Published online Jul 28, 2021. doi: 10.4329/wjr.v13.i7.223
Peer-review started: March 15, 2021
First decision: April 6, 2021
Revised: April 10, 2021
Accepted: June 18, 2021
Article in press: June 18, 2021
Published online: July 28, 2021
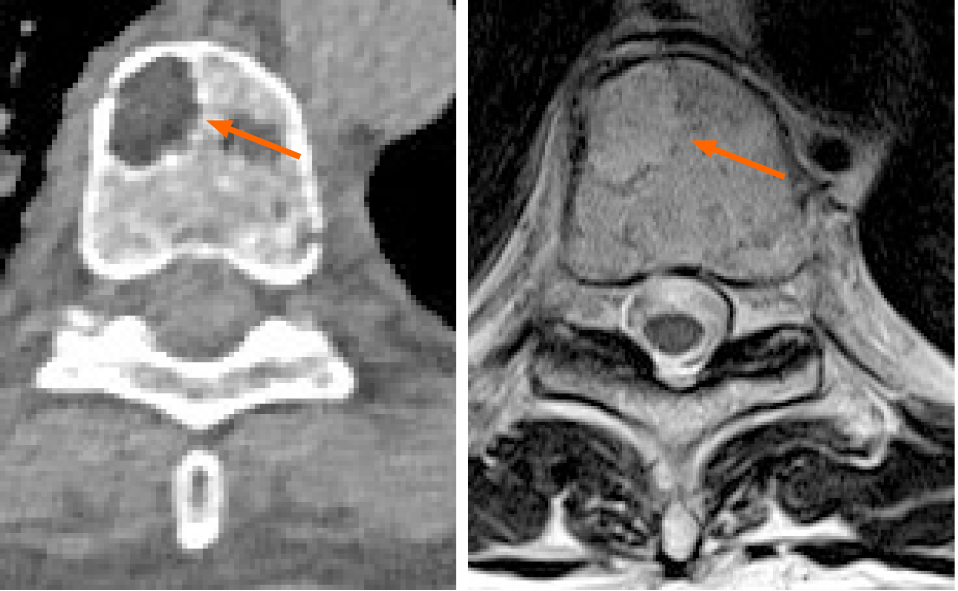
Multiple myeloma (MM) is the second most common type of hematological disease with its incidence rising in the elderly. In MM, the extent of the bone disease increases both morbidity and mortality. The detection of lytic bone lesions on imaging, especially computerized tomography (CT) and magnetic resonance imaging (MRI) is crucial to separate asymptomatic from symptomatic MM patients even when no clinical symptoms are present. Although radiology is essential in the staging and management of patients with MM there is still high variability in the choice between MRI and CT. In addition, there is still suboptimal agreement among readers. The potential of medical imaging in MM is largely under-evaluated: artificial intelligence, radiomics and new quantitative methods to report CT and MRI will improve imaging usage.
Core Tip: Introduction of new quantitative scores and biomarkers to predict multiple myeloma (MM) prognosis, possibly outperforming current staging methods to create new reliable standards for disease prediction and monitoring is an opportunity for further research in MM imaging.
- Citation: Tagliafico AS. Imaging in multiple myeloma: Computed tomography or magnetic resonance imaging? World J Radiol 2021; 13(7): 223-226
- URL: https://www.wjgnet.com/1949-8470/full/v13/i7/223.htm
- DOI: https://dx.doi.org/10.4329/wjr.v13.i7.223
Multiple myeloma belongs to the so-called plasma cell dyscrasias which are patholo
Firstly, it must be underlined that the detection of lytic bone lesions with a diameter > 5 mm can be done with both CT and MRI and no study directly compared the two modalities regarding patients’ outcomes after CT or MRI. At least in theory, MRI could have some advantages, such as the possibility to introduce functional sequences such as diffusion weighted sequences, but, no clear advantage of one technique over another has been found, even when a systematic review approach was adopted[11,12]. Regelink et al[12] found that there was only few additional lesions detected by both PET and MRI if CT was used as reference test (detection rate 1.00 and 1.00-1.25 respectively). In addition, the review by Regelink et al[12] review was limited by the suboptimal methodological quality of the involved studies due to lack of a technical details. It could be suggested that both MRI and CT have equal diagnostic value and there is no clear advantage to prefer one of the two techniques (Table 1). The scientific community is waiting for thorough comparative future studies, possibly focusing on prognostic value and follow-up. Furthermore, an analysis of multiple bone lesions detected on CT and MRI could be performed using artificial intelligence and radiomics[13]. Up-to-date, radiomics[14] is a quantitative radiological promising technique, with the ultimate goal to improve cancer treatment by improving prognostic capabilities of medical imaging. Radiomics is a complex, quantitative feature-based tool for image analysis described as the conversion of images to higher dimensional data and the subsequent mining of these data for improved decision support[14]. In MM, a recent application of radiomics improved the radiological evaluation of focal and diffuse pattern on CT by increasing the area under the curve of radiologists[15]. Accuracy of radiologists compared to the reference standard was lower (64%) than the accuracy using a radiomics approach (79%)[15]. In addition, machine learning-based classifiers resulted a satisfactory in differentiating MM lesions from those of tumor metastasis of the spine evaluated on MRI[16]. Radiomics was also on PET/CT in MM to elaborate a prognosis model predicting outcome in transplant-eligible newly diagnosed patients[17]. Finally, radiomics has been used with MRI to correlate features with the clinical and hematological response in multiple myeloma patients undergoing systemic treatment. In detail, one textural feature (GLSZM large area low gray level emphasis), in the study by Ekert et al[18] resulted to be correlated also with the bioptic degree of bone marrow infiltration.
| Availability | Reader expertise | Radiation dose | Repeatability among different readers | Repeatability among different scanners | Availability of reporting guidelines | Ability to detect > 5 mm focal lesions | Exam duration | |
| CT | High | Medium | Similar to total body CT | High | Medium | Low | High | Less than 10 min |
| MRI | Medium | Low | None | Medium | Medium | Low | High | More than 30 min |
Introduction of new quantitative scores and biomarkers to refine diagnosis, to predict MM prognosis, possibly outperforming current staging methods to create new reliable standards for disease prediction and monitoring is an opportunity for further research in MM imaging.
Manuscript source: Invited manuscript
Specialty type: Radiology, nuclear medicine and medical imaging
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C, C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Karavaş E, Xiao B S-Editor: Zhang H L-Editor: Filipodia P-Editor: Yuan YY
| 1. | Jameson J, Fauci A, Kasper D, Hauser S, Longo D. Harrison’s Principles of Internal Medicine. 20th ed. McGraw Hill, 2018. [Cited in This Article: ] |
| 2. | Rasche L, Chavan SS, Stephens OW, Patel PH, Tytarenko R, Ashby C, Bauer M, Stein C, Deshpande S, Wardell C, Buzder T, Molnar G, Zangari M, van Rhee F, Thanendrarajan S, Schinke C, Epstein J, Davies FE, Walker BA, Meissner T, Barlogie B, Morgan GJ, Weinhold N. Spatial genomic heterogeneity in multiple myeloma revealed by multi-region sequencing. Nat Commun. 2017;8:268. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 192] [Cited by in F6Publishing: 242] [Article Influence: 34.6] [Reference Citation Analysis (0)] |
| 3. | Neben K, Jauch A, Hielscher T, Hillengass J, Lehners N, Seckinger A, Granzow M, Raab MS, Ho AD, Goldschmidt H, Hose D. Progression in smoldering myeloma is independently determined by the chromosomal abnormalities del(17p), t(4;14), gain 1q, hyperdiploidy, and tumor load. J Clin Oncol. 2013;31:4325-4332. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 157] [Cited by in F6Publishing: 161] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 4. | Fonseca R, Bergsagel PL, Drach J, Shaughnessy J, Gutierrez N, Stewart AK, Morgan G, Van Ness B, Chesi M, Minvielle S, Neri A, Barlogie B, Kuehl WM, Liebisch P, Davies F, Chen-Kiang S, Durie BG, Carrasco R, Sezer O, Reiman T, Pilarski L, Avet-Loiseau H; International Myeloma Working Group. International Myeloma Working Group molecular classification of multiple myeloma: spotlight review. Leukemia. 2009;23:2210-2221. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 614] [Cited by in F6Publishing: 624] [Article Influence: 41.6] [Reference Citation Analysis (0)] |
| 5. | Kyle RA, Remstein ED, Therneau TM, Dispenzieri A, Kurtin PJ, Hodnefield JM, Larson DR, Plevak MF, Jelinek DF, Fonseca R, Melton LJ 3rd, Rajkumar SV. Clinical course and prognosis of smoldering (asymptomatic) multiple myeloma. N Engl J Med. 2007;356:2582-2590. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 609] [Cited by in F6Publishing: 574] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 6. | Rajkumar SV, Dimopoulos MA, Palumbo A, Blade J, Merlini G, Mateos MV, Kumar S, Hillengass J, Kastritis E, Richardson P, Landgren O, Paiva B, Dispenzieri A, Weiss B, LeLeu X, Zweegman S, Lonial S, Rosinol L, Zamagni E, Jagannath S, Sezer O, Kristinsson SY, Caers J, Usmani SZ, Lahuerta JJ, Johnsen HE, Beksac M, Cavo M, Goldschmidt H, Terpos E, Kyle RA, Anderson KC, Durie BG, Miguel JF. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014;15:e538-548. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2320] [Cited by in F6Publishing: 2676] [Article Influence: 267.6] [Reference Citation Analysis (0)] |
