kitada S, Noguchi H, Yamada K, Takeshita M, Sasaguri Y. Primary splenic histiocytic sarcoma complicated with prolonged idiopathic thrombocytopenia and secondary bone marrow involvement: a unique surgical case presenting with splenomegaly but non-nodular lesions. Diagn Pathol. 2012;7:143. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5] [Cited by in F6Publishing: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 19. | Yamamoto S, Tsukamoto T, Kanazawa A, Shimizu S, Morimura K, Toyokawa T, Xiang Z, Sakurai K, Fukuoka T, Yoshida K, Takii M, Inoue K. Laparoscopic splenectomy for histiocytic sarcoma of the spleen. World J Gastrointest Surg. 2013;5:129-134. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 5] [Cited by in F6Publishing: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |