Published online Jul 18, 2013. doi: 10.5312/wjo.v4.i3.114
Revised: May 21, 2013
Accepted: June 5, 2013
Published online: July 18, 2013
The incidence of cancer is increasing worldwide, with the advent of a myriad of new treatment options, so is the overall survival of these patients. However, from an orthopaedic perspective, there comes the challenge of treating more patients with a variety of metastatic bone lesions. The consequences of such lesions can be significant to the patient, from pain and abnormal blood results, including hypercalcemia, to pathological fracture. Given the multiple options available, the treatment of bone metastasis should be based on a patient-by patient manner, as is the case with primary bone lesions. It is imperative, given the various lesion types and locations, treatment of bone metastasis should be performed in an individualised manner. We should consider the nature of the lesion, the effect of treatment on the patient and the overall outcome of our decisions. The dissemination of primary lesions to distant sites is a complex pathway involving numerous cytokines within the tumour itself and the surrounding microenvironment. To date, it is not fully understood and we still base a large section of our knowledge on Pagets historic “seed and soil” theory. As we gain further understanding of this pathway it will allow us develop more medical based treatments. The treatment of primary cancers has long been provided in a multi-disciplinary setting to achieve the best patient outcomes. This should also be true for the treatment of bone metastases. Orthopaedic surgeons should be involved in the multidisciplinary treatment of such patients given that there are a variety of both surgical fixation methods and non-operative methods at our disposal.
Core tip: This paper discusses the pathophysiology and patient implications of bone metastasis. We aim to describe the orthopaedic input into the management of this condition, especially in a multi-disciplinary setting. We believe that orthopaedics do not have a significant enough involvement in the treatment of long bone metastasis, although from this paper we feel we have many options to offer. The future of metastasis treatment may be targeted at the molecular level but current management options do require an understanding of musculoskeletal oncology to obtain best patient outcomes through operative and non-operative means.
- Citation: Molloy AP, O’Toole GC. Orthopaedic perspective on bone metastasis. World J Orthop 2013; 4(3): 114-119
- URL: https://www.wjgnet.com/2218-5836/full/v4/i3/114.htm
- DOI: https://dx.doi.org/10.5312/wjo.v4.i3.114
The American Cancer Society estimates that 1.64 million cases of cancer will be diagnosed in the United States in 2012[1]. Approximately 50% of these cases involve tumours of the breast, prostate, lung, kidney and thyroid. These tumours commonly metastasize to bone and account for 80% of all skeletal metastases[2]. This compares to an estimated 2890 cases of primary bone tumours that will be diagnosed during the same period[1]. These figures emphasize the importance of being able to recognise, investigate, manage and intervene appropriately in the course of metastatic disease in order to preserve function and quality of life while minimizing complications.
The microenvironment associated with bone is ideal for tumour progression. Bone is a highly vascular mineral which produces adhesion molecules and is a source of angiogenic and bone resorbing molecules, all of which are conducive to the spread and development of tumours[3,4]. It also contains immobilised growth factors, which, when released, further enhance tumour cell proliferation[3,5].
Earliest evidence of skeletal metastases dates from 400 BC[6]. The term “metastasis” was first used by Hippocratic physicians, is of Greek origin and means the “change in the seat of a disease”.
Throughout the 19th century, further investigation was carried out to elicit the mechanism behind the development of metastases. This era gave rise to pagets “seed and soil” theory, which postulated that tissues (“soil”) receiving tumour cells could either be congenial or hostile[7]. The preferential development of bone metastases first postulated by Stephen Paget in 1889 noted that “in a cancer of the breast the bones suffer in a special way, which cannot be explained by any theory of embolism alone”. Hence he proposed the widely acknowledged “seed and soil hypothesis”. Circulating disseminated cancer cells activate bone to provide the ideal “soil” in which the aforementioned “seeds” can grow.
Others explained metastases on a purely stochastic basis[8]. It is now accepted that both methods occur, approximately 60% of metastatic sites can be predicted on a purely haematological and/or lymphatic route basis, the remainder of metastases involve intricate interactions between the tumour and host sites at the cellular and molecular level.
The identification of bone metastases is a significant development for patients. Their treatment can change completely as does their outcome. Not only can this news have a physical effect on patients’ lives but also an emotional effect. As the treatment of metastatic disease is multidisciplinary in nature, it is imperative that orthopaedic surgeons are involved at an early stage and not just following pathological fracture or the development spinal stenosis.
Bone metastases and their associated complications (bone pain, pathological fractures, spinal cord compression, loss of independence and mobility and abnormal electrolytes) are the major morbidities associated with advanced disease and the symptoms with which patients will present[9].
The critical aspect in the investigation of a patient with potential metastases involves recognition of the above symptoms as possible progression of the primary tumour to bone. Imaging has an important role in the detection, diagnosis, prognostication, treatment planning, and follow-up monitoring of bone metastases.
Despite the relative insensitivity of plain radiographs in detecting small or early metastases, initial investigations should always include plain radiography. The presence of sclerosis or osteolysis on the X-ray can aid in diagnosis of the metastatic lesion, with sclerosis typically indicating a prostatic lesion and osteolytic lesions secondary to a breast primary[9,10].
Technetium-99m (99mTc) bone scintiscanning (i.e., radionuclide bone scanning) is the most cost-effective and widely available whole-body screening test for the assessment of bone metastases. Combined analysis with plain radiography and 99mTc bone scintiscans improves diagnostic accuracy in detecting bone metastases and assessing the response to therapy[10].
Computed tomography (CT) scanning is an invaluable modality in those cases where bone scan confirms a focal abnormality but plain radiography cannot confirm any metastases. All bony metastatic lesions are depicted well on CT, including those of an osteoblastic, osteolytic and mixed nature.
Despite the expensive nature of the modality, magnetic resonance imaging (MRI) is very sensitive in the detection of metastases. Although some studies have suggested whole body MRI as a possible alternative to 99mTc bone scanning in the skeletal evaluation of bone metastases, this would be an impractical and expensive choice. MRI is primarily used in the evaluation of vertebral metastases for spinal cord compression or soft tissue involvement[11].
Histological diagnosis of metastases can be obtained from core biopsy of the effected bone, or CT guided biopsy should the former prove difficult. Alternatively, in the case of a patient presenting with a pathological fracture in the setting of known metastases, bone reamings at the time of surgical fixation can also be histologically analysed for the presence of circulating tumour cells.
Metastasis involves a number of complex cell-cell interactions that ultimately leads to the development and growth of cancer cells in a distant visceral or bony site. Cells from the primary tumour must detach and extravasate. Following this they must migrate through the endothelium into the surrounding blood vessels, attach to the endothelium of a distant site after surviving the turbulent arterial blood supply, then migrate through the endothelium and extracellular matrix of the distant organ. Finally these circulating tumour cells must develop in the distant organ and facilitate the growth of further cancer cells[12].
The capacity to enter the circulation requires that neoplastic cells must have intrinsic properties that facilitate this process. The tumour cell must have the ability to induce neovascularization and be capable of crossing from the tumour stroma to the vasculature by invading the basement membrane of the vascular endothelium[13]. This process is facilitated by cell adhesion molecules (CAMs). Several categories of CAMs exist, including intercellular adhesion molecules (ICAMs), selectins and cadherins[14,15].
Once in the circulation, embolization of the tumour cell is facilitated by adhesion to P- and L-selectins, located on platelets and leucocytes respectively. Adhesion to the endothelium of the metastatic tissue is mediated via E-selectin. Upon adhesion, an integrin signalling pathway is initiated, the net result of which is up-regulation of both the anti-apoptotic machinery and proteolytic activity in the microenvironment, thus facilitating the extravasation of tumour cells out of the circulation, and their invasion into the host tissue[16].
Disseminated tumour cells also contain integrins, a transmembrane receptor family which allows their attachment to several peptide sequences present on certain bone matrix proteins. These cell-surface molecules are involved in signal transduction and have been implicated in the mediation of cell migration, differentiation and apoptosis. Many studies have shown the correlation of increased integrin expression with malignant potential[17].
While our understanding of the molecular mechanisms of metastases has improved significantly since the earliest observations of Billroth[18], we remain ignorant of the intricacies of metastases. Selective therapeutic agents targeted exclusively at metastatic cells have yet to be developed[19] and much remains to be discovered about the critical determinants of metastatic process. However, the accelerated advances in the fields of molecular biology and genetics augurs well for the future.
Current medical options for the treatment of bone metastases primarily involves the use of bisphosphonates[20]. These are potent inhibitors of osteoclast activity and bone resorption and are widely used in both metabolic bone disease and metastatic disease. The mechanism of action of bisphosphonates targets the key stage of metastatic development where the disseminated circulating tumour cell stimulates further bone resorption. The disruption of this interaction, for either a palliative or preventative means, decreases the amplification of the metastatic process.
More recent studies have investigated the direct anti-tumour effects of bisphosphonates. It is believed that along with their inhibition of bone resorption, bisphosphonates may induce apoptosis of certain disseminated cancer cells, such as breast cancer cells. Furthermore, it is now believed from in vivo studies that bisphosphonates alter the properties of adhesion molecules in the bone matrix thus inhibiting the direct attachment of circulating tumour cells to the bone microenvironment[12,20]. However, despite their benefits in the treatment of symptomatic metastatic disease, they have not improved survival in patients with bone metastases[20].
There is no strict rationale governing the surgical management of skeletal metastases. Clinical, medical, radiological and surgical factors, coupled with the inherent biology of the primary tumour all contribute to the decision making process. Furthermore, surgery in the setting of metastatic disease requires reliable data about patient survival and quality of life[21]. Earliest recommendations were simple and called for surgical intervention in the clinical scenario where fractures were “predicable”[22], the idea that the patient should have a reasonable life expectancy before considering surgery is relatively new[23].
Protocols for the treatment of bone metastases of the appendicular skeleton have been published. Mirels described a weighted scoring system in an attempt at quantifying the risk of sustaining a pathological fracture[24] and consequently the relative urgency for orthopaedic surgical intervention (Table 1). Mirels’ system remains in wide use today despite the recent introduction of newer protocols[25]. The newer system describes the guidelines to surgical indications, types of surgery and recommended implants. Capanna classifies all patients with bony metastases into 4 categories (Table 2). Patients accumulate a representative numerical value for their metastatic lesion depending on figures awarded for potential survival (Table 3), the size of the lesion, the biomechanics of the bone involved and the potential response to adjuvant therapy. This dictates the recommended surgical intervention and the prosthetic implant to be used (Table 4).
| Variable | Score | ||
| 1 | 2 | 3 | |
| Site | Upper Limb | Lower Limb | Peritrochanter |
| Pain | Mild | Moderate | Functional |
| Lesion | Blastic | Mixed | Lytic |
| Size | < 1/3 | 1/3-2/3 | > 2/3 |
| Classification according to tumour type | |
| Class 1 | Solitary metastatic lesion |
| Primary with good prognosis | |
| Interval after primary over 3 years | |
| Class 2 | Pathological fracture at any site |
| Class 3 | Impending fracture in a major weight bearing bone |
| Class 4 | Osteoblastic lesions at all sites |
| Osteolytic or mixed lesions in non-structural bones | |
| Osteolytic lesion with no impending fracture in major weight-bearing bone | |
| Lesions of the iliac wing, anterior pelvis or scapula | |
| Survival | Sources of metastasis |
| < 1 yr (1 point) | Unknown Primary |
| Melanoma | |
| Lung | |
| Pancreas | |
| Thyroid (undifferentiated) | |
| Stomach | |
| 1-2 yr (3 points) | Colon |
| Breast (not responding to adjuvants) | |
| Liver | |
| Uterus (responding to adjuvants) | |
| > 2 yr (6 points) | Thyroid (differentiated) |
| Myeloma | |
| Lymphoma | |
| Breast (responding to adjuvants) | |
| Rectum | |
| Prostate | |
| Kidney |
| Survival | Biomechanics | Size defect | Response to adjuvant therapy |
| < 1 yr = 1 pts | Tibia = 1pt | Small (1/3) = 1 pt | Yes = 0 |
| 1-2 yr = 3 pts | Femur, humerus = 2 pts | Large (1/2) = 2 pts | No = 3 |
| > 2 yr = 6 pts | Subtrochanteric, supracondylar = 3 pts | Defective or pathological fracture = 3 pts | |
| < 5 points = | Minimal or simple osteosynthesis | ||
| 5-10 points = | Reinforced osteosynthesis | ||
| 10-15 points = | Megaprosthesis or intercalary spacer |
However, despite involved classification systems and resultant surgical recommendations, each case of metastatic disease warrants treatment on an individual basis. Huge strides have been made in the techniques of surgical management for achieving secure fixation of pathological fractures despite what is often extensive bony destruction[26,27]. The use of internal fixation devices and prostheses along with methyl methacrylate has greatly assisted the orthopaedic surgeon in managing pathological fractures. Despite improved fixation, healing of pathological fractures remains a challenge, overall 35% of pathological fractures can be expected to heal in 6 mo[28], highest healing rates are seen with multiple myeloma (67%) and the lowest rates were seen with lung carcinoma (0%).
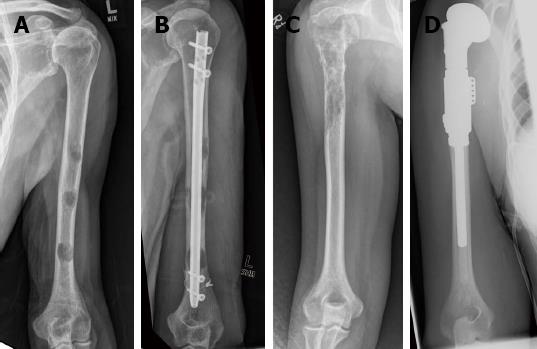
It is imperative that whatever fixation device is used the construct should be durable enough to reliably last the remainder of the patient’s life expectancy[29] and it is our recommendation that the entire diseased bone be stabilised at one operative sitting (Figure 1).
Although the general orthopaedic surgeon will commonly deal with pathological fractures in their day to day practice, we are commonly referred patients with spinal metastases. As with any patient, a complete history and examination is necessary, including a thorough neurological examination. Radiology should include an MRI to assess spinal cord compression and the extent of spinal metastases. Neurological status may necessitate urgent decompression with stabilisation of the adjacent vertebrae. However, prior to major surgery, it is important to liaise with the patients’ oncology service to ascertain overall outcome. In palliative cases, radiotherapy may be an option, if the patient is medically unfit to undergo and survive spinal surgery.
The indications to treat bone metastases with radiation therapy include pain, risk of pathological fracture and spinal cord compression. The goals of radiation therapy are to palliate pain, decrease the use of narcotic analgesics, improve ambulation and restore function and prevent complications of pathological fracture.
External-beam radiation is the most common treatment and remains the cornerstone of palliative treatment with hundreds of thousands of patients undergoing treatment each year in the United States. Radiotherapy for bone metastases attempts to exploit the radiosensitivity characteristics inherent to tumour cells such as significant vascularization, high rates of proliferation and non-differentiation[30]. The exact mechanism of action of radiation therapy is unknown and remains speculative[31]. It was only recently that animal models established that radiation had its effect on tumour cells and that the benefit did not accrue from an indirect effect on the peripheral surrounding normal cells[32,33].
Clinically, several choices exist regarding the use of radiation therapy for bone metastases. Opinions differ on the best regimen for each patient, the most suitable radiation dosage, the appropriate adjuvant therapies and their timings and the best delivery mechanism.
The radiation therapy oncology group (RTOG) conducted a prospective randomized trial (RTOG 74-02)[34]. Patients with a solitary metastasis were randomized to receive 2000 cGy using 4 Gy fractions delivered over a short 5-day period or 4050 cGy delivered using 2.7 Gy fractions over a 3 wk period. There was no significant difference in outcomes measured by pain relief. Similar results were seen in patients with multiple metastases who were randomized to receive 3000 cGy in 10 fractions over 2 wk or 1500 cGy in 5 fractions over 1 wk or 2000 cGy in 5 fractions over 1 wk or 2500 cGy in 5 fractions over 1 wk.
Since the RTOG trial[34] there have been several trials evaluating dose fractionation schemes[34,35] with no schedule or dose demonstrating significantly better outcome. Single-fraction radiotherapy has been advocated as a cost effective way to palliate bony metastases. A single dose of 8 Gy has been shown to have significantly better response rates when compared to a single dose of 4 Gy[36]. When a single dose regimen was compared to a multi-fraction regimen (20 Gy/5 fractions or 30 Gy/10 fractions), no differences were noted in time to symptomatic improvement, time to complete pain relief or time to first increase in pain up to 12 mo post-treatment[37].
It is now accepted that accelerated regimens may be appropriate in certain clinical settings for instance if the life expectancy is less than 3 years or where social circumstances decree that the patient cannot return on a daily basis. A protracted course may be more appropriate where the disease is more indolent or where the patient has a good performance status with a longer life expectancy or a solitary bone metastasis where the primary is well controlled[38].
The decision to use chemotherapy for the management of bone metastases relies on several factors. Firstly, the histology of the tumour must be known and secondly, it is important to know whether the patient has previously received chemotherapy because even the most chemosensitive tumours, such as lymphomas, are frequently resistant on relapse[38].
Certain tumours are considered highly chemosensitive. Such tumours frequently respond rapidly and often chemotherapy results in a significant reduction in the tumour burden. Complete or near complete remissions can be seen in certain chemosensitive tumours. Highly chemosensitive bone tumours may be considered for a trial of chemotherapy unless the involved bone is mechanically unstable.
Chemotherapy should rarely be considered for the management of metastatic tumours if the primary tumour is not chemoresponsive or chemosensitive. Response rates for these tumour types are so low that responses are considered anecdotal and it is reasonable to consider the tumour to have no effective chemotherapy.
Unfortunately the incidence of primary tumours is increasing, with that comes the challenge of dealing with metastatic disease. An individualised approach is recommended for each patient, taking into account the nature and biology of the primary lesion, life expectancy and the most appropriate surgical option. An increased understanding of the biology of the metastatic process may produce new treatment options to arrest this pathway at a variety of positions. Surgical management relies upon basic principles, but also on a fundamental knowledge of the nature of bone metastases.
P- Reviewers Brooks D, Iwamoto J, Nyland J S- Editor Huang XZ L- Editor A E- Editor Ma S
| 1. | American Cancer Society. Cancer Facts and Figures. Atlanta: American Cancer Society 2012; . [Cited in This Article: ] |
| 2. | Buckwalter JA, Brandser EA. Metastatic disease of the skeleton. Am Fam Physician. 1997;55:1761-1768. [PubMed] [Cited in This Article: ] |
| 3. | Hauschka PV, Mavrakos AE, Iafrati MD, Doleman SE, Klagsbrun M. Growth factors in bone matrix. Isolation of multiple types by affinity chromatography on heparin-Sepharose. J Biol Chem. 1986;261:12665-12674. [PubMed] [Cited in This Article: ] |
| 4. | Kahn D, Weiner GJ, Ben-Haim S, Ponto LL, Madsen MT, Bushnell DL, Watkins GL, Argenyi EA, Hichwa RD. Positron emission tomographic measurement of bone marrow blood flow to the pelvis and lumbar vertebrae in young normal adults. Blood. 1994;83:958-963. [PubMed] [Cited in This Article: ] |
| 5. | van der Pluijm G, Sijmons B, Vloedgraven H, Deckers M, Papapoulos S, Löwik C. Monitoring metastatic behavior of human tumor cells in mice with species-specific polymerase chain reaction: elevated expression of angiogenesis and bone resorption stimulators by breast cancer in bone metastases. J Bone Miner Res. 2001;16:1077-1091. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 103] [Cited by in F6Publishing: 102] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 6. | Urteaga O, Pack GT. On the antiquity of melanoma. Cancer. 1966;19:607-610. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 7. | Ribatti D, Mangialardi G, Vacca A. Stephen Paget and the ‘seed and soil’ theory of metastatic dissemination. Clin Exp Med. 2006;6:145-149. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 121] [Cited by in F6Publishing: 121] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 8. | Ewing J. Neoplastic diseases: A Textbook on Tumours. Philadelphia and London: W. B. Saunders Co 1919; . [Cited in This Article: ] |
| 9. | Harvey HA, Cream Lv. Biology of bone metastases: causes and consequences. Clin Breast Cancer. 2007;7 Suppl 1:S7-S13. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 30] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 10. | Cook GJ, Fogelman I. The role of nuclear medicine in monitoring treatment in skeletal malignancy. Semin Nucl Med. 2001;31:206-211. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 54] [Cited by in F6Publishing: 58] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 11. | Bombardieri E, Gianni L. The choice of the correct imaging modality in breast cancer management. Eur J Nucl Med Mol Imaging. 2004;31 Suppl 1:S179-S186. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 12. | Coleman RE. Metastatic bone disease: clinical features, pathophysiology and treatment strategies. Cancer Treat Rev. 2001;27:165-176. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1313] [Cited by in F6Publishing: 1274] [Article Influence: 55.4] [Reference Citation Analysis (0)] |
| 13. | Liotta LA, Kohn E. Cancer invasion and metastases. JAMA. 1990;263:1123-1126. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 20] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 14. | Mundy GR. Mechanisms of bone metastasis. Cancer. 1997;80:1546-1556. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 15. | Mareel MM, Behrens J, Birchmeier W, De Bruyne GK, Vleminckx K, Hoogewijs A, Fiers WC, Van Roy FM. Down-regulation of E-cadherin expression in Madin Darby canine kidney (MDCK) cells inside tumors of nude mice. Int J Cancer. 1991;47:922-928. [PubMed] [Cited in This Article: ] |
| 16. | Orr FW, Wang HH, Lafrenie RM, Scherbarth S, Nance DM. Interactions between cancer cells and the endothelium in metastasis. J Pathol. 2000;190:310-329. [PubMed] [Cited in This Article: ] |
| 17. | Berry MG, Gui GP, Wells CA, Carpenter R. Integrin expression and survival in human breast cancer. Eur J Surg Oncol. 2004;30:484-489. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 24] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 18. | Billroth T. General Surgical Pathology and Therapeutics, in Fifty Lectures: A Textbook for Students and Physicians. New York: D. Appleton and Company 1871; 233-243. [Cited in This Article: ] |
| 19. | Freije JM, Lawrence JA, Hollingshead MG, De la Rosa A, Narayanan V, Grever M, Sausville EA, Paull K, Steeg PS. Identification of compounds with preferential inhibitory activity against low-Nm23-expressing human breast carcinoma and melanoma cell lines. Nat Med. 1997;3:395-401. [PubMed] [Cited in This Article: ] |
| 20. | Senaratne SG, Colston KW. Direct effects of bisphosphonates on breast cancer cells. Breast Cancer Res. 2002;4:18-23. [PubMed] [Cited in This Article: ] |
| 21. | Kohno N. Treatment of breast cancer with bone metastasis: bisphosphonate treatment - current and future. Int J Clin Oncol. 2008;13:18-23. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 30] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 22. | Nathan SS, Healey JH, Mellano D, Hoang B, Lewis I, Morris CD, Athanasian EA, Boland PJ. Survival in patients operated on for pathologic fracture: implications for end-of-life orthopedic care. J Clin Oncol. 2005;23:6072-6082. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 150] [Cited by in F6Publishing: 161] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 23. | Beals RK, Lawton GD, Snell WE. Prophylactic internal fixation of the femur in metastatic breast cancer. Cancer. 1971;28:1350-1354. [PubMed] [Cited in This Article: ] |
| 24. | Cohen DB, Riley LH. Management of metastatic carcinoma to the spine: Surgical treatment. Orthopaedic Knowledge Update: Musculoskeletal Tumours. 1st ed. Rosemont IL: American Academy of Orthopaedic Surgeons 2002; 331-348. [Cited in This Article: ] |
| 25. | Mirels H. Metastatic disease in long bones: A proposed scoring system for diagnosing impending pathologic fractures. 1989. Clin Orthop Relat Res. 2003;S4-13. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 108] [Cited by in F6Publishing: 114] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 26. | Capanna R, Campanacci DA. Indications for the surgical treatment of long bone metastases. Textbook of Bone Metastases. Chichester: Wiley and Sons 2005; 135-146. [DOI] [Cited in This Article: ] |
| 27. | Galasko CSB, Banks AJ: The stabilization of skeletal metastases. In: Hellman K, Eccles SA: Treatment of Metastasis: Problems and Prospects. London: Taylor and Francis 1985; 85-88. [Cited in This Article: ] |
| 28. | Marco RA, Sheth DS, Boland PJ, Wunder JS, Siegel JA, Healey JH. Functional and oncological outcome of acetabular reconstruction for the treatment of metastatic disease. J Bone Joint Surg Am. 2000;82:642-651. [PubMed] [Cited in This Article: ] |
| 29. | Gainor BJ, Buchert P. Fracture healing in metastatic bone disease. Clin Orthop Relat Res. 1983;297-302. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 87] [Cited by in F6Publishing: 88] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 30. | Böhm P, Huber J. The surgical treatment of bony metastases of the spine and limbs. J Bone Joint Surg Br. 2002;84:521-529. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 126] [Cited by in F6Publishing: 141] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 31. | Presad KN. Handbook of Radiology. 2nd ed. Boca Raton: CRC press 1995; . [Cited in This Article: ] |
| 32. | Hoskin PJ. Scientific and clinical aspects of radiotherapy in the relief of bone pain. Cancer Surv. 1988;7:69-86. [PubMed] [Cited in This Article: ] |
| 33. | Goblirsch M, Lynch C, Mathews W, Manivel JC, Mantyh PW, Clohisy DR. Radiation treatment decreases bone cancer pain through direct effect on tumor cells. Radiat Res. 2005;164:400-408. [PubMed] [Cited in This Article: ] |
| 34. | Tong D, Gillick L, Hendrickson FR. The palliation of symptomatic osseous metastases: final results of the Study by the Radiation Therapy Oncology Group. Cancer. 1982;50:893-899. [PubMed] [Cited in This Article: ] |
| 35. | Okawa T, Kita M, Goto M, Nishijima H, Miyaji N. Randomized prospective clinical study of small, large and twice-a-day fraction radiotherapy for painful bone metastases. Radiother Oncol. 1988;13:99-104. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 81] [Cited by in F6Publishing: 83] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 36. | Niewald M, Tkocz HJ, Abel U, Scheib T, Walter K, Nieder C, Schnabel K, Berberich W, Kubale R, Fuchs M. Rapid course radiation therapy vs. more standard treatment: a randomized trial for bone metastases. Int J Radiat Oncol Biol Phys. 1996;36:1085-1089. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 103] [Cited by in F6Publishing: 105] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 37. | Jeremic B, Shibamoto Y, Acimovic L, Milicic B, Milisavljevic S, Nikolic N, Aleksandrovic J, Igrutinovic I. A randomized trial of three single-dose radiation therapy regimens in the treatment of metastatic bone pain. Int J Radiat Oncol Biol Phys. 1998;42:161-167. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 144] [Cited by in F6Publishing: 145] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 38. | 8 Gy single fraction radiotherapy for the treatment of metastatic skeletal pain: randomised comparison with a multifraction schedule over 12 months of patient follow-up. Bone Pain Trial Working Party. Radiother Oncol. 1999;52:111-121. [PubMed] [Cited in This Article: ] |