| 7. | Rajkumar SV. Evolving diagnostic criteria for multiple myeloma. Hematology Am Soc Hematol Educ Program. 2015;2015:272-278. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 42] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 8. | Hillengass J. Evolving Concepts in the Diagnosis and Staging of Multiple Myeloma. Natl Compr Cancer Netw. 2020;18:1770-1772. [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 9. | Tagliafico AS, Belgioia L, Bonsignore A, Rossi F, Succio G, Bignotti B, Dominietto A. Subspecialty Second-Opinion in Multiple Myeloma CT: Emphasis on Clinically Significant Lytic Lesions. Medicina (Kaunas). 2020;56. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 4] [Cited by in F6Publishing: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | Tagliafico AS, Dominietto A, Belgioia L, Campi C, Schenone D, Piana M. Quantitative Imaging and Radiomics in Multiple Myeloma: A Potential Opportunity? Medicina (Kaunas). 2021;57. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Caers J, Withofs N, Hillengass J, Simoni P, Zamagni E, Hustinx R, Beguin Y. The role of positron emission tomography-computed tomography and magnetic resonance imaging in diagnosis and follow up of multiple myeloma. Haematologica. 2014;99:629-637. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 46] [Cited by in F6Publishing: 51] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 12. | Regelink JC, Minnema MC, Terpos E, Kamphuis MH, Raijmakers PG, Pieters-van den Bos IC, Heggelman BG, Nievelstein RJ, Otten RH, van Lammeren-Venema D, Zijlstra JM, Arens AI, de Rooy JW, Hoekstra OS, Raymakers R, Sonneveld P, Ostelo RW, Zweegman S. Comparison of modern and conventional imaging techniques in establishing multiple myeloma-related bone disease: a systematic review. Br J Haematol. 2013;162:50-61. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 141] [Cited by in F6Publishing: 133] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 13. | Fiz F, Marini C, Campi C, Massone AM, Podestà M, Bottoni G, Piva R, Bongioanni F, Bacigalupo A, Piana M, Sambuceti G, Frassoni F. Allogeneic cell transplant expands bone marrow distribution by colonizing previously abandoned areas: an FDG PET/CT analysis. Blood. 2015;125:4095-4102. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 14. | Gillies RJ, Kinahan PE, Hricak H. Radiomics: Images Are More than Pictures, They Are Data. Radiology. 2016;278:563-577. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 4541] [Cited by in F6Publishing: 4471] [Article Influence: 558.9] [Reference Citation Analysis (2)] |
| 15. | Tagliafico AS, Cea M, Rossi F, Valdora F, Bignotti B, Succio G, Gualco S, Conte A, Dominietto A. Differentiating diffuse from focal pattern on Computed Tomography in multiple myeloma: Added value of a Radiomics approach. Eur J Radiol. 2019;121:108739. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 16. | Xiong X, Wang J, Hu S, Dai Y, Zhang Y, Hu C. Differentiating Between Multiple Myeloma and Metastasis Subtypes of Lumbar Vertebra Lesions Using Machine Learning-Based Radiomics. Front Oncol. 2021;11:601699. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 17. | Jamet B, Morvan L, Nanni C, Michaud AV, Bailly C, Chauvie S, Moreau P, Touzeau C, Zamagni E, Bodet-Milin C, Kraeber-Bodéré F, Mateus D, Carlier T. Random survival forest to predict transplant-eligible newly diagnosed multiple myeloma outcome including FDG-PET radiomics: a combined analysis of two independent prospective European trials. Eur J Nucl Med Mol Imaging. 2021;48:1005-1015. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 18. | Ekert K, Hinterleitner C, Baumgartner K, Fritz J, Horger M. Extended Texture Analysis of Non-Enhanced Whole-Body MRI Image Data for Response Assessment in Multiple Myeloma Patients Undergoing Systemic Therapy. Cancers (Basel). 2020;12. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 34] [Article Influence: 8.5] [Reference Citation Analysis (0)] |