Published online Jul 6, 2022. doi: 10.12998/wjcc.v10.i19.6636
Peer-review started: January 5, 2022
First decision: March 7, 2022
Revised: March 19, 2022
Accepted: May 8, 2022
Article in press: May 8, 2022
Published online: July 6, 2022
Retroperitoneal liposarcoma (RLPS) is a rare malignant tumor of the connective tissue and usually grows to a large size, undetected. Diagnosis is currently based on collective findings from clinical examinations and computed tomography (CT) and magnetic resonance imaging, the latter of which show a fat density mass and possible surrounding organ involvement. Surgical resection is the main therapeutic strategy. The efficacy and safety of further therapeutic choices, such as chemotherapy and radiotherapy, are still controversial.
A 61-year-old man presented with complaint of a large left inguinal mass that had appeared suddenly, after a slight exertion. Ultrasonography revealed an omental inguinal hernia. During further clinical examination, an enormous palpable abdominal mass, continuing from the left inguinal location, was observed. CT revealed a giant RLPS, with remarkable mass effect and wide visceral dislocation. After multidisciplinary consultation, surgical intervention was performed. Subsequent neoadjuvant chemotherapy and radiotherapy were precluded by the mass’ large size and retroperitoneal localization, features typically associated with non-response to these types of treatment. Instead, the patient underwent conservative treatment via radical surgical excision. After 1 year, his clinical condition remained good, with no radiological signs of recurrence.
Conservative treatment via surgery resulted in a successful outcome for a large RLPS.
Core Tip: Giant retroperitoneal liposarcoma (RLPS) is a rare condition. The tumor arises from retroperitoneal fatty tissue in the space between the peritoneum and posterior abdominal wall. Owing to the absence of any functional impairment for a long time, RLPS can reach large dimensions before the onset of symptoms. We report the case of a man who received a misdiagnosis of inguinal hernia and was scheduled for intervention. After a careful clinic approach and further radiological examinations, the tumor was removed by radical surgery without any damage to surrounding organs. At the 1-year follow-up no recurrence was present.
- Citation: Lieto E, Cardella F, Erario S, Del Sorbo G, Reginelli A, Galizia G, Urraro F, Panarese I, Auricchio A. Giant retroperitoneal liposarcoma treated with radical conservative surgery: A case report and review of literature. World J Clin Cases 2022; 10(19): 6636-6646
- URL: https://www.wjgnet.com/2307-8960/full/v10/i19/6636.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v10.i19.6636
Soft tissue sarcomas are rare mesenchymal tumors, with an annual incidence of 0.3-0.4 newly diagnosed cases per 100000[1,2]. Stemming from connective tissue cells[3], they represent a heterogeneous family of malignant tumors[2] (Table 1). Since they can arise from any mesenchymal cell line, forming fat, muscle, blood vessels, nerves, and bone tissue[4], a wide variety are currently recognized[5]. Moreover, among the more than 70 different histological subtypes known, most show distinctive clinical behaviors and responses to therapy. The World Health Organization (WHO) classification[3] of these tumors is based upon the specific involved cellular line (with a wide range of different histotypes for each stem cell category), the particular status of cellular differentiation (G), and the tumor’s dimensions[6], which underlie the different biological behaviors cited above[7]. According to the histological features, these tumors are clinically classified as low-grade (stage 1) or high-grade (stage 2 or 3)[8], with progressively worse prognoses.
| No. | World Health Organization classification |
| 1 | Adipocytic tumors |
| 2 | Fibroblastic/myofibroblastic tumors |
| 3 | Fibrohistiocytic tumors |
| 4 | Vascular tumors |
| 5 | Smooth muscle tumors |
| 6 | Skeletal muscle tumors |
| 7 | Gastrointestinal stromal tumors |
| 8 | Chondro-osseous tumors |
| 9 | Peripheral nerve sheath tumors |
| 10 | Tumors of uncertain differentiation |
Although soft tissue sarcomas are ubiquitous among the sexes, they are more frequently diagnosed in adult males[9], with peak of incidence occurring in the 5th-6th decades of life. The most common histotypes are liposarcoma, leiomyosarcoma, leiomyoma, and dedifferentiated pleomorphic sarcoma[10], and the most frequent sites of onset are limbs, trunk, and retroperitoneum[6].
Currently, radical surgery is the cornerstone of the therapeutic strategy[11]. Although this method aims for tumor-free resection margins (R0 resection), the infiltrating nature of the tumor, with possible spreading into different surrounding organs, can make radical resection a daunting challenge for surgeons. Distant metastases are relatively common, occurring in 20%-25% of reported cases[8,12]. Local recurrence is not uncommon, even up to 10 years from the time of radical resection; indeed, the reported frequency of 35% contributes to the low cumulative 10-year survival (46%)[13]. Although, it is important to note here that recurrence and survival are dependent on the anatomical site of origin and the histopathological type of the soft tissue sarcoma[13].
The most common type of soft tissue sarcoma (liposarcoma) represents about 45% of all diagnosed cases[14-16] and in about 30% of those the tumor was determined to have arisen from the retroperitoneum, specifically from adipocytic cells, expanding in the space outside the parietal peritoneum of the posterior abdominal wall[17]. This particular site would allow for a relatively hidden growth of the tumor, in essence enlarging its mass to a greater extent before causing any symptoms[18]. Cellular differentiation[19,20] of the tumor (Table 2) is one of the most impactful prognostic factors, with a better differentiation being predictive of a better outcome usually. Considering this parameter, liposarcomas can be classified into the five subtypes of atypical lipomatous tumor, well-differentiated liposarcoma (WDLPS), dedifferentiated liposarcoma (DDLPS), myxoid liposarcoma (MLPS), pleomorphic liposarcoma (PLPS), and myxoid PLPS, representing progressively worse biological behavior in terms of local recurrence, invasiveness, and response to therapy[19].
| No. | Histopathological classification |
| 1 | Well-differentiated liposarcoma (lipoma-like) |
| 2 | Undifferentiated liposarcoma |
| 3 | Myxoid liposarcoma |
| 4 | Pleomorphic liposarcoma |
| 5 | Myxoid pleomorphic liposarcoma |
The retroperitoneal liposarcoma (RLPS) is usually a WDLPS and typically occurs in adulthood[21] with a completely non-specific symptomatology[22,23]. Prognosis is variable, and 5-year survival rates range widely, from 40% to 90% according to the tumor dimension and histological type[24]. Local recurrence and distant metastases are the main tumor-related causes of death, affecting about 50% of cases by 5 years from diagnosis[25].
The so-called giant liposarcomas reach over 20 kg in weight[13]; the large size and invasive growth towards proximal organs can impact the symptomologic profile. These huge-dimension tumors are associated with significantly lower survival and higher incidences of local recurrence and distant metastasis.
Contrast-enhanced computed tomography (CT) is the gold standard for diagnosis of RLPS[19]. In such, the tumor appears as an encapsulated heterogeneous mass, with alternating areas of density and septa. Magnetic resonance imaging (MRI) can offer a better definition of the tumor’s relationship with surrounding organs[2]. In clinical practice, an RLPS must be suspected when these imaging modalities detect a large-sized, low-density mass localized in the retroperitoneal space that has displaced surrounding structures and shows scarce contrast enhancement[19]. When imaging provides a strong confidence of the RLPS diagnosis[4], direct biopsy can be avoided, which protects the patient against possible tumoral seeding from such[18].
Radical surgery, extended to surrounding involved organs, is the only effective treatment[26] for RLPS, because the reported outcomes of chemotherapy as well as radiotherapy have been inconsistent and incomplete[27,28]; it appears, however, that better outcomes are achieved for soft tissue sarcomas of the limbs[29,30] as opposed to retroperitoneal ones. Regardless, prognosis depends on both the completeness of the surgical resection and the histological subtype itself, with the WDLPS form conferring a greater likelihood for longer survival and a lesser risk for recurrence[22]. However, the importance of margin status of the resected specimen is still under debate[31], given the high recurrence rate (> 40%)[32] observed in cases that received extended resections. Although demolitive surgery has been suggested repeatedly[33-35], less extensive resections are admitted[26,36], since prognosis of RLPS is more likely associated with tumor size and histological differentiation rather than the extent of the resection.
Perioperative radio-chemotherapy[28] has been suggested in more complicated cases, with effective results reported for many. Radiotherapy alone has instead been proposed for local disease control; however, its role is still debated due to the risk of negative effects on renal function and ongoing lack of strong supporting data in the scientific literature[37]. Chemotherapy alone may be effective for cases afflicted with chemo-sensitive subtypes[18], such as MLPS, but the overall small number of treated patients reported in the literature and the generally wide histological variation of soft tissue sarcomas precludes a definitive recommendation. The combination of radio-chemotherapy can be useful in supposed unresectable lesions, and a more effective result may be achieved in cases of soft tissue sarcomas of the extremities and in cases of DDLPS types[26]. Adjuvant radiotherapy has been also proposed for cases of macroscopic or microscopic non-radical surgical resection, in which the invasion of vital structures, such as vena cava or aorta, contraindicates an extensive surgical demolition.
We report herein the case of a giant well-differentiated RLPS that had filled the left abdominal cavity and caused severe visceral dislocation and impaired renal function, penetrating the ipsilateral inguinal canal and scrotum. To the best of our knowledge, this case represents the largest RLPS ever described in the scientific literature. The giant RLPS was initially diagnosed as an inguinal hernia through an incomplete imaging work-up; after further diagnostic assessment, however, it was accurately diagnosed and successfully treated by radical surgery, with an organ-sparing approach. The current clinical case is particularly interesting, in our opinion, because it clearly shows the limited diagnostic power of ultrasonography for retroperitoneal giant sarcoma and the utility of CT scan in more meaningful investigations and decision-making for effective treatment modalities to address and resolve a rare but prognostically-negative disease.
A 61-year-old patient presented with scrotal swelling, abdominal pain, respiratory discomfort, and difficulty moving.
The patient reported that about 1 mo prior, a left inguino-scrotal mass had suddenly appeared after a slight exertion, and was accompanied by a moderate feeling of local tension pain. During a subsequent urological check, the patient had been diagnosed with a gross omental hernia, confirmed by ultrasonography. He had been placed on the waitlist for immediate intervention (a proposed surgical operation for the inguinal hernia). In response, the patient sought a second opinion.
Comorbidities included hypertension, diabetes mellitus, hyperlipidemia, and psoriasis. No previous surgical history was reported.
Nothing relevant was revealed.
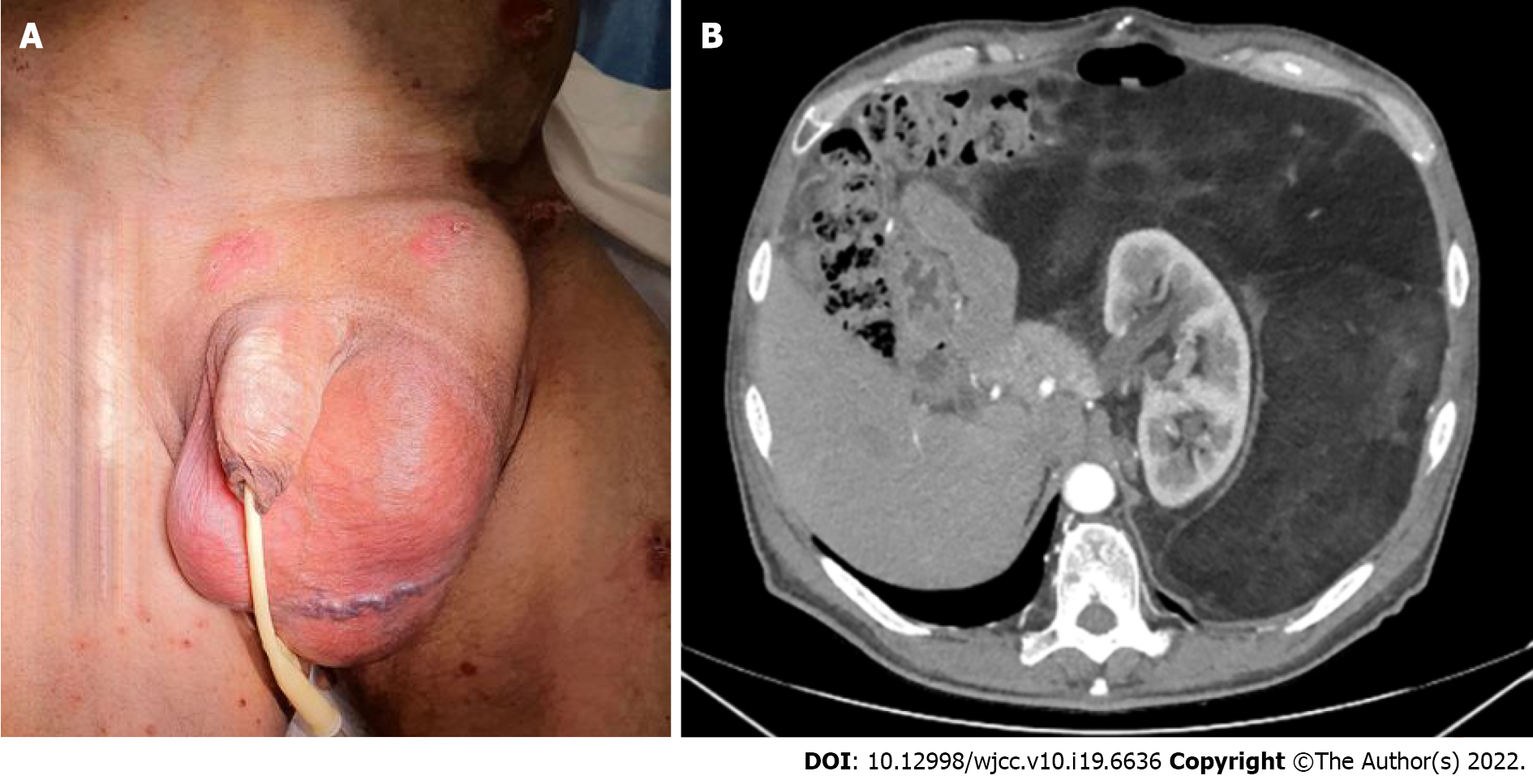
Physical examination detected an obvious huge irreducible inguino-scrotal distension covered by ulcerated skin. A palpable abdominal mass was also found. In both sites, a fixed hard texture mass was evident (Figure 1A). The enormous solid mass did not allow a thorough examination of the abdominal organs. Both pulmonary bases were lifted and poorly mobile.
All laboratory tests, including tumor markers, were normal.
A CT scan of the thorax and abdomen did not show any lesion suspected as metastasis, either in lung or the lymph node stations. On the other hand, the CT scan did reveal a giant retroperitoneal capsulated liposarcoma occupying the left hemi-abdomen and mesogastrium extensively. The tumor, displacing the left kidney anteromedially and digestive structures contralaterally, extended to the ipsilateral inguinal canal. Signal characteristics were consistent with an inhomogeneous fatty mass with internal septa, and there was no excretion of the contrast medium through the left ureter (Figure 1B). Since the imaging findings were strictly consistent with diagnosis of giant RLPS, biopsy was not performed.
The multidisciplinary team consisted of oncologic surgeons and radiologists. Considering the absence of any sign of systemic involvement, the large size of the tumor and the highly symptomatic disease profile, an upfront surgical approach was decided on. Preoperative chemotherapeutic treatment was determined to be unnecessary. Also, preoperative radiotherapy was not considered due to the risk of chronic kidney disease. Given the huge dimensions of the tumor, a laparoscopic approach was excluded.
Giant RLPS with visceral displacement and mechanical obstruction of the left ureter.
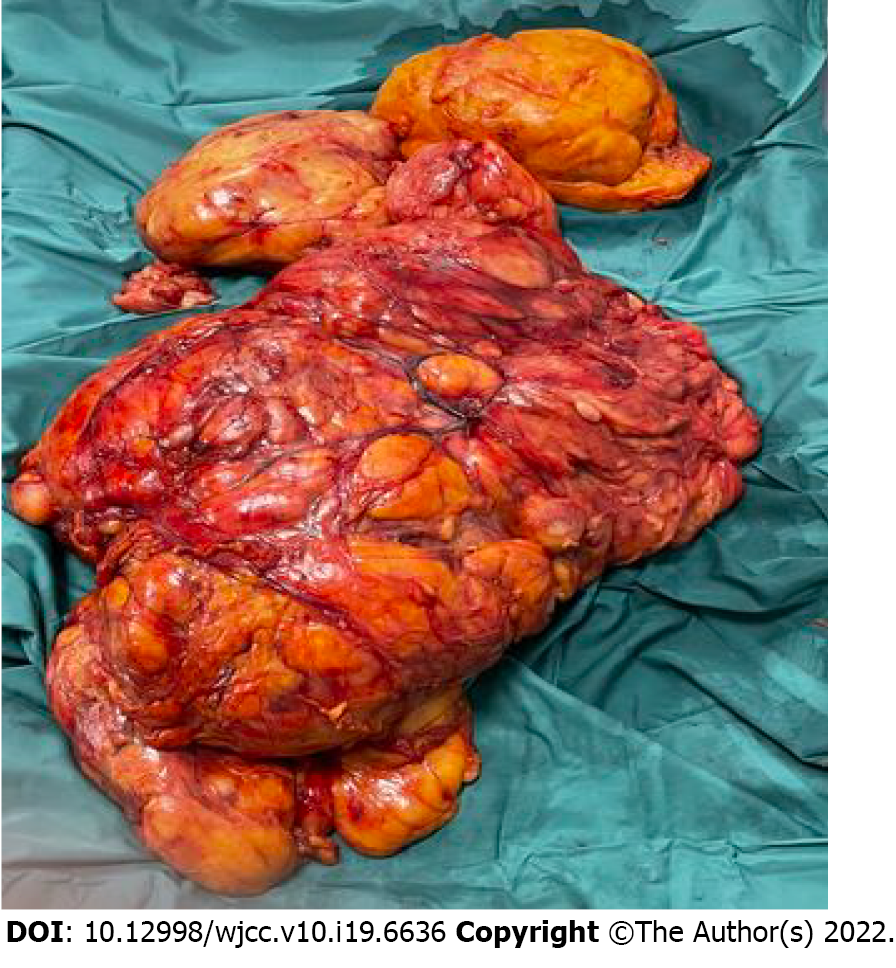
A long median laparotomy was chosen in order to optimize the surgeon’s control over the intraperitoneal organs. An irregularly shaped lesion (70 cm at the largest diameter) was found in the left retroperitoneal space and observed to involve the abdomen, from the diaphragm to the left scrotum. The colon, left kidney and entire bowel were dislocated to the right.
After incision of the left Toldt’s fascia, the entire mass was completely removed (Figure 2) while preserving the integrity of its capsule. The spleen, left kidney and corresponding urinary tract, left spermatic vessels, aorta, left iliac vessels, spermatic cord and left testicle were accurately cleaved from the mass and spared. Bilateral preventive check of the ureters was absolutely mandatory, in order to avoid possible ureteral iatrogenic damage. As both ureters were clearly visible throughout the entire operation, ureteral stenting was not performed; preventive ureteral stenting is usually excluded, in such cases, in order to avoid the risk of complications related to the procedure itself.
A cholecystectomy was also performed to address the concomitant symptomatic cholelithiasis.
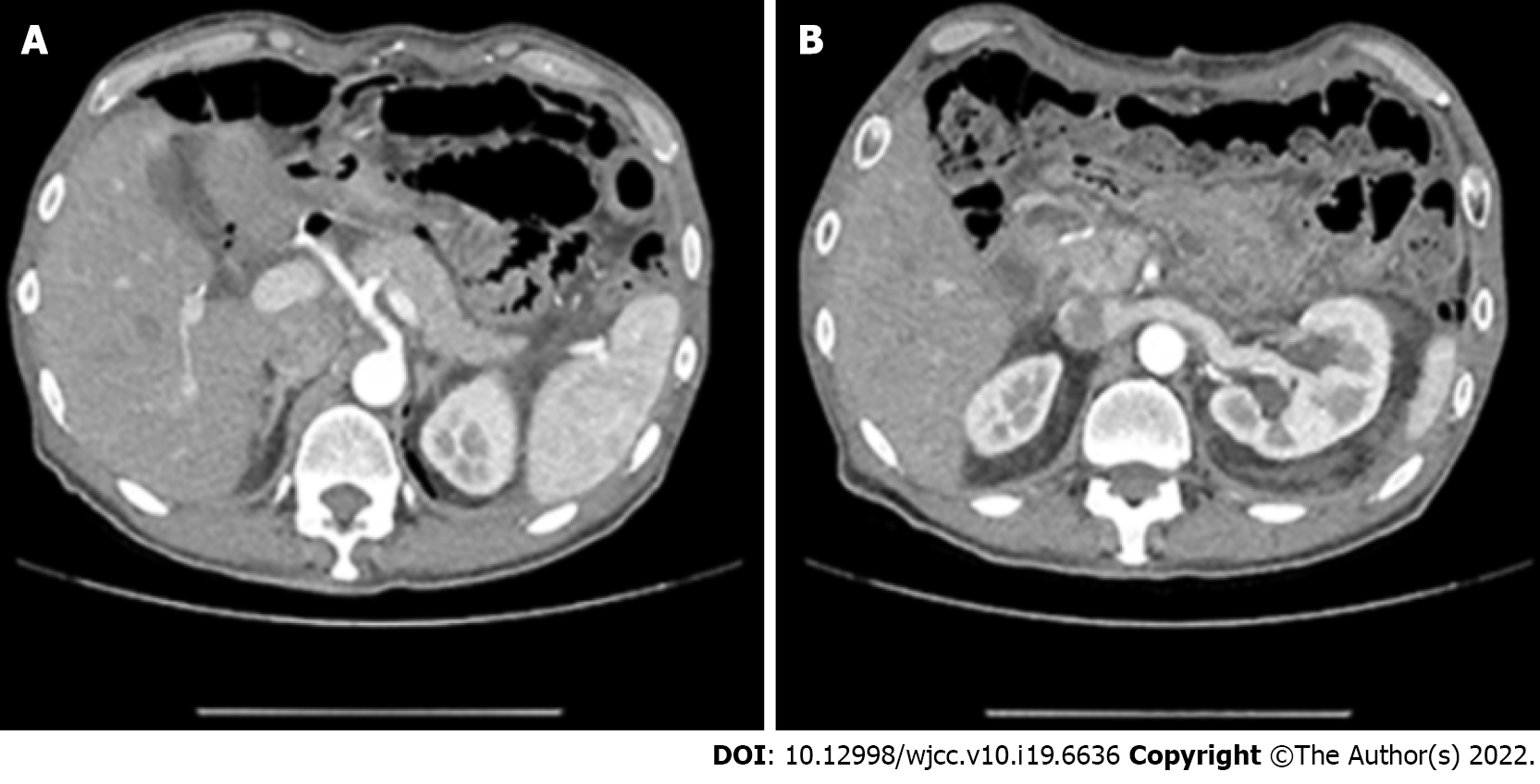
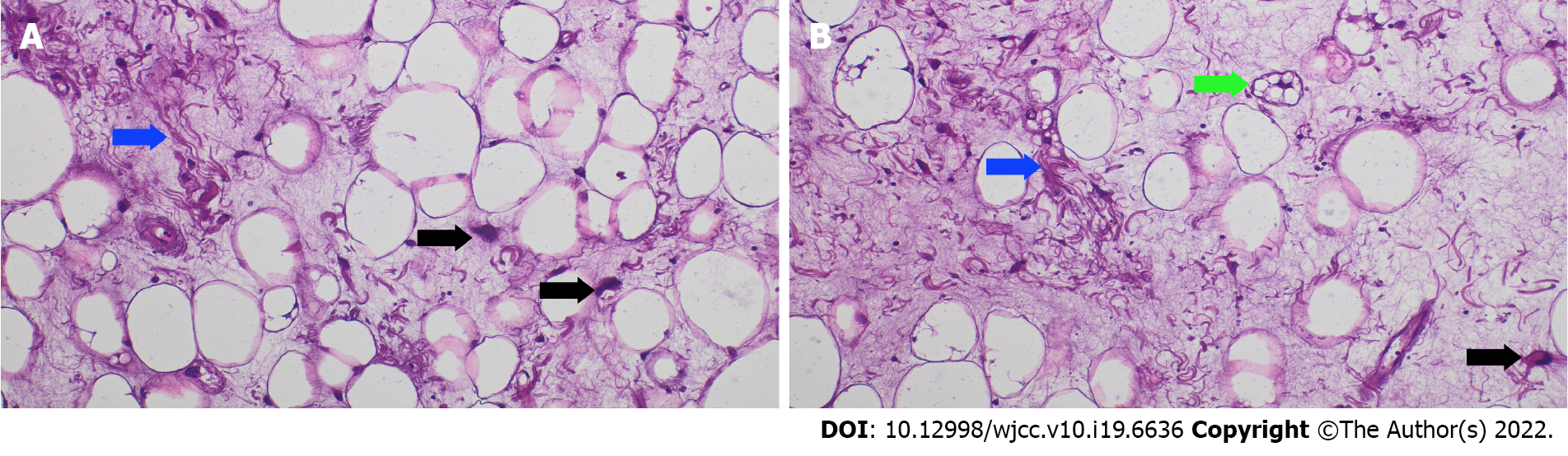
The postoperative course was uneventful. Renal function normalized rapidly, and the patient was discharged on the 7th postoperative day. A further CT scan showed no residual disease and revealed regular function of the left kidney with normal excretion of iodate urine (Figure 3). Pathological examination demonstrated a WDLPS with sclerosing features according to the 5th edition WHO guidelines (Figure 4). Amplification of the Murine Double Minute clone 2 (MDM2) gene was determined by fluorescence in situ hybridization (commonly known as FISH)[20]. The definitive pathological stage was IIIB TNM (pT4, N0, M0–G1).
A strict follow-up with the aim of detecting any possible tumoral recurrence has been proposed, consisting of abdominal ultrasonography and annual contrast-enhanced CT and/or MRI. Because surgical radicality is the most consistent prognostic factor[38] and WDLPSs have a low recurrence rate[12], we are confident that the long-term prognosis for this patient will be good. Indeed, after the 1st year of follow-up, the patient has remained asymptomatic, with CT scan showing negativity for recurrence.
The location of RLPS often causes a late diagnosis, when the tumor has already reached a huge size. The case we present herein, to our knowledge representing the largest sized RLPS reported in the literature with a distinctive clinic history, began with a clinically diagnosed inguinal hernia, confirmed by ultrasonography.
Usually, diagnosis of inguinal hernia is quite easy. Patients present with a reducible mass, without soreness, that corresponds to the external inguinal ring; if any complication (strangulation or hernia clogging) has manifested, pain and/or peritonitic symptomatology complete the clinical presentation. In our case, a correct anamnestic harvesting and a complete clinical evaluation could have immediately demonstrated the contemporary presence of both the inguinal and abdominal mass, and thus the diagnostic mistake could have been avoided.
The ineffectiveness of the ultrasonographic examination was likely due both to the giant dimension of the tumor (more than 70 cm), which did not allow for a complete circumferential visualization, and the predominantly fatty composition, which appeared anechoic. Although the hernia was strongly symptomatic, the patient did not present with abdominal discomfort and/or any abdominal circumference enhancement until the later months; in fact, the tumor had been growing asymptomatically for a long time. The patient had experienced respiratory impairment and some difficulty in normal activities only in the last month before presentation to our clinic.
The latest guidelines[39] suggest that an imaging-guided preoperative diagnosis is mandatory because it can avoid a direct biopsy, thereby preventing hemorrhagic complications or tumoral seeding. The abdomino-pelvic CT scan, which allows for a complete view of the abdominal cavity, gave the correct diagnosis, as it showed the retroperitoneal tumor and its relationship with the surrounding organs. The radiological appearance of RLPS is characterized as an inhomogeneous mass with internal septa and with a complete surrounding capsule. The lack of contrast enhancement, as we observed in the presented case, is due to scarce tumoral vascularization. Imaging features can be used to predict cell differentiation grade[19,40]; for example, the non-enhancement during the arterial phase of CT generally indicates a well-differentiated form[12]. Postoperative histological study confirmed this for the presented case.
Our choice of a primary resection surgery was dictated by the CT images of a giant retroperitoneal encapsulated liposarcoma occupying the left retroperitoneal cavity and the corresponding inguinal canal and displacing the left kidney and intestinal loops. An adjuvant chemotherapy treatment[33] was immediately excluded in the multidisciplinary evaluation because of the dimension of the tumor together with the patient’s impaired clinical condition. According to current practice for clinical management of sarcomas, radiotherapy is mainly indicated as adjuvant therapy when tumor size exceeds 5 cm and an incomplete surgical resection has been performed[41]. In our case, apart from the completeness of the resection and the lack of any histological sign of other-organ invasion, radiotherapy was excluded because of the extensively wide tumoral site which would have necessitated a large irradiation field that would have affected the patient’s renal function. Also, the role of perioperative chemotherapy is still under debate; it has been proposed for high-risk patients (classified according to comorbidities, incomplete resection, organ spreading, and/or cellular dedifferentiation) and has shown some greater benefit in patients with limb and chest sarcomas compared to patients with retroperitoneal sarcomas, and in patients with metastatic disease[11]. Neoadjuvant chemotherapy with antracycline plus ipofosfamide may also represent a therapeutic option for high-risk patients with the aim of achieving preoperative disease downstaging; moreover, doxorubicin is indicated in palliative therapy for metastatic disease[41]. Based on the excellent results of imatinib for treatment of gastrointestinal stromal tumors, immunotherapy may represent a concrete therapeutic alternative for soft tissue sarcomas in the near future[42-44].
Despite the dimension and irregular edge-shape of the mass in our patient, complete removal was possible due to the complete capsulization that allowed for cleavage from the surrounding organs. The lack of any sign of macroscopic infiltration suggested a limited resection with consequent organ sparing. The histological examination confirmed a WDLPS with no sign of capsular invasion.
The MDM2 amplification observed in the resected specimen is an interesting diagnostic aspect of the liposarcoma in our patient. MDM2, a chromosomal 12q15-located oncogene, is the most important negative regulator of p53, the tumor suppressor gene whose mutation is frequently found in many human cancers[45-48]. MDM2 amplification[49] is frequently seen in soft tissue sarcomas, and it can specifically be a hallmark for WDLS, even if the lack of positivity for this marker by genetic testing does not necessarily exclude that diagnosis[50]. Currently, FISH detection of MDM2 amplification is considered adequately sensitive and specific for WDLS diagnosis[51]. In the presence of a p53 mutation, MDM2 promotes cancer growth by enhancing the cell cycle; in absence of a p53 mutation, the overexpression of MDM2 by the tumor cells induces p53 degradation and inactivity[52]. In our case, the MDM2 amplification served to confirm a diagnosis of liposarcoma, which can be difficult at times[3]. Nutlin, an anti-MDM2, is a promising drug for the targeted therapy of liposarcomas[53] and could be an interesting therapeutic choice in case of recurrence.
The advanced disease stage of our case was exclusively due to the tumor’s huge dimension (> 15 cm); otherwise, both the histological type and the differentiation configured a low-grade sarcoma[8], with a limited risk of recurrence[54]. Furthermore, both the absence of any surrounding organ invasion and the complete capsular lining make us confident in a less negative prognostic judgment. The decision for a strict follow-up program should allow us to detect any recurrences in the early stage.
This case is unique due to the dimensions of the Retroperitoneal Sarcoma. To the best of our knowledge, the size and unusual clinical presentation have not been described before. In such cases when an irreducible hernia suddenly appears, complete preoperative imaging is mandatory to reach a correct diagnosis. If the diagnosis of such is based only upon an ultrasonography examination, the patient would be indicated for surgery to simply address an inguinal hernia and not managed for a giant RLPS, which would continue growing unabated. Furthermore, as shown in our case, the CT scan features facilitate the prediction of the tumor’s cell differentiation status and consequently support a relatively better prognosis through a more timely diagnosis and accurate clinical management.
The authors wish to extend a special thanks to Gennaro Galizia for final supervision of this case and its report.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Medicine, research and experimental
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): D, D
Grade E (Poor): 0
P-Reviewer: García-Compeán D, Mexico; Shenoy S, United States; Tian YT, China; Wang G, China S-Editor: Fan JR L-Editor: Filipodia CL P-Editor: Fan JR
| 1. | Hazen B, Cocieru A. Giant Retroperitoneal Sarcoma. J Gastrointest Surg. 2017;21:602-603. [PubMed] [DOI] [Cited in This Article: ] |
| 2. | Peterson JJ, Kransdorf MJ, Bancroft LW, O'Connor MI. Malignant fatty tumors: classification, clinical course, imaging appearance and treatment. Skeletal Radiol. 2003;32:493-503. [PubMed] [DOI] [Cited in This Article: ] |
| 3. | Sbaraglia M, Bellan E, Dei Tos AP. The 2020 WHO Classification of Soft Tissue Tumours: news and perspectives. Pathologica. 2021;113:70-84. [PubMed] [DOI] [Cited in This Article: ] |
| 4. | von Mehren M, Randall RL, Benjamin RS, Boles S, Bui MM, Ganjoo KN, George S, Gonzalez RJ, Heslin MJ, Kane JM, Keedy V, Kim E, Koon H, Mayerson J, McCarter M, McGarry SV, Meyer C, Morris ZS, O'Donnell RJ, Pappo AS, Paz IB, Petersen IA, Pfeifer JD, Riedel RF, Ruo B, Schuetze S, Tap WD, Wayne JD, Bergman MA, Scavone JL. Soft Tissue Sarcoma, Version 2.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16:536-563. [PubMed] [DOI] [Cited in This Article: ] |
| 5. | Fletcher CDM, Bridge JA, Hogendoorn PCW, Mertens F. Editors. WHO Classification of Soft Tissue and Bone. Switzerland: WHO, 2013. [Cited in This Article: ] |
| 6. | Brennan MF, Antonescu CR, Moraco N, Singer S. Lessons learned from the study of 10,000 patients with soft tissue sarcoma. Ann Surg. 2014;260:416-21; discussion 421. [PubMed] [DOI] [Cited in This Article: ] |
| 7. | Korovessis P. Expert's comment concerning Grand Rounds case entitled "Scoliosis correction surgery for patients with McCune-Albright syndrome using pedicle screws: a report of two cases with different characteristics and a review of the literature" (K. Yamane, M. Tanaka, Y. Sugimoto, H. Misawa and T. Ozaki). Eur Spine J. 2015;24:1368-1369. [PubMed] [DOI] [Cited in This Article: ] |
| 8. | Coindre JM, Terrier P, Guillou L, Le Doussal V, Collin F, Ranchère D, Sastre X, Vilain MO, Bonichon F, N'Guyen Bui B. Predictive value of grade for metastasis development in the main histologic types of adult soft tissue sarcomas: a study of 1240 patients from the French Federation of Cancer Centers Sarcoma Group. Cancer. 2001;91:1914-1926. [PubMed] [DOI] [Cited in This Article: ] |
| 9. | Huh WW, Yuen C, Munsell M, Hayes-Jordan A, Lazar AJ, Patel S, Wang WL, Barahmani N, Okcu MF, Hicks J, Debelenko L, Spunt SL. Liposarcoma in children and young adults: a multi-institutional experience. Pediatr Blood Cancer. 2011;57:1142-1146. [PubMed] [DOI] [Cited in This Article: ] |
| 10. | Blay JY, Honoré C, Stoeckle E, Meeus P, Jafari M, Gouin F, Anract P, Ferron G, Rochwerger A, Ropars M, Carrere S, Marchal F, Sirveaux F, Di Marco A, Le Nail LR, Guiramand J, Vaz G, Machiavello JC, Marco O, Causeret S, Gimbergues P, Fiorenza F, Chaigneau L, Guillemin F, Guilloit JM, Dujardin F, Spano JP, Ruzic JC, Michot A, Soibinet P, Bompas E, Chevreau C, Duffaud F, Rios M, Perrin C, Firmin N, Bertucci F, Le Pechoux C, Le Loarer F, Collard O, Karanian-Philippe M, Brahmi M, Dufresne A, Dupré A, Ducimetière F, Giraud A, Pérol D, Toulmonde M, Ray-Coquard I, Italiano A, Le Cesne A, Penel N, Bonvalot S. Surgery in reference centers improves survival of sarcoma patients: a nationwide study. Ann Oncol. 2019;30:1407. [PubMed] [DOI] [Cited in This Article: ] |
| 11. | Bourcier K, Le Cesne A, Tselikas L, Adam J, Mir O, Honore C, de Baere T. Basic Knowledge in Soft Tissue Sarcoma. Cardiovasc Intervent Radiol. 2019;42:1255-1261. [PubMed] [DOI] [Cited in This Article: ] |
| 12. | Gamboa AC, Gronchi A, Cardona K. Soft-tissue sarcoma in adults: An update on the current state of histiotype-specific management in an era of personalized medicine. CA Cancer J Clin. 2020;70:200-229. [PubMed] [DOI] [Cited in This Article: ] |
| 13. | Gronchi A, Strauss DC, Miceli R, Bonvalot S, Swallow CJ, Hohenberger P, Van Coevorden F, Rutkowski P, Callegaro D, Hayes AJ, Honoré C, Fairweather M, Cannell A, Jakob J, Haas RL, Szacht M, Fiore M, Casali PG, Pollock RE, Raut CP. Variability in Patterns of Recurrence After Resection of Primary Retroperitoneal Sarcoma (RPS): A Report on 1007 Patients From the Multi-institutional Collaborative RPS Working Group. Ann Surg. 2016;263:1002-1009. [PubMed] [DOI] [Cited in This Article: ] |
| 14. | Linch M, Miah AB, Thway K, Judson IR, Benson C. Systemic treatment of soft-tissue sarcoma-gold standard and novel therapies. Nat Rev Clin Oncol. 2014;11:187-202. [PubMed] [DOI] [Cited in This Article: ] |
| 15. | Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7-30. [PubMed] [DOI] [Cited in This Article: ] |
| 16. | Xie TH, Ren XX, Fu Y, Ha SN, Liu LT, Jin XS. Multiple well-differentiated retroperitoneal liposarcomas with different patterns of appearance on computed tomography: A case report. World J Clin Cases. 2021;9:1661-1667. [PubMed] [DOI] [Cited in This Article: ] |
| 17. | Osman S, Lehnert BE, Elojeimy S, Cruite I, Mannelli L, Bhargava P, Moshiri M. A comprehensive review of the retroperitoneal anatomy, neoplasms, and pattern of disease spread. Curr Probl Diagn Radiol. 2013;42:191-208. [PubMed] [DOI] [Cited in This Article: ] |
| 18. | Xiao J, Liu J, Chen M, Liu W, He X. Diagnosis and Prognosis of Retroperitoneal Liposarcoma: A Single Asian Center Cohort of 57 Cases. J Oncol. 2021;2021:7594027. [PubMed] [DOI] [Cited in This Article: ] |
| 19. | Haddox CL, Riedel RF. Recent advances in the understanding and management of liposarcoma. Fac Rev. 2021;10:1. [PubMed] [DOI] [Cited in This Article: ] |
| 20. | WHO classification of tumors Editorial Board. WHO classification of tumours of soft tissue and bone, 5th ed Lyon, France; IARC Press; 2020. [Cited in This Article: ] |
| 21. | Hashimoto Y, Hatakeyama S, Tachiwada T, Yoneyama T, Koie T, Kamimura N, Yanagisawa T, Hakamada K, Ohyama C. Surgical treatment of a giant liposarcoma in a Japanese man. Adv Urol. 2010;2010:943073. [PubMed] [DOI] [Cited in This Article: ] |
| 22. | Vijay A, Ram L. Retroperitoneal liposarcoma: a comprehensive review. Am J Clin Oncol. 2015;38:213-219. [PubMed] [DOI] [Cited in This Article: ] |
| 23. | Chouairy CJ, Abdul-Karim FW, MacLennan GT. Retroperitoneal liposarcoma. J Urol. 2007;177:1145. [PubMed] [DOI] [Cited in This Article: ] |
| 24. | Park JO, Qin LX, Prete FP, Antonescu C, Brennan MF, Singer S. Predicting outcome by growth rate of locally recurrent retroperitoneal liposarcoma: the one centimeter per month rule. Ann Surg. 2009;250:977-982. [PubMed] [DOI] [Cited in This Article: ] |
| 25. | Nathan H, Raut CP, Thornton K, Herman JM, Ahuja N, Schulick RD, Choti MA, Pawlik TM. Predictors of survival after resection of retroperitoneal sarcoma: a population-based analysis and critical appraisal of the AJCC staging system. Ann Surg. 2009;250:970-976. [PubMed] [DOI] [Cited in This Article: ] |
| 26. | Saponara M, Stacchiotti S, Gronchi A. Pharmacological therapies for Liposarcoma. Expert Rev Clin Pharmacol. 2017;10:361-377. [PubMed] [DOI] [Cited in This Article: ] |
| 27. | Suarez-Kelly LP, Baldi GG, Gronchi A. Pharmacotherapy for liposarcoma: current state of the art and emerging systemic treatments. Expert Opin Pharmacother. 2019;20:1503-1515. [PubMed] [DOI] [Cited in This Article: ] |
| 28. | Bonvalot S, Gronchi A, Le Péchoux C, Swallow CJ, Strauss D, Meeus P, van Coevorden F, Stoldt S, Stoeckle E, Rutkowski P, Rastrelli M, Raut CP, Hompes D, De Paoli A, Sangalli C, Honoré C, Chung P, Miah A, Blay JY, Fiore M, Stelmes JJ, Dei Tos AP, Baldini EH, Litière S, Marreaud S, Gelderblom H, Haas RL. Preoperative radiotherapy plus surgery versus surgery alone for patients with primary retroperitoneal sarcoma (EORTC-62092: STRASS): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2020;21:1366-1377. [PubMed] [DOI] [Cited in This Article: ] |
| 29. | Kasper B, Ho AD, Egerer G. Is there an indication for high-dose chemotherapy in the treatment of bone and soft-tissue sarcoma? Oncology. 2005;68:115-121. [PubMed] [DOI] [Cited in This Article: ] |
| 30. | Pasquali S, Gronchi A. Neoadjuvant chemotherapy in soft tissue sarcomas: latest evidence and clinical implications. Ther Adv Med Oncol. 2017;9:415-429. [PubMed] [DOI] [Cited in This Article: ] |
| 31. | Howard JH, Pollock RE. Intra-Abdominal and Abdominal Wall Desmoid Fibromatosis. Oncol Ther. 2016;4:57-72. [PubMed] [DOI] [Cited in This Article: ] |
| 32. | Sun P, Ma R, Liu G, Wang L, Chang H, Li Y. Pathological prognostic factors of retroperitoneal liposarcoma: comprehensive clinicopathological analysis of 124 cases. Ann Transl Med. 2021;9:574. [PubMed] [DOI] [Cited in This Article: ] |
| 33. | Kuperus JM, Steensma MR, Khachaturov V, Lane BR. Surgical management of a large retroperitoneal liposarcoma: A case study. Urol Case Rep. 2021;34:101502. [PubMed] [DOI] [Cited in This Article: ] |
| 34. | Fairweather M, Gonzalez RJ, Strauss D, Raut CP. Current principles of surgery for retroperitoneal sarcomas. J Surg Oncol. 2018;117:33-41. [PubMed] [DOI] [Cited in This Article: ] |
| 35. | Gronchi A, Lo Vullo S, Fiore M, Mussi C, Stacchiotti S, Collini P, Lozza L, Pennacchioli E, Mariani L, Casali PG. Aggressive surgical policies in a retrospectively reviewed single-institution case series of retroperitoneal soft tissue sarcoma patients. J Clin Oncol. 2009;27:24-30. [PubMed] [DOI] [Cited in This Article: ] |
| 36. | Yokoyama Y, Nishida Y, Ikuta K, Nagino M. A case of retroperitoneal dedifferentiated liposarcoma successfully treated by neoadjuvant chemotherapy and subsequent surgery. Surg Case Rep. 2020;6:105. [PubMed] [DOI] [Cited in This Article: ] |
| 37. | Bachmann R, Eckert F, Gelfert D, Strohäker J, Beltzer C, Ladurner R. Perioperative strategy and outcome in giant retroperitoneal dedifferentiated liposarcoma-results of a retrospective cohort study. World J Surg Oncol. 2020;18:296. [PubMed] [DOI] [Cited in This Article: ] |
| 38. | Mussi C, Collini P, Miceli R, Barisella M, Mariani L, Fiore M, Casali PG, Gronchi A. The prognostic impact of dedifferentiation in retroperitoneal liposarcoma: a series of surgically treated patients at a single institution. Cancer. 2008;113:1657-1665. [PubMed] [DOI] [Cited in This Article: ] |
| 39. | von Mehren M, Kane JM, Bui MM, Choy E, Connelly M, Dry S, Ganjoo KN, George S, Gonzalez RJ, Heslin MJ, Homsi J, Keedy V, Kelly CM, Kim E, Liebner D, McCarter M, McGarry SV, Meyer C, Pappo AS, Parkes AM, Paz IB, Petersen IA, Poppe M, Riedel RF, Rubin B, Schuetze S, Shabason J, Sicklick JK, Spraker MB, Zimel M, Bergman MA, George GV. NCCN Guidelines Insights: Soft Tissue Sarcoma, Version 1.2021. J Natl Compr Canc Netw. 2020;18:1604-1612. [PubMed] [DOI] [Cited in This Article: ] |
| 40. | Messiou C, Moskovic E, Vanel D, Morosi C, Benchimol R, Strauss D, Miah A, Douis H, van Houdt W, Bonvalot S. Primary retroperitoneal soft tissue sarcoma: Imaging appearances, pitfalls and diagnostic algorithm. Eur J Surg Oncol. 2017;43:1191-1198. [PubMed] [DOI] [Cited in This Article: ] |
| 41. | Casali PG, Abecassis N, Aro HT, Bauer S, Biagini R, Bielack S, Bonvalot S, Boukovinas I, Bovee JVMG, Brodowicz T, Broto JM, Buonadonna A, De Álava E, Dei Tos AP, Del Muro XG, Dileo P, Eriksson M, Fedenko A, Ferraresi V, Ferrari A, Ferrari S, Frezza AM, Gasperoni S, Gelderblom H, Gil T, Grignani G, Gronchi A, Haas RL, Hassan B, Hohenberger P, Issels R, Joensuu H, Jones RL, Judson I, Jutte P, Kaal S, Kasper B, Kopeckova K, Krákorová DA, Le Cesne A, Lugowska I, Merimsky O, Montemurro M, Pantaleo MA, Piana R, Picci P, Piperno-Neumann S, Pousa AL, Reichardt P, Robinson MH, Rutkowski P, Safwat AA, Schöffski P, Sleijfer S, Stacchiotti S, Sundby Hall K, Unk M, Van Coevorden F, van der Graaf WTA, Whelan J, Wardelmann E, Zaikova O, Blay JY; ESMO Guidelines Committee and EURACAN. Soft tissue and visceral sarcomas: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29:iv51-iv67. [PubMed] [DOI] [Cited in This Article: ] |
| 42. | Mir O, Brodowicz T, Italiano A, Wallet J, Blay JY, Bertucci F, Chevreau C, Piperno-Neumann S, Bompas E, Salas S, Perrin C, Delcambre C, Liegl-Atzwanger B, Toulmonde M, Dumont S, Ray-Coquard I, Clisant S, Taieb S, Guillemet C, Rios M, Collard O, Bozec L, Cupissol D, Saada-Bouzid E, Lemaignan C, Eisterer W, Isambert N, Chaigneau L, Cesne AL, Penel N. Safety and efficacy of regorafenib in patients with advanced soft tissue sarcoma (REGOSARC): a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Oncol. 2016;17:1732-1742. [PubMed] [DOI] [Cited in This Article: ] |
| 43. | Gounder MM, Mahoney MR, Van Tine BA, Ravi V, Attia S, Deshpande HA, Gupta AA, Milhem MM, Conry RM, Movva S, Pishvaian MJ, Riedel RF, Sabagh T, Tap WD, Horvat N, Basch E, Schwartz LH, Maki RG, Agaram NP, Lefkowitz RA, Mazaheri Y, Yamashita R, Wright JJ, Dueck AC, Schwartz GK. Sorafenib for Advanced and Refractory Desmoid Tumors. N Engl J Med. 2018;379:2417-2428. [PubMed] [DOI] [Cited in This Article: ] |
| 44. | Bourcier K, Italiano A. Newer therapeutic strategies for soft-tissue sarcomas. Pharmacol Ther. 2018;188:118-123. [PubMed] [DOI] [Cited in This Article: ] |
| 45. | Galizia G, Ferraraccio F, Lieto E, Orditura M, Castellano P, Imperatore V, Romano C, Vollaro M, Agostini B, Pignatelli C, De Vita F. Prognostic value of p27, p53, and vascular endothelial growth factor in Dukes A and B colon cancer patients undergoing potentially curative surgery. Dis Colon Rectum. 2004;47:1904-1914. [PubMed] [DOI] [Cited in This Article: ] |
| 46. | Cissé MY, Pyrdziak S, Firmin N, Gayte L, Heuillet M, Bellvert F, Fuentes M, Delpech H, Riscal R, Arena G, Chibon F, Le Gellec S, Maran-Gonzalez A, Chateau MC, Theillet C, Carrere S, Portais JC, Le Cam L, Linares LK. Targeting MDM2-dependent serine metabolism as a therapeutic strategy for liposarcoma. Sci Transl Med. 2020;12. [PubMed] [DOI] [Cited in This Article: ] |
| 47. | Kommoss FK, Chang KT, Stichel D, Banito A, Jones DT, Heilig CE, Fröhling S, Sahm F, Stenzinger A, Hartmann W, Mechtersheimer G, Sinn HP, Schmidt D, Kommoss F, von Deimling A, Koelsche C. Endometrial stromal sarcomas with BCOR-rearrangement harbor MDM2 amplifications. J Pathol Clin Res. 2020;6:178-184. [PubMed] [DOI] [Cited in This Article: ] |
| 48. | Kato S, Ross JS, Gay L, Dayyani F, Roszik J, Subbiah V, Kurzrock R. Analysis of MDM2 Amplification: Next-Generation Sequencing of Patients With Diverse Malignancies. JCO Precis Oncol. 2018;2018. [PubMed] [DOI] [Cited in This Article: ] |
| 49. | Kulkarni AS, Wojcik JB, Chougule A, Arora K, Chittampalli Y, Kurzawa P, Mullen JT, Chebib I, Nielsen GP, Rivera MN, Ting DT, Deshpande V. MDM2 RNA In Situ Hybridization for the Diagnosis of Atypical Lipomatous Tumor: A Study Evaluating DNA, RNA, and Protein Expression. Am J Surg Pathol. 2019;43:446-454. [PubMed] [DOI] [Cited in This Article: ] |
| 50. | Sciot R. MDM2 Amplified Sarcomas: A Literature Review. Diagnostics (Basel). 2021;11. [PubMed] [DOI] [Cited in This Article: ] |
| 51. | Kimura H, Dobashi Y, Nojima T, Nakamura H, Yamamoto N, Tsuchiya H, Ikeda H, Sawada-Kitamura S, Oyama T, Ooi A. Utility of fluorescence in situ hybridization to detect MDM2 amplification in liposarcomas and their morphological mimics. Int J Clin Exp Pathol. 2013;6:1306-1316. [PubMed] [DOI] [Cited in This Article: ] |
| 52. | Momand J, Wu HH, Dasgupta G. MDM2--master regulator of the p53 tumor suppressor protein. Gene. 2000;242:15-29. [PubMed] [DOI] [Cited in This Article: ] |
| 53. | Wang W, Qin JJ, Rajaei M, Li X, Yu X, Hunt C, Zhang R. Targeting MDM2 for novel molecular therapy: Beyond oncology. Med Res Rev. 2020;40:856-880. [PubMed] [DOI] [Cited in This Article: ] |
| 54. | Singal R, Trehan S, Mittal A, Roy M, Kusum J. Sarcoma retroperitoneal gigante: patologia actual y exito quirurgico. Acta Gastroeneterologica Latino-Americana, 2018. [Cited in This Article: ] |