Published online Jul 15, 2018. doi: 10.4251/wjgo.v10.i7.184
Peer-review started: March 16, 2018
First decision: April 9, 2018
Revised: April 23, 2018
Accepted: May 23, 2018
Article in press: May 23, 2018
Published online: July 15, 2018
Since solid pseudopapillary tumor of the pancreas (SPTP) was officially classified by the World Health Organization in 1996, SPTP has recently received special attention in the literature. Studies have shown that SPTP is a heterogeneous tumor, with a small percentage of patients harboring aggressive behaviors. However, criteria for malignancy grade in SPTP have not been well established. The prognosis of SPTP is generally good, with cases having a chance for long-term survival even with recurrence and/or metastasis after surgical resection. The current American Joint Committee on Cancer/Union for International Cancer Control tumor, node, metastasis staging system is not specific to SPTP. The lack of a predictive staging classification that accurately describes the heterogeneity of this disease hinders meaningful research into optimal individualized therapy. Here we summarize and discuss the associated factors proposed for appraisal of the malignant potential and adverse outcome of SPTP.
Core tip: Solid pseudopapillary tumor of the pancreas (SPTP) is a heterogeneous tumor, with a small percentage of patients harboring aggressive behaviors. Its prognosis is generally good, with cases having a chance for long-term survival even with recurrence and/or metastasis after surgical resection. The lack of a predictive staging classification that accurately describes the heterogeneity of this disease hinders meaningful research into optimal individualized therapy. Here we summarize and discuss the associated factors proposed for appraisal of the malignant potential and adverse outcome of SPTP.
- Citation: You L, Yang F, Fu DL. Prediction of malignancy and adverse outcome of solid pseudopapillary tumor of the pancreas. World J Gastrointest Oncol 2018; 10(7): 184-193
- URL: https://www.wjgnet.com/1948-5204/full/v10/i7/184.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v10.i7.184
Since solid pseudopapillary tumor of the pancreas (SPTP) was officially classified by the World Health Organization (WHO) in 1996, SPTP has been accepted worldwide. It had also been called Frantz tumor, papillary cystic tumor/neoplasm/carcinoma, solid and papillary neoplasm, solid and papillary epithelial neoplasm, solid and cystic tumor, and solid and cystic papillary epithelial neoplasm. Most of the tumors are found in young women in their second or third decade while it is rare in male patients, accounting for 12.05% of all cases. More than half of the patients are under the age of 25 years[1]. Occasionally, it occurs in children[2]. There was no significant difference in age between male and female patients. Approximately one-third of patients were asymptomatic, with the tumors incidentally discovered during physical examination or in work-up for unrelated diseases[1]. Although several genetic alterations such as somatic mutations in exon 3 of CTNNB1, and upregulated genes activated in Wnt/β-catenin, Hedgehog, and androgen receptor signaling pathways have been identified[3-5], the tumorigenesis of SPTP is still not clear. The incidence of SPTP seems to be increasing, and study of this rare tumor is thus of clinical significance.
Previously, SPTP was mostly considered as a benign tumor, but not until the 2010 version of the WHO classification was issued, all SPTPs are considered as low-grade malignant tumors. Studies have shown that SPTP is a heterogeneous tumor, with a small percentage of patients harboring aggressive behaviors[6-8]. Even if the tumor has no evidence of malignant potential, such as perineural invasion, vascular invasion, invasion of pancreatic parenchyma, and infiltration of peripancreatic tissue, it may metastasize to the liver or recur after surgery. Long-term survival can be achieved in SPTP patients with advanced or metastatic disease, which reveals that SPTP is a relatively indolent disease compared with other pancreatic carcinomas. It is difficult to elucidate the natural course of SPTP and to predict its malignancy and outcome after surgery due to limited follow-up studies. As such, SPTP remains a pancreatic surgical enigma and studies have failed to identify prognostic factors predicting its malignant behavior.
The incidence of SPTP has increased markedly in recent years, possibly due to the ready use of modern imaging, diagnostic endoscopy, