Published online Apr 10, 2016. doi: 10.4239/wjd.v7.i7.153
Peer-review started: August 30, 2015
First decision: September 28, 2015
Revised: November 2, 2015
Accepted: February 14, 2016
Article in press: February 16, 2016
Published online: April 10, 2016
The burden of diabetic foot disease (DFD) is expected to increase in the future. The incidence of DFD is still rising due to the high prevalence of DFD predisposing factors. DFD is multifactorial in nature; however most of the diabetic foot amputations are preceded by foot ulceration. Diabetic peripheral neuropathy (DPN) is a major risk factor for foot ulceration. DPN leads to loss of protective sensation resulting in continuous unconscious traumas. Patient education and detection of high risk foot are essential for the prevention of foot ulceration and amputation. Proper assessment of the diabetic foot ulceration and appropriate management ensure better prognosis. Management is based on revascularization procedures, wound debridement, treatment of infection and ulcer offloading. Management and type of dressing applied are tailored according to the type of wound and the foot condition. The scope of this review paper is to describe the diabetic foot syndrome starting from the evaluation of the foot at risk for ulceration, up to the new treatment modalities.
Core tip: Foot at risk evaluation is crucial to diabetic foot ulceration prevention. Diabetic foot ulcer treatment includes standard wound care procedures, as well as, other novel treatment modalities always as add on therapy.
- Citation: Amin N, Doupis J. Diabetic foot disease: From the evaluation of the “foot at risk” to the novel diabetic ulcer treatment modalities. World J Diabetes 2016; 7(7): 153-164
- URL: https://www.wjgnet.com/1948-9358/full/v7/i7/153.htm
- DOI: https://dx.doi.org/10.4239/wjd.v7.i7.153
The diabetic foot syndrome or disease (DFD) includes several pathologies, mainly diabetic peripheral neuropathy and peripheral arterial disease which result in foot ulceration. Diabetic foot ulceration may ultimately lead to amputation, especially when wound infection or osteomyelitis are involved. Diabetic foot ulcer is defined as a full-thickness wound which is present at a level distal to the ankle in patients with diabetes[1,2]. Special categories like Charcot neuroarthropathy are also included in the DFD[3]. Patients with diabetic foot are also more likely to present with other diabetes-related complications such as nephropathy, retinopathy, ischemic heart disease and cerebrovascular disease[4].
DFD is a common complication which is multifactorial in nature. A good understanding of its various predisposing risk factors would help in both prevention and treatment of this devastating medical condition. The present review paper attempts to address the major challenges and barriers for a better approach of the DFD.
DFD occurs in all types of diabetes showing higher prevalence among males and in patients more than 60 years old[3]. The burden of DFD is expected to rise in the future, giving that the prevalence of its predisposing factors - mainly the diabetic peripheral neuropathy and peripheral limb ischemia are continually increasing[5]. Epidemiological studies for the DFD incidence and prevalence, present various conduction difficulties mostly related to the diagnostic tests used and the population selection[6].
The annual incidence of foot ulceration is estimated to be approximately 1%-4%[7,8], and its prevalence ranges from 4% to 10%, whereas, the lifetime risk for the development of a diabetic foot ulcer in patients with diabetes ranges from 15% to as high as 25%[7,9].
The presence of foot ulceration is considered to be the main precursor of a lower extremity amputation among patients with diabetes[10]. Apart from the diabetic peripheral neuropathy and the peripheral vascular disease, several other risk factors were identified such as, the limited joint mobility, the foot deformities and any previous ulceration or amputation at the same or contralateral limb. Other risk factors are related to the patient’s general condition including; impaired visual acuity, older age, chronic renal disease, long duration of diabetes & sustained uncontrolled hyperglycemia[11-14].
According to the International Consensus Group on Neuropathy; the diabetic neuropathy is defined as the detection of manifestations of peripheral nerve dysfunction in people with diabetes, after excluding other possible causes of peripheral neuropathy[15]. It is not an uncommon condition; in fact it is one of the most common long term complications of diabetes and the most common form of neuropathy in many parts of the world.
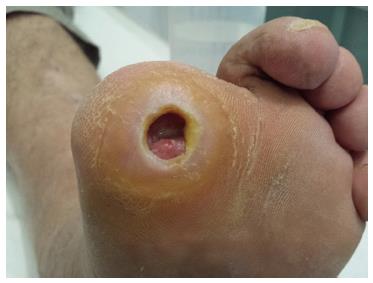
The presence of diabetic peripheral neuropathy, even with trivial trauma, is the initiating factor of the development of foot ulceration in patients with diabetes. It has been reported that the risk for diabetic foot ulceration increases by seven fold in patients with peripheral diabetic neuropathy[16,17]. It is also estimated that 45% to 60% of all ulcerations in patients with diabetes are mainly due to neuropathy, while 45% of the ulcers are due to combined neuropathic and ischemic factors (Figure 1)[12,16,18,19].
This form of neuropathy is the commonest presentation among patients with diabetes. It usually starts in the lower limbs while the upper limbs may be also involved too in some cases. It has a progressive course, starting distally and then proceeds proximally as the severity of nerve dysfunction increases. It usually presents in a glove and stocking pattern of abnormal sensations[20].
The distal symmetrical diabetic neuropathy may present with different clinical symptoms. Patients may describe it as symptoms of unpleasant sensations such as tingling, burning, prickling, electric shocks, lancinating pain, hyperalgesia (exaggerated perception of pain on application of a painful stimulus) or even allodynia (contact pain or pain perception due to a non-painful stimulus). Some patients may report abnormal cold or hot feelings in their feet or persistent painful cramp-like sensations even at rest[21]. It is worth mentioning that most of the patients may be completely asymptomatic and unaware of having peripheral neuropathy. Patients may present with diabetic foot ulceration even without any preceding neuropathic complaints[12].
Although the sensory nerve fibers are the most commonly affected fibers, motor nerve fibers are sometimes affected too, leading to muscle denervation. During the early course of the disease, the muscle power is preserved except mild muscle weakness in the toe extensors. As the disease progresses muscle weakness becomes more generalized affecting small muscles in both feet and hands. This muscle wasting can result in altering the normal foot dynamics and pressure distribution. Wasting and atrophy of small muscles in the foot lead to loss of joint stability and the development of foot deformities. Foot deformities may take several forms such as equinus or varus deformity, hammer toes, cocked-up toes and flat foot changes. These changes lead to pressure distribution disturbance, increased shear stress and friction, ultimately leading in foot ulceration[22-25]. Diabetic peripheral neuropathy is also characterized by the loss of the deep sensation, such as vibration perception and proprioception which in severe cases might lead to sensory ataxia and a positive Romberg’s sign. Deep tendon reflexes are usually impaired or lost starting with ankle reflex and progressing proximally to the knee reflex[21].
Additionally diabetic autonomic neuropathy may result in sudomotor dysfunction leading to abnormal sweating and dry skin with cracking and fissuring facilitating the bacterial infection of the foot[26]. Autonomic neuropathy is also associated with thermoregulatory dysfunction and abnormal tissue perfusion. Autonomic neuropathy is also in many cases associated with an unexplained foot edema which is resistant to diuretics. This edema results from shunt opening and hyperkinetic circulation, further adding to the risk of foot ulceration[27].
Diabetic peripheral neuropathy is diagnosed through careful patient history review and physical examination of the feet. Using the combination of patient’s neuropathic symptoms, clinical signs and electrodiagnostic tests would be the best predictor for diabetic peripheral neuropathy[28].
Symptom scores: Various verbal descriptive scales and simple visual analog scales are used in clinical practice to assess and follow up the neuropathic symptoms in response to treatment[29,30]. Symptom scores are used for the evaluation of painful diabetic neuropathy. The most widely used ones are: The Neuropathy Symptom Score (NSS), which is widely used in clinical practice has shown high validity and sensitivity[31-34]. Several other adaptations are also available such as the Neuropathy Symptom Profile, the modified NSS scores of Veves and Young, the Michigan Neuropathy Screening Instrument, and Diabetic NSS[17,35-38].
Semmes-Weinstein monofilament: Semmes-Weinstein monofilament is a widely used tool for the assessment of the diabetic peripheral neuropath in every day clinical practice[39,40]. It assesses the protective ability (evaluates A-beta fibers, determining the patient’s threshold for light touch and pressure) of the foot through the application of gentle pressure to the handle until the nylon filament is buckled for 2 s. Many different sizes of filaments are available with the 10-g pressure monofilament (5.07 monofilament) to be the most commonly used for pressure sensation evaluation. Around 90% of the patients with insensate diabetic foot could be identified on testing four planter sites (great toe and the base of first, third and fifth metatarsals)[41]. Monofilament test has shown a sensitivity of (66%-91%) in detection of diabetic patients at high risk for foot ulceration in several studies[18,42,43]. The monofilament test is a quick and painless method, easily acceptable from the patient, easy to administer by the physicians, portable and inexpensive[40].
Vibration perception: The impairment of vibration perception is usually one of the earliest signs of peripheral diabetic neuropathy. Vibration assessment evaluates the large diameter fibers (A-beta fibers). There are several ways for examining the vibration perception threshold (VPT), including: (1) 128 Hz tuning fork: It assesses the vibration perception through application on distal bony prominences of the great toe bilaterally and proceeds proximally on other bony prominences such as the medial malleolus and tibial tuberosity if impairment is noted. Tuning fork gives around 53% sensitivity and there is evidence suggesting that compared to the monofilament test, tuning fork is less predictive for development of foot ulceration[18]; (2) graduated rydel-seiffer tuning fork: The graduated tuning fork depends on optical visual illusion. The fork has 0-8 graded scale, where the examiner can detect the point of vibration impairment or disappearance[44]. Application of the graduated tuning fork detects the presence of vibration perception impairment and the intensity of this impairment. The reduction of vibration perception to less than 4/8 was present in 95% of diabetic foot ulcerations due to peripheral neuropathy[44,45]; (3) neurothesiometer; and (4) biothesiometer.
The neurothesiometer and biothesiometer are electronic devices. They depend on sending vibrations of various strengths through a probe applied to the bony prominence of the great toe. The vibrations are measured in volts per micrometer. As the VPT of the patient increases, the risk for diabetic foot ulceration due to neuropathy increases. A vibration threshold of more than 25 V has been reported to have a sensitivity of 83%. The risk of ulceration in the group of patients with sensitivity between 25 and 33 V was increased by eight times compared to twenty-fold increase associated with values of more than 42 V[46,47].
Nerve conduction studies: Nerve electrophysiologic conduction studies are not routinely used in clinical practice for diagnosis of diabetic neuropathy. They are objective, non-invasive, highly reliable parametric measures which are useful in monitoring the progression of diabetic peripheral neuropathy especially in asymptomatic patients[48]. They are also extremely useful in atypical presentations of neuropathy and in superimposed forms of mononeuropathies[49]. Electromyography and nerve conduction studies such as NCV and F waves can detect the type of nerve injury, extend, symmetry and severity of the lesion[50].
Other methods of assessment: (1) Nerve biopsy: It is an invasive procedure used for diagnosis of peripheral neuropathy and atypical presentations in patients with diabetes, usually using sural nerve biopsy[51]. Nerve biopsy has many complications such as postoperative pain at the site of nerve biopsy, parasthesia, allodynia and sensory disturbances at the sites of nerve distribution especially in patients with diabetes[52]. Assessment of efficacy of treatment and disease progression can be determined depending on morphological parameters such as axonal atrophy, density of myelinated fibres and axo-glial dysjunction[53,54]; (2) Skin biopsy: It is another less invasive technique alternative to nerve biopsy for studying small nerve fibers using a 3-mm skin biopsy in clinical studies[55]. Several neuronal markers are used to immunostain skin nerves, such asneurone-specific enolase and somatostatin. The best cytoplasmic axonal marker has been proposed is the protein gene product-9.5. Formalin-fixed frozen sections are used in clinical research to visualize and assess the density of intraepidermal nerve fibers[56,57].
Validated scores are available in order to standardize the clinical assessment for the severity of symptoms and the grade of neuropathic impairment. The validated scores include; Neuropathy Disability Score for neuropathic deficits (impairments), NSS for neuropathic symptoms[17] and the Michigan Neuropathy Screening Instrument[36]. According to the Neuropathy Disability Score and the NSS, minimum criteria required for the clinical diagnosis of neuropathy are: (1) the presence of moderate signs of neuropathy in the presence or absence of symptoms; (2) the detection of mild signs in the presence of moderate symptoms.
Peripheral arterial disease (PAD) is one of the multifactorial causes leading to the diabetic foot disease. The presence of PAD alters the normal body response to foot ulcerations and leads to persistent non-healing foot ulcers, when there is an increased need for blood supply. PAD leads to progression of infection, increases tissue break down and insufficient delivery of oxygen, nutrition and antibiotics. All these factors further contribute to a potential foot amputation[58].
PAD shows higher prevalence among patients with diabetes than the general population. PAD among patients with diabetes is characterized by onset at an earlier age, increased severity, a more rapid progression and equal sex distribution[59]. About 20% of patients having symptomatic PAD had diabetes as reported by the Framingham Heart Study[60]. In patients with diabetes, the risk of PAD is increased by advanced age, duration of diabetes, uncontrolled hyperglycemia and the association with diabetic peripheral neuropathy. The presence of diabetes is mostly associated with below knee PAD such as tibial, popliteal and femoral arterial affection, in contrast to more proximal PAD in the aorto-iliofemoral vessels associated with other risk factors such as hypertension and smoking[61].
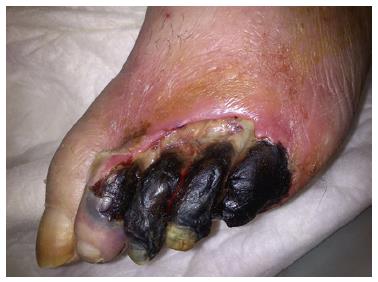
PAD is characterized by the presence of intermittent claudication, which is defined as cramping or aching pains usually in the calf muscles, but can also be present in thighs or the buttocks. Intermittent claudication is aggravated by walking exercise that it forces the patient to stop walking and relieved by rest. In severe cases of PAD, pain may be present even at rest, limb may show gangrenous changes, tissue loss; which is known as critical limb ischemia (Figure 2)[62].
The ankle-brachial index: The ankle-brachial index (ABI) is a simple bed-side screening tool for the presence of PAD. PAD simply depends on the calculation of the ratio between the systolic pressure of the ankle arteries and the systolic pressure at the brachial arteries[63]. ABI is an inexpensive method that can assess the severity of PAD as it usually correlates with the patient’s reported symptoms and functional status. The normal range is of ABI is between 0.9-1.3, falsely elevated values of ABI can result in cases of calcified, non-compressible arteries. Thus the ABI method may lead to underestimation of the severity of the disease in patients with diabetes[64].
The toe-brachial index: The toe-brachial index is calculated similar to the ABI, where the systolic pressure is measured using a small cuff and a Doppler probe. Measuring the toe-brachial index is helpful especially in cases of ABI values more than 1.30, as the small arteries of the lower limb are less likely to be calcified. A toe-brachial index lower than 0.70 is diagnostic for PAD[65].
Segmental limb pressure assessment and pulse volume recordings: The technique depends on plethysmographic cuffs situated over the brachial arteries and different points on the lower limb. The extent and location of PAD can be detected from segmental systolic pressure assessment using a Doppler probe[66].
Ultrasound velocity spectroscopy and imaging: The normal arterial Doppler velocity shows a triphasic signal. When an arterial obstruction is present proximal to the probe, there is loss of the normal reversed flow component on transforming the waveform associated with decreased amplitude, attenuation of all parts of the spectrum and delayed upstroke[67].
Duplex ultrasound depends on combining the B-mode and the pulsed Doppler ultrasound to assess arterial flow and localized velocity information at stenotic sites. Duplex ultrasonography is widely used nowadays detecting with high sensitivity and specificity the arterial patency and extends of obstruction[68]. Duplex ultrasound has certain limitations mainly difficulty in identifying close multiple separate lesions, some difficulty when assessing infrapopliteal, common and external iliac arteries[69,70].
Transcutaneous oximetry and laser - doppler flowmetry: These techniques are used mainly to assess cutaneous blood flow. Cutaneous blood flow is usually normal until late stages of proximal arterial ischemia of the atherosclerotic type, thus, this type of vascular evaluation is not used in every day practice[64].
Magnetic resonance angiography (MRA)[71,72].
Computed tomographic angiography: CTA is superior to MRA as it can detect the presence of calcification, which is advantageous on planning revascularization strategies. The ACC/AHA guidelines recommend CTA on deciding the revascularization techniques in cases of PAD, offering faster image than MRA[63].
Contrast angiography: Although it is the gold standard for the diagnosis of PAD, is rarely required as a diagnostic tool due to the risks associated with invasive procedures. Computer-enhanced digital subtraction angiography can be useful in patients who present with localized stenosis so as to minimize the amount of contrast material injected and for better image resolution[63].
Diabetic foot ulcers.
The presence of diabetic foot ulceration is the main leading risk for amputation in patients with diabetes. Proper assessment and classification of a diabetic foot ulcer is an essential part for the management of the diabetic foot. A prompt and adequate ulcer treatment may lead to foot amputation prevention, preserving the life quality of the patient.
Several classifications have been proposed for the categorization of diabetic foot ulcers. The most important ones are described below.
It is one of the earliest and most widely used classifications. It classifies the diabetic foot ulceration depending on how deep the wound is, includes 6 grades: (1) Grade 0: The skin is intact; (2) Grade 1: Presence of ulcer which is superficial; (3) Grade 2: Presence of ulcer which is deep; (4) Grade 3: Deep ulcer with abscess, bone involvement or osteomyelitis; (5) Grade 4: Gangrene in the forefoot; and (6) Grade 5: Whole foot gangrene.
Wagner-Meggitt classification has shown several disadvantages: (1) cannot address all patterns of diabetic foot ulcerations and infections; (2) the presence of infection is addressed in only one stage, thus, the superficial ulcers if infected or ischemic are not properly categorized in this system; and (3) this system does not properly assess the presence of peripheral ischemia in categorization of foot ulcers.
It classifies diabetic foot ulcers into 4 grades (0-4) according to their depth, and then stages every grade of them according to the presence or absence of infection and ischemia (A-D).
The University of Texas Classification has been validated and has prognostic advantages as it included both infection and ischemia but showed some difficulty in application in day to day practice.
This classification grades the diabetic foot ulceration according to five ulcer features (size, depth, sepsis, arteriopathy, and denervation) on a 4-point scale (0-3).
SAD classification differs from the other earlier systems by considering both size of ulcer and the presence of neuropathy. It has been validated by demonstrating differences between baselines variable and clinical outcome. Its major drawback is the complexity in practical use.
The PEDIS system has been proposed by the International Working Group on the Diabetic Foot. This system grades the wound based on five features: (1) perfusion (arterial blood supply); (2) extent (area of the ulcer); (3) depth of the wound; (4) presence of infection; and (5) sensation.
These guidelines sub classified the infected diabetic foot into three categories: (1) mild: Involvement is restricted to skin and subcutaneous tissues; (2) moderate: Involvement is more extensive or affecting deeper tissues; and (3) Severe: Diabetic foot ulceration is accompanied by systemic signs of infection or metabolic decompensation.
Debridement of diabetic foot ulcers is an important initial step in the management of the wound. Several benefits can result from proper debridement including the removal of the necrotic and non-viable tissues and keeping a healthy granular wound bed. One should be careful on the assessment of the ulcer if ischemia is suspected. A revascularization intervention may be necessary before a debridement is performed. Debridement is also stimulating the release of growth factors to promote advancing healing edges[77,78]. Various methods are used for wound debridement.
Surgical debridement: It is the gold standard method in diabetic foot ulceration. To obtain optimal results, healthy tissue loss should be minimized, foot function should be preserved, and deformities which can precipitate recurrence of ulcers should be prevented. Surgical debridement is typically done for ulcers with large amount of necrotic and non-viable tissues. Debridement is performed using a scalpel blade with the tip pointed in a 45-degree angle or a tissue nipper to remove all necrotic and non-viable tissues until a bleeding healthy base is obtained[79].
Enzymatic debridement: Enzymatic debridement is based on the application of topical agents on the ulcer. These agents are usually applied once daily. Their action is based on the necrotic tissue degradation using proteolytic digestive enzymes such as streptokinases, trypsin, papain, fibrinolysin-DNase, collagenase, papain-urea and streptodornase. Data from clinical studies have shown conflicting results about the efficacy of these topical agents, thus, their additional benefits to standard wound care remains unclear. Putting into consideration the need of long time application, as well as, the high cost, their use is usually limited to slowly soften large eschars or debridement of some decubitus ulcerations in sensate limbs. In order to improve efficacy of these agents, a scalpel blade is applied to crosshatch eschars[80].
Mechanical debridement: Although it is a simple and an inexpensive tool, it can remove both viable and also non-viable tissues leading to pain in sensate foot. The wet gauze dressing is applied to the wound bed and then kept to dry. The necrotic debris embedded in the gauze is mechanically stripped from the wound bed on gauze removal[81].
Biological debridement (Maggot therapy): Recently the use of Maggot therapy has re-emerged showing benefits in necrotic tissue debridement, decreasing bacterial load and stimulation of wound healing. Several studies showed the efficacy of Maggot therapy[82-84].
Other modalities: Ongoing research is evaluating other methods such as low energy ultrasound mist for the debridement of diabetic foot ulcers[84].
The pressure offloading relieves abnormal pressure applied to the ulcer promoting the wound healing. Several methods have been applied for offloading including; total contact casting (TCC), short leg walkers, half shoes and felted foam dressings. TCC is based on a well-molded plaster cast, resulting in equal pressure distribution to the whole lower limb. This method is very effective with a good wound healing rate when applied properly and changed at least weekly. Although it is an effective method it has significant disadvantages which may limit their use and the choice of other alternatives. Disadvantages of TCC include; time and skill required to be applied properly, secondary skin irritation and ulceration resulting from the cast applied, and impossible daily assessment of the wound[85,86].
Other alternative to TCC is the Scotch-cast boot with a cast sandal to increase mobility and at the same time ensure ulcer relief from pressure. Commercial devices such as the short leg walker and half shoe are readily available, they are preferred by the patients with a better acceptance, simple, easy to apply and inexpensive. Their major disadvantage is that patients can remove them thus cannot ensure patient’s compliance with less significant pressure relieving results compared to the TCC[87].
Felted foam dressings allow a customized pressure relief through a felt-foam pad with an opening over the ulceration through which wound assessment and care can be done. The felted foam is used in combination with half-shoe or surgical shoe and when used properly and changed every 10-14 d, has shown more efficacy compared to half-shoe or short walkers alone[88].
The presence of infection is a common finding in diabetic foot ulcers which act as an entry route for pathogens. Infections must be diagnosed and treated promptly and adequately as they may rapidly progress to a limb-threatening condition[78]. Also high levels of bacteria can delay or event prevent wound healing and impede surgical closure of diabetic ulcers[89].
Diagnosis of diabetic foot ulcer infection: Diagnosis of infection is based initially on clinical signs such as redness, temperature, pain, tenderness, edema and the presence of suspected discharge. On clinical suspicion of infection, properly taken cultures from the wound area may be helpful in proper antibiotic treatment selection. It is important to point out that uninfected ulcers is not necessary to be cultured as the results will only indicate the colonizing flora. The most common pathogens in diabetic foot ulcers are aerobic gram positive cocci and gram negative bacteria. Anaerobic organisms are frequently isolated too[78]. Staphylococcus and streptococci are the most frequently causative agents for non-threatening limb infections while limb-threatening infections are mostly polymicrobial in nature[90].
The use of antibiotics in infected diabetic foot ulcer should be carefully applied, in order to be assured that the patient will receive the appropriate antibiotic therapy, for an adequate period of time, along with wound debridement and drainage[90].
To be able to provide an effective plan for diabetic foot syndrome prevention and treatment, a multidisciplinary team approach is required.
This multidisciplinary team approach includes[10,91]: (1) diabetologist/Endocrinologist to optimize the metabolic control for patients with diabetes; (2) diabetes educator and a qualified nurse: To provide special education and assurance; (3) podiatrist who would guide the patient to prevent diabetic foot lesions and provide appropriate treatment; (4) vascular surgeon to assess the vascularity of the lower limbs and provide interventional management whenever required; (5) orthotist: Help in choosing the appropriate foot wear or custom foot wear to allow adequate pressure distribution and thus rapid wound healing; (6) infection disease specialist: For appropriate choice of antibiotics regimen based on culture results; and (7) nutritionist consultation to help in adequate glycemic control, weight loss and also wound healing.
In the recent years, apart from the standard wound care, new diabetic ulcer treatment modalities have been developed[92,93].
The ideal wound dressing[94]: (1) should be sterile and does not contaminate the ulcer with foreign particles; (2) should be readily available, easy to use and cost effective; (3) should keep a moist environment for adequate wound healing; (4) should be able to absorb excess exudates from the wound; (5) should not adhere to the wound floor, also should be non-allergic and non-toxic; (6) should be able to protect the wound from microorganisms and also provides mechanical protection; and (7) should maintain adequate tissue gaseous exchange and control wound odor.
Wet to dry dressing (simple saline dressing): Wet to dry dressing is included in standard wound care and is considered a method for mechanical debridement, since it presents a good debriding effect in removal of the necrotic tissue and wound preparation[95]. In order to minimize irritation and discomfort, adequate moistening of the dressings with normal saline is done when treating granulating wound tissues to avoid trauma and bleeding[96].
Local antibacterial agents: Antibacterial agents can be used alone or in combination with other dressings except for dry necrotic ulcers. For effective anaerobic coverage, metronidazole gel is used and maintains a moist environment for wound healing[97,98]. Several antibiotics have effective antibacterial action on topical application such as Neomycin, Gentamycin, and Mupirocin. Silver dressings and polyherbal topical preparations have shown good antibacterial action[99]. For effective antibacterial action against Pseudomonas, other gram-negative bacilli, and beta hemolytic streptococci wound infections Sisomycin and acetic acid can be used. Special precautions should be considered when using povidone iodine solution dressings, iodine has been found to be toxic at high concentrations to bacteria and fungi as well as human cells[100,101].
Tulle dressings: Tulle dressings are used mainly for skin grafts and superficial wounds. They can be safely used in granulating and epithelializing tissues as they are impregnated with paraffin, having low dressing adherence properties[102]. Evidence from several previous studies have shown better and faster re-epithelialization rates compared to dry dressing[102-104].
Hydrogel dressings: Hydrogel dressings are considered the best choice for dry wounds with necrotic eschar. Hydrogels provide fluid and good hydration to dry and slough wounds. Although they are very good at absorbing exudates, they should be avoided in diabetic foot planter ulcers as they may cause maceration of the skin surrounding the wound[105-107].
Polyurethane films: Polyurethane films are transparent films coated with a water-proof adhesive dressing. They provide permeable films allowing diffusion of gases and vapor thus an adequately moist wound environment. They have the advantage of being transparent, thus can monitor the wound progression. They also can be used for low exudates wounds, but they may cause maceration of the skin surrounding the wound[108].
Polyurethane foam: Polyurethane foam is highly used in diabetic foot ulcers. It can absorb large amounts of exudates in a non-adherent nature thus does not cause wound sloughing or trauma on removal. They maintain moisture environment thus allow proper preparation of wound bed and promotes better wound healing[109].
Alginate dressings: Two forms of alginate dressings are available; calcium alginate and calcium sodium alginate. Alginate dressings can absorb large amounts of exudates up to 20 times their weight as shown by several clinical studies[110].
Honey-impregnated dressings: The anti-inflammatory and anti-microbial actions have been shown in vitro studies but further studies are required to support strong evidence in vivo[111,112].
Growth factors have shown promising results in diabetic ulcer healing. Growth factors stimulate angiogenesis, cellular proliferation and migration, together with promoting enzymatic production. Several growth factors have been studied including; platelet-derived growth factor (PDGF), epidermal growth factor (EGF), transforming growth factor (TGF)-β, TGF-α and insulin-like growth factor…, etc.[113]. A recombinant human (rh)-PDGF dressing is available for diabetic foot lesions when added to conventional[114]. EGF in the form of local injections of rh-EGF showed favorable results in neuropathic vs ischemic ulceration[115].
The Food and Drug Administration (FDA) announced some warnings regarding the use of a recombinant human platelet-derived growth factor, which contains becaplermin. In both clinical studies and post marketing users, becaplermin was associated with malignancies distant from the application site. Also increased mortality rate from systemic malignancies was reported on using 3 or more tubes of becaplermin gel. Topical enzymes: Several prepared ointments containing enzymes such as fibrinolysin, collagenase or papain have been used in enzymatic debridement of the sloughy tissues and promoting granulation tissue formation. Papain-urea has shown better enzymatic debridement effect when compared to collagenase[116].
Vacuum-assisted closure: Vacuum-assisted devices have shown efficacy in exudates removal and edema reduction. Ideally a pressure of 125 mmHg can generate a negative topical pressure over the diabetic foot wound. It has the advantage of leaving the wound surface moist. It has several limitations; it is contraindicated in cases of osteomyelitis, ischemia, deep tissues exposure such as tendons, bones and blood vessels, presence of necrotic tissues and fistulas[117]. Vacuum-assisted devices are also effective in promoting closure and wound healing in patients with treated infections and treated osteomyelitis[118,119].
Hyperbaric oxygen therapy, Do we have evidence A systemic treatment where oxygen is breathed but at a higher pressure than the local atmospheric pressure[120]. HBOT has shown increased healing rates of diabetic foot ulcers, however it still controversial whether it can be used as adjuvant treatment or not[121]. Hyperbaric oxygen therapy (HBOT) have the advantage of reduction of tissue hypoxia, edema, increase angiogenesis and erythrocytes deformability, antimicrobial effects and increase fibroblastic activity[122-124]. HBOT is approved as an adjunctive treatment to be used in chronic non-healing ulcers by the Undersea and Hyperbaric Medical Society[125]. The European Committee for Hyperbaric Medicine has set a type 2 recommendation for the use of HBOT in the management of diabetic foot ulcers including patients with ischemic wounds without a surgically treatable arterial lesion or as a complement after vascular surgery, in presence of non-healing wounds[126].
The role of stem cell therapy in PAD: It is worth mentioning that our skeletal muscles have a regenerative capacity as the have multipotentaial and progenitor cells. In cases of critical limb peripheral arterial disease, the transplantation of progenitor cells- derived from bone marrow- has beneficial effects on angiogenesis and ulcer healing as shown in phase I and II studies. The role therapeutic angiogenesis is a promising and a safe method for management of PAD and limb salvage[127].
Concluding, diabetic foot ulceration is generally preventable. The first step in ulcer prevention is the careful screening for foot problems and detection of patients at high risk. More research is still required to improve the diagnosis of conditions leading to foot ulceration. Diversity in the diagnostic criteria and the lack of cut off hinders the standardization of management plans. Multi-disciplinary team approach is required to effectively manage the different aspects of diabetic foot syndrome. Standard wound care is recommended, while modern treatment modalities have shown some promising results in recent studies.
P- Reviewer: Tamemoto H, Zhao J S- Editor: Kong JX L- Editor: A E- Editor: Wu HL
| 1. | Reiber GE, Ledoux WR. Epidemiology of diabetic foot ulcers and amputations: Evidence for prevention. The evidence base for diabetes care. Chichester; Hoboken (NJ): John Wiley & Sons, Ltd 2003; 641-665. [DOI] [Cited in This Article: ] |
| 2. | Apelqvist J, Bakker K, van Houtum WH, Nabuurs-Franssen MH, Schaper NC. International consensus and practical guidelines on the management and the prevention of the diabetic foot. International Working Group on the Diabetic Foot. Diabetes Metab Res Rev. 2000;16 Suppl 1:S84-S92. [PubMed] [DOI] [Cited in This Article: ] |
| 3. | Rathur HM, Boulton AJ. The diabetic foot. Clin Dermatol. 2007;25:109-120. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 73] [Cited by in F6Publishing: 78] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 4. | Khanolkar MP, Bain SC, Stephens JW. The diabetic foot. QJM. 2008;101:685-695. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 95] [Cited by in F6Publishing: 91] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 5. | UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352:837-853. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14327] [Cited by in F6Publishing: 12343] [Article Influence: 474.7] [Reference Citation Analysis (0)] |
| 6. | Williams DR, Airey M. The size of the problem: Epidemiological and economical aspects of the diabetic foot. Chichester England; New York: Wiley 2000; 3-17. [Cited in This Article: ] |
| 7. | Bartus CL, Margolis DJ. Reducing the incidence of foot ulceration and amputation in diabetes. Curr Diab Rep. 2004;4:413-418. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 56] [Cited by in F6Publishing: 56] [Article Influence: 2.8] [Reference Citation Analysis (2)] |
| 8. | Armstrong DG, Lipsky BA. Advances in the treatment of diabetic foot infections. Diabetes Technol Ther. 2004;6:167-177. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 57] [Cited by in F6Publishing: 59] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 9. | Boulton AJ, Vileikyte L, Ragnarson-Tennvall G, Apelqvist J. The global burden of diabetic foot disease. Lancet. 2005;366:1719-1724. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1532] [Cited by in F6Publishing: 1427] [Article Influence: 75.1] [Reference Citation Analysis (0)] |
| 10. | American Diabetes Association. Consensus Development Conference on Diabetic Foot Wound Care: 7-8 April 1999, Boston, Massachusetts. American Diabetes Association. Diabetes Care. 1999;22:1354-1360. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 388] [Cited by in F6Publishing: 399] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 11. | Frykberg RG. Diabetic foot ulcers: pathogenesis and management. Am Fam Physician. 2002;66:1655-1662. [PubMed] [Cited in This Article: ] |
| 12. | Boulton AJ, Kirsner RS, Vileikyte L. Clinical practice. Neuropathic diabetic foot ulcers. N Engl J Med. 2004;351:48-55. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 394] [Cited by in F6Publishing: 397] [Article Influence: 19.9] [Reference Citation Analysis (1)] |
| 13. | Boulton AJ. The diabetic foot: from art to science. The 18th Camillo Golgi lecture. Diabetologia. 2004;47:1343-1353. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 216] [Cited by in F6Publishing: 224] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 14. | Frykberg RG, Zgonis T, Armstrong DG, Driver VR, Giurini JM, Kravitz SR, Landsman AS, Lavery LA, Moore JC, Schuberth JM. Diabetic foot disorders. A clinical practice guideline (2006 revision). J Foot Ankle Surg. 2006;45:S1-S66. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 510] [Cited by in F6Publishing: 438] [Article Influence: 24.3] [Reference Citation Analysis (1)] |
| 15. | Katsilambros N. Who is the patient at risk for foot ulceration. Chichester; Hoboken, NJ: Wiley 2003; 1-21. [DOI] [Cited in This Article: ] |
| 16. | Reiber GE, Vileikyte L, Boyko EJ, del Aguila M, Smith DG, Lavery LA, Boulton AJ. Causal pathways for incident lower-extremity ulcers in patients with diabetes from two settings. Diabetes Care. 1999;22:157-162. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 700] [Cited by in F6Publishing: 607] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 17. | Young MJ, Boulton AJ, MacLeod AF, Williams DR, Sonksen PH. A multicentre study of the prevalence of diabetic peripheral neuropathy in the United Kingdom hospital clinic population. Diabetologia. 1993;36:150-154. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 961] [Cited by in F6Publishing: 897] [Article Influence: 28.9] [Reference Citation Analysis (0)] |
| 18. | Boyko EJ, Ahroni JH, Stensel V, Forsberg RC, Davignon DR, Smith DG. A prospective study of risk factors for diabetic foot ulcer. The Seattle Diabetic Foot Study. Diabetes Care. 1999;22:1036-1042. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 430] [Cited by in F6Publishing: 389] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 19. | Abbott CA, Carrington AL, Ashe H, Bath S, Every LC, Griffiths J, Hann AW, Hussein A, Jackson N, Johnson KE. The North-West Diabetes Foot Care Study: incidence of, and risk factors for, new diabetic foot ulceration in a community-based patient cohort. Diabet Med. 2002;19:377-384. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 706] [Cited by in F6Publishing: 607] [Article Influence: 27.6] [Reference Citation Analysis (1)] |
| 20. | Tesfaye S. Diabetic neuropathy. Chichester, England; Hoboken, NJ: Wiley 2006; 30-40. [DOI] [Cited in This Article: ] |
| 21. | Tesfaye S. Clinical features of diabetic polyneuropathy. Totowa, NJ: Humana Press 2007; 243-257. [DOI] [Cited in This Article: ] |
| 22. | Hutton W, Stokes I. The mechanics of the foot. Oxford, England: Blackwell Scientific 1991; 11-25. [Cited in This Article: ] |
| 23. | Boulton A. Peripheral neuropathy and the diabetic foot. The Foot. 1992;2:67-72. [DOI] [Cited in This Article: ] |
| 24. | Frykberg RG, Lavery LA, Pham H, Harvey C, Harkless L, Veves A. Role of neuropathy and high foot pressures in diabetic foot ulceration. Diabetes Care. 1998;21:1714-1719. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 286] [Cited by in F6Publishing: 232] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 25. | Bus SA, Yang QX, Wang JH, Smith MB, Wunderlich R, Cavanagh PR. Intrinsic muscle atrophy and toe deformity in the diabetic neuropathic foot: a magnetic resonance imaging study. Diabetes Care. 2002;25:1444-1450. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 144] [Cited by in F6Publishing: 116] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 26. | Shaw JE, Boulton AJ. The pathogenesis of diabetic foot problems: an overview. Diabetes. 1997;46 Suppl 2:S58-S61. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 87] [Cited by in F6Publishing: 87] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 27. | Nabuurs-Franssen MH, Houben AJ, Tooke JE, Schaper NC. The effect of polyneuropathy on foot microcirculation in Type II diabetes. Diabetologia. 2002;45:1164-1171. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 34] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 28. | England JD, Gronseth GS, Franklin G, Miller RG, Asbury AK, Carter GT, Cohen JA, Fisher MA, Howard JF, Kinsella LJ. Distal symmetrical polyneuropathy: a definition for clinical research. A report of the American Academy of Neurology, the American Association of Electrodiagnostic Medicine, and the American Academy of Physical Medicine and Rehabilitation. Arch Phys Med Rehabil. 2005;86:167-174. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 28] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 29. | Didangelos T, Doupis J, Veves A. Painful diabetic neuropathy: clinical aspects. Handb Clin Neurol. 2014;126:53-61. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 30. | Ziegler D, Mayer P, Gries FA. Evaluation of thermal, pain, and vibration sensation thresholds in newly diagnosed type 1 diabetic patients. J Neurol Neurosurg Psychiatry. 1988;51:1420-1424. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 70] [Cited by in F6Publishing: 74] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 31. | Dyck PJ. Detection, characterization, and staging of polyneuropathy: assessed in diabetics. Muscle Nerve. 1988;11:21-32. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 362] [Cited by in F6Publishing: 351] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 32. | Dyck PJ, Sherman WR, Hallcher LM, Service FJ, O’Brien PC, Grina LA, Palumbo PJ, Swanson CJ. Human diabetic endoneurial sorbitol, fructose, and myo-inositol related to sural nerve morphometry. Ann Neurol. 1980;8:590-596. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 368] [Cited by in F6Publishing: 364] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 33. | Dyck PJ, Karnes JL, Daube J, O’Brien P, Service FJ. Clinical and neuropathological criteria for the diagnosis and staging of diabetic polyneuropathy. Brain. 1985;108:861-880. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 222] [Cited by in F6Publishing: 237] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 34. | Dyck PJ, Kratz KM, Lehman KA, Karnes JL, Melton LJ, O’Brien PC, Litchy WJ, Windebank AJ, Smith BE, Low PA. The Rochester Diabetic Neuropathy Study: design, criteria for types of neuropathy, selection bias, and reproducibility of neuropathic tests. Neurology. 1991;41:799-807. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 208] [Cited by in F6Publishing: 205] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 35. | Dyck PJ, Karnes J, O’Brien PC, Swanson CJ. Neuropathy Symptom Profile in health, motor neuron disease, diabetic neuropathy, and amyloidosis. Neurology. 1986;36:1300-1308. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 67] [Cited by in F6Publishing: 67] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 36. | Feldman EL, Stevens MJ, Thomas PK, Brown MB, Canal N, Greene DA. A practical two-step quantitative clinical and electrophysiological assessment for the diagnosis and staging of diabetic neuropathy. Diabetes Care. 1994;17:1281-1289. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 821] [Cited by in F6Publishing: 831] [Article Influence: 27.7] [Reference Citation Analysis (0)] |
| 37. | Veves A, Manes C, Murray HJ, Young MJ, Boulton AJ. Painful neuropathy and foot ulceration in diabetic patients. Diabetes Care. 1993;16:1187-1189. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 70] [Cited by in F6Publishing: 72] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 38. | Meijer JW, Smit AJ, Sonderen EV, Groothoff JW, Eisma WH, Links TP. Symptom scoring systems to diagnose distal polyneuropathy in diabetes: the Diabetic Neuropathy Symptom score. Diabet Med. 2002;19:962-965. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 187] [Cited by in F6Publishing: 173] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 39. | Valk GD, de Sonnaville JJ, van Houtum WH, Heine RJ, van Eijk JT, Bouter LM, Bertelsmann FW. The assessment of diabetic polyneuropathy in daily clinical practice: reproducibility and validity of Semmes Weinstein monofilaments examination and clinical neurological examination. Muscle Nerve. 1997;20:116-118. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 2] [Reference Citation Analysis (0)] |
| 40. | Mayfield JA, Sugarman JR. The use of the Semmes-Weinstein monofilament and other threshold tests for preventing foot ulceration and amputation in persons with diabetes. J Fam Pract. 2000;49:S17-S29. [PubMed] [Cited in This Article: ] |
| 41. | Smieja M, Hunt DL, Edelman D, Etchells E, Cornuz J, Simel DL. Clinical examination for the detection of protective sensation in the feet of diabetic patients. International Cooperative Group for Clinical Examination Research. J Gen Intern Med. 1999;14:418-424. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 96] [Cited by in F6Publishing: 84] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 42. | Rith-Najarian SJ, Stolusky T, Gohdes DM. Identifying diabetic patients at high risk for lower-extremity amputation in a primary health care setting. A prospective evaluation of simple screening criteria. Diabetes Care. 1992;15:1386-1389. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 182] [Cited by in F6Publishing: 149] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 43. | Pham H, Armstrong DG, Harvey C, Harkless LB, Giurini JM, Veves A. Screening techniques to identify people at high risk for diabetic foot ulceration: a prospective multicenter trial. Diabetes Care. 2000;23:606-611. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 419] [Cited by in F6Publishing: 348] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 44. | Liniger C, Albeanu A, Bloise D, Assal JP. The tuning fork revisited. Diabet Med. 1990;7:859-864. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 76] [Cited by in F6Publishing: 78] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 45. | Thivolet C, el Farkh J, Petiot A, Simonet C, Tourniaire J. Measuring vibration sensations with graduated tuning fork. Simple and reliable means to detect diabetic patients at risk of neuropathic foot ulceration. Diabetes Care. 1990;13:1077-1080. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 39] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 46. | Boulton AJ, Kubrusly DB, Bowker JH, Gadia MT, Quintero L, Becker DM, Skyler JS, Sosenko JM. Impaired vibratory perception and diabetic foot ulceration. Diabet Med. 1986;3:335-337. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 118] [Cited by in F6Publishing: 123] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 47. | Young MJ, Breddy JL, Veves A, Boulton AJ. The prediction of diabetic neuropathic foot ulceration using vibration perception thresholds. A prospective study. Diabetes Care. 1994;17:557-560. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 417] [Cited by in F6Publishing: 368] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 48. | Arezzo JC. The use of electrophysiology for the assessment of diabetic neuropathy. Neurosci Res Comm. 1997;21:13-23. [DOI] [Cited in This Article: ] |
| 49. | Edwards JL, Vincent AM, Cheng HT, Feldman EL. Diabetic neuropathy: mechanisms to management. Pharmacol Ther. 2008;120:1-34. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 506] [Cited by in F6Publishing: 461] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 50. | Gooch C, Podwall D. The diabetic neuropathies. Neurologist. 2004;10:311-322. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 73] [Cited by in F6Publishing: 76] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 51. | Thomas PK. Nerve biopsy. Diabet Med. 1997;14:345-346. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 52. | Dahlin LB, Eriksson KF, Sundkvist G. Persistent postoperative complaints after whole sural nerve biopsies in diabetic and non-diabetic subjects. Diabet Med. 1997;14:353-356. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 53. | Greene DA, Arezzo JC, Brown MB. Effect of aldose reductase inhibition on nerve conduction and morphometry in diabetic neuropathy. Zenarestat Study Group. Neurology. 1999;53:580-591. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 202] [Cited by in F6Publishing: 180] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 54. | Sima AA, Calvani M, Mehra M, Amato A. Acetyl-L-carnitine improves pain, nerve regeneration, and vibratory perception in patients with chronic diabetic neuropathy: an analysis of two randomized placebo-controlled trials. Diabetes Care. 2005;28:89-94. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 159] [Cited by in F6Publishing: 149] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 55. | McCarthy BG, Hsieh ST, Stocks A, Hauer P, Macko C, Cornblath DR, Griffin JW, McArthur JC. Cutaneous innervation in sensory neuropathies: evaluation by skin biopsy. Neurology. 1995;45:1848-1855. [PubMed] [Cited in This Article: ] |
| 56. | Holland NR, Crawford TO, Hauer P, Cornblath DR, Griffin JW, McArthur JC. Small-fiber sensory neuropathies: clinical course and neuropathology of idiopathic cases. Ann Neurol. 1998;44:47-59. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 244] [Cited by in F6Publishing: 209] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 57. | Polydefkis M, Griffin JW, McArthur J. New insights into diabetic polyneuropathy. JAMA. 2003;290:1371-1376. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 104] [Cited by in F6Publishing: 108] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 58. | Akbari CM, Macsata R, Smith BM, Sidawy AN. Overview of the diabetic foot. Semin Vasc Surg. 2003;16:3-11. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1] [Cited by in F6Publishing: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 59. | Al-Delaimy WK, Merchant AT, Rimm EB, Willett WC, Stampfer MJ, Hu FB. Effect of type 2 diabetes and its duration on the risk of peripheral arterial disease among men. Am J Med. 2004;116:236-240. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 86] [Cited by in F6Publishing: 83] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 60. | Murabito JM, D’Agostino RB, Silbershatz H, Wilson WF. Intermittent claudication. A risk profile from The Framingham Heart Study. Circulation. 1997;96:44-49. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 436] [Cited by in F6Publishing: 367] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 61. | McDermott MM, Greenland P, Liu K, Guralnik JM, Criqui MH, Dolan NC, Chan C, Celic L, Pearce WH, Schneider JR. Leg symptoms in peripheral arterial disease: associated clinical characteristics and functional impairment. JAMA. 2001;286:1599-1606. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 609] [Cited by in F6Publishing: 574] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 62. | American Diabetes Association. Peripheral arterial disease in people with diabetes. Diabetes Care. 2003;26:3333-3341. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 671] [Cited by in F6Publishing: 624] [Article Influence: 29.7] [Reference Citation Analysis (0)] |
| 63. | Hirsch AT, Haskal ZJ, Hertzer NR, Bakal CW, Creager MA, Halperin JL, Hiratzka LF, Murphy WR, Olin JW, Puschett JB. White CJ, White J, White RA, Antman EM, Smith SC, Jr., Adams CD, Anderson JL, Faxon DP, Fuster V, Gibbons RJ, Halperin JL, Hiratzka LF, Hunt SA, Jacobs AK, Nishimura R, Ornato JP, Page RL, Riegel B, American Association for Vascular S, Society for Vascular S, Society for Cardiovascular A, Interventions, Society for Vascular M, Biology, Society of Interventional R, Guidelines AATFoP, American Association of C, Pulmonary R, National Heart L, Blood I, Society for Vascular N, TransAtlantic Inter-Society C, Vascular Disease F. Acc/aha 2005 guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): Executive summary a collaborative report from the american association for vascular surgery/society for vascular surgery, society for cardiovascular angiography and interventions, society for vascular medicine and biology, society of interventional radiology, and the acc/aha task force on practice guidelines (writing committee to develop guidelines for the management of patients with peripheral arterial disease) endorsed by the american association of cardiovascular and pulmonary rehabilitation; national heart, lung, and blood institute; society for vascular nursing; transatlantic inter-society consensus; and vascular disease foundation. J Am Coll Cardiol. 2006;47:1239-1312. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 791] [Cited by in F6Publishing: 735] [Article Influence: 40.8] [Reference Citation Analysis (0)] |
| 64. | Halperin JL. Evaluation of patients with peripheral vascular disease. Thromb Res. 2002;106:V303-V311. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 39] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 65. | Brooks B, Dean R, Patel S, Wu B, Molyneaux L, Yue DK. TBI or not TBI: that is the question. Is it better to measure toe pressure than ankle pressure in diabetic patients. Diabet Med. 2001;18:528-532. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 164] [Cited by in F6Publishing: 134] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 66. | Rutherford RB, Lowenstein DH, Klein MF. Combining segmental systolic pressures and plethysmography to diagnose arterial occlusive disease of the legs. Am J Surg. 1979;138:211-218. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 84] [Cited by in F6Publishing: 88] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 67. | Strandness Jr. DE. Peripheral arterial system. Philadelphia, PA: Lippincott Williams & Wilkins 2002; 118-143. [DOI] [Cited in This Article: ] |
| 68. | Whelan JF, Barry MH, Moir JD. Color flow Doppler ultrasonography: comparison with peripheral arteriography for the investigation of peripheral vascular disease. J Clin Ultrasound. 1992;20:369-374. [PubMed] [DOI] [Cited in This Article: ] |
| 69. | Allard L, Cloutier G, Durand LG, Roederer GO, Langlois YE. Limitations of ultrasonic duplex scanning for diagnosing lower limb arterial stenoses in the presence of adjacent segment disease. J Vasc Surg. 1994;19:650-657. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 71] [Cited by in F6Publishing: 75] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 70. | Larch E, Minar E, Ahmadi R, Schnürer G, Schneider B, Stümpflen A, Ehringer H. Value of color duplex sonography for evaluation of tibioperoneal arteries in patients with femoropopliteal obstruction: a prospective comparison with anterograde intraarterial digital subtraction angiography. J Vasc Surg. 1997;25:629-636. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 79] [Cited by in F6Publishing: 80] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 71. | Carpenter JP, Baum RA, Holland GA, Barker CF. Peripheral vascular surgery with magnetic resonance angiography as the sole preoperative imaging modality. J Vasc Surg. 1994;20:861-869; discussion 869-871. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 101] [Cited by in F6Publishing: 102] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 72. | Leiner T, Kessels AG, Nelemans PJ, Vasbinder GB, de Haan MW, Kitslaar PE, Ho KY, Tordoir JH, van Engelshoven JM. Peripheral arterial disease: comparison of color duplex US and contrast-enhanced MR angiography for diagnosis. Radiology. 2005;235:699-708. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 78] [Cited by in F6Publishing: 80] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 73. | Oyibo SO, Jude EB, Tarawneh I, Nguyen HC, Harkless LB, Boulton AJ. A comparison of two diabetic foot ulcer classification systems: the Wagner and the University of Texas wound classification systems. Diabetes Care. 2001;24:84-88. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 374] [Cited by in F6Publishing: 323] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 74. | Treece KA, Macfarlane RM, Pound N, Game FL, Jeffcoate WJ. Validation of a system of foot ulcer classification in diabetes mellitus. Diabet Med. 2004;21:987-991. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 94] [Cited by in F6Publishing: 94] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 75. | Schaper NC. Diabetic foot ulcer classification system for research purposes: a progress report on criteria for including patients in research studies. Diabetes Metab Res Rev. 2004;20 Suppl 1:S90-S95. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 320] [Cited by in F6Publishing: 323] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 76. | Lipsky BA, Berendt AR, Deery HG, Embil JM, Joseph WS, Karchmer AW, LeFrock JL, Lew DP, Mader JT, Norden C. Diagnosis and treatment of diabetic foot infections. Clin Infect Dis. 2004;39:885-910. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 649] [Cited by in F6Publishing: 572] [Article Influence: 28.6] [Reference Citation Analysis (0)] |
| 77. | Falabella AF. Debridement and wound bed preparation. Dermatol Ther. 2006;19:317-325. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 81] [Cited by in F6Publishing: 69] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 78. | Veves A, Giurini J, LoGerfo F. The diabetic foot: Medical and surgical management. Totowa, NJ: Humana Press 2006; . [DOI] [Cited in This Article: ] |
| 79. | Steed DL, Donohoe D, Webster MW, Lindsley L. Effect of extensive debridement and treatment on the healing of diabetic foot ulcers. Diabetic Ulcer Study Group. J Am Coll Surg. 1996;183:61-64. [PubMed] [Cited in This Article: ] |
| 80. | Bale S. A guide to wound debridement. J Wound Care. 1997;6:179-182. [PubMed] [Cited in This Article: ] |
| 81. | Hultén L. Dressings for surgical wounds. Am J Surg. 1994;167:42S-44S; discussion 44S-45S. [PubMed] [Cited in This Article: ] |
| 82. | Sherman RA. Maggot therapy for treating diabetic foot ulcers unresponsive to conventional therapy. Diabetes Care. 2003;26:446-451. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 171] [Cited by in F6Publishing: 176] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 83. | Mumcuoglu KY, Ingber A, Gilead L, Stessman J, Friedmann R, Schulman H, Bichucher H, Ioffe-Uspensky I, Miller J, Galun R. Maggot therapy for the treatment of intractable wounds. Int J Dermatol. 1999;38:623-627. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 99] [Cited by in F6Publishing: 103] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 84. | Sherman RA, Wyle F, Vulpe M. Maggot therapy for treating pressure ulcers in spinal cord injury patients. J Spinal Cord Med. 1995;18:71-74. [PubMed] [Cited in This Article: ] |
| 85. | Doupis J, Veves A. Classification, diagnosis, and treatment of diabetic foot ulcers. Wounds. 2008;20:117-126. [PubMed] [Cited in This Article: ] |
| 86. | Nabuurs-Franssen MH, Sleegers R, Huijberts MS, Wijnen W, Sanders AP, Walenkamp G, Schaper NC. Total contact casting of the diabetic foot in daily practice: a prospective follow-up study. Diabetes Care. 2005;28:243-247. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 101] [Cited by in F6Publishing: 103] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 87. | Lavery LA, Vela SA, Lavery DC, Quebedeaux TL. Reducing dynamic foot pressures in high-risk diabetic subjects with foot ulcerations. A comparison of treatments. Diabetes Care. 1996;19:818-821. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 143] [Cited by in F6Publishing: 141] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 88. | Birke J, Fred B, Krieger L, Sliman K. The effectiveness of an accommodative dressing in offloading pressure over areas of previous metatarsal head ulceration. Wounds. 2003;15:33-39. [Cited in This Article: ] |
| 89. | Younes NA, Bakri FG. Diabetic foot infection. Saudi Med J. 2006;27:596-603. [PubMed] [Cited in This Article: ] |
| 90. | Lipsky BA, Pecoraro RE, Wheat LJ. The diabetic foot. Soft tissue and bone infection. Infect Dis Clin North Am. 1990;4:409-432. [PubMed] [Cited in This Article: ] |
| 91. | Sumpio BE, Aruny J, Blume PA. The multidisciplinary approach to limb salvage. Acta Chir Belg. 2004;104:647-653. [PubMed] [Cited in This Article: ] |
| 92. | Queen D, Orsted H, Sanada H, Sussman G. A dressing history. Int Wound J. 2004;1:59-77. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 156] [Cited by in F6Publishing: 149] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 93. | Sibbald RG, Torrance G, Hux M, Attard C, Milkovich N. Cost-effectiveness of becaplermin for nonhealing neuropathic diabetic foot ulcers. Ostomy Wound Manage. 2003;49:76-84. [PubMed] [Cited in This Article: ] |
| 94. | Moura LI, Dias AM, Carvalho E, de Sousa HC. Recent advances on the development of wound dressings for diabetic foot ulcer treatment--a review. Acta Biomater. 2013;9:7093-7114. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 444] [Cited by in F6Publishing: 433] [Article Influence: 39.4] [Reference Citation Analysis (1)] |
| 95. | Bryant RA. Acute and chronic wounds. St. Louis, MO: Mosby 2000; . [Cited in This Article: ] |
| 96. | McDonald A, Lesage P. Palliative management of pressure ulcers and malignant wounds in patients with advanced illness. J Palliat Med. 2006;9:285-295. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 50] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 97. | Kalinski C, Schnepf M, Laboy D, Hernandez L, Nusbaum J, McGrinder B, Comfort C, Alvarez OM. Effectiveness of a topical formulation containing metronidazole for wound odor and exudate control. Wounds. 2005;17:84-90. [Cited in This Article: ] |
| 98. | Newman V, Allwood M, Oakes R. The use of metronidazole gel to control the smell of malodorous lesions. Palliat Med. 1989;3:303-305. [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 99. | Viswanathan V, Kesavan R, Kavitha KV, Kumpatla S. A pilot study on the effects of a polyherbal formulation cream on diabetic foot ulcers. Indian J Med Res. 2011;134:168-173. [PubMed] [Cited in This Article: ] |
| 100. | Geronemus RG, Mertz PM, Eaglstein WH. Wound healing. The effects of topical antimicrobial agents. Arch Dermatol. 1979;115:1311-1314. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 108] [Cited by in F6Publishing: 109] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 101. | Kashyap A, Beezhold D, Wiseman J, Beck WC. Effect of povidone iodine dermatologic ointment on wound healing. Am Surg. 1995;61:486-491. [PubMed] [Cited in This Article: ] |
| 102. | Innes ME, Umraw N, Fish JS, Gomez M, Cartotto RC. The use of silver coated dressings on donor site wounds: a prospective, controlled matched pair study. Burns. 2001;27:621-627. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 142] [Cited by in F6Publishing: 131] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 103. | Kilinç H, Sensöz O, Ozdemir R, Unlü RE, Baran C. Which dressing for split-thickness skin graft donor sites. Ann Plast Surg. 2001;46:409-414. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 39] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 104. | Field FK, Kerstein MD. Overview of wound healing in a moist environment. Am J Surg. 1994;167:2S-6S. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 331] [Cited by in F6Publishing: 337] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 105. | Schultz GS, Sibbald RG, Falanga V, Ayello EA, Dowsett C, Harding K, Romanelli M, Stacey MC, Teot L, Vanscheidt W. Wound bed preparation: a systematic approach to wound management. Wound Repair Regen. 2003;11 Suppl 1:S1-28. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 830] [Cited by in F6Publishing: 700] [Article Influence: 33.3] [Reference Citation Analysis (1)] |
| 106. | Trudgian J. Investigating the use of Aquaform Hydrogel in wound management. Br J Nurs. 2000;9:943-948. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 15] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 107. | Mulder GD. Cost-effective managed care: gel versus wet-to-dry for debridement. Ostomy Wound Manage. 1995;41:68-70, 72, 74 passim. [PubMed] [Cited in This Article: ] |
| 108. | Kavitha KV, Tiwari S, Purandare VB, Khedkar S, Bhosale SS, Unnikrishnan AG. Choice of wound care in diabetic foot ulcer: A practical approach. World J Diabetes. 2014;5:546-556. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 83] [Cited by in F6Publishing: 70] [Article Influence: 7.0] [Reference Citation Analysis (3)] |
| 109. | Lohmann M, Thomsen JK, Edmonds ME, Harding KG, Apelqvist J, Gottrup F. Safety and performance of a new non-adhesive foam dressing for the treatment of diabetic foot ulcers. J Wound Care. 2004;13:118-120. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 110. | Cowan T. Wound care handbook. London: Mark Allen Healthcare 2011; . [Cited in This Article: ] |
| 111. | Molan PC, Betts JA. Using honey to heal diabetic foot ulcers. Adv Skin Wound Care. 2008;21:313-316. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 19] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 112. | Molan PC. The evidence supporting the use of honey as a wound dressing. Int J Low Extrem Wounds. 2006;5:40-54. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 225] [Cited by in F6Publishing: 205] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 113. | Fitton AR, Drew P, Dickson WA. The use of a bilaminate artificial skin substitute (Integra) in acute resurfacing of burns: an early experience. Br J Plast Surg. 2001;54:208-212. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 51] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 114. | Veerapur BG, Uppin I, Babu YR. Chronic diabetic wound healing: Recombinant pdgf v/s normal saline. J Pharmaceut Biomed. 2012;24:118-120. [Cited in This Article: ] |
| 115. | Fernández-Montequín JI, Valenzuela-Silva CM, Díaz OG, Savigne W, Sancho-Soutelo N, Rivero-Fernández F, Sánchez-Penton P, Morejón-Vega L, Artaza-Sanz H, García-Herrera A. Intra-lesional injections of recombinant human epidermal growth factor promote granulation and healing in advanced diabetic foot ulcers: multicenter, randomised, placebo-controlled, double-blind study. Int Wound J. 2009;6:432-443. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 73] [Cited by in F6Publishing: 79] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 116. | Vijaykumar H, Pai SA, Pandey V, Kamble P. Comparative study of collagenase and papain-urea based preparations in the management of chronic nonhealing limb ulcers. Indian J Sci Technol. 2011;4:1096-1100. [DOI] [Cited in This Article: ] |
| 117. | Andros G, Armstrong DG, Attinger CE, Boulton AJ, Frykberg RG, Joseph WS, Lavery LA, Morbach S, Niezgoda JA, Toursarkissian B. Consensus statement on negative pressure wound therapy (V.A.C. Therapy) for the management of diabetic foot wounds. Ostomy Wound Manage. 2006;Suppl:1-32. [PubMed] [Cited in This Article: ] |
| 118. | Venturi ML, Attinger CE, Mesbahi AN, Hess CL, Graw KS. Mechanisms and clinical applications of the vacuum-assisted closure (VAC) Device: a review. Am J Clin Dermatol. 2005;6:185-194. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 238] [Cited by in F6Publishing: 254] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 119. | Petrie N, Potter M, Banwell P. The management of lower extremity wounds using topical negative pressure. Int J Low Extrem Wounds. 2003;2:198-206. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 120. | Kot J, Desola J, Simao AG, Gough-Allen R, Houman R, Meliet JL, Galland F, Mortensen C, Mueller PH, Sippinen S. A European code of good practice for hyperbaric oxygen therapy. Int Marit Health. 2004;55:121-130. [PubMed] [Cited in This Article: ] |
| 121. | Strauss MB. Hyperbaric oxygen as an intervention for managing wound hypoxia: its role and usefulness in diabetic foot wounds. Foot Ankle Int. 2005;26:15-18. [PubMed] [Cited in This Article: ] |
| 122. | Mathieu D. Role of hyperbaric oxygen therapy in the management of lower extremity wounds. Int J Low Extrem Wounds. 2006;5:233-235. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 123. | Kessler L, Bilbault P, Ortéga F, Grasso C, Passemard R, Stephan D, Pinget M, Schneider F. Hyperbaric oxygenation accelerates the healing rate of nonischemic chronic diabetic foot ulcers: a prospective randomized study. Diabetes Care. 2003;26:2378-2382. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 163] [Cited by in F6Publishing: 169] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 124. | Fernandes TD. Hyperbaric medicine. Acta Med Port. 2009;22:323-334. [PubMed] [Cited in This Article: ] |
| 125. | Shah J. Hyperbaric oxygen therapy. J Am Col Certif Wound Spec. 2010;2:9-13. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 126. | Goulon M, Bakker D, Marroni A, Mathieu D, Wattel F, Bitterman N. 7th European Consensus Conference on Hyperbaric Medicine (2004). Available from: http://www.echm.org/documents/ECHM 7th Consensus Conference Lille 2004.pdf. [Cited in This Article: ] |
| 127. | Hart CA, Tsui J, Khanna A, Abraham DJ, Baker DM. Exp Biol Med (Maywood). 2013;238:1118-1126. [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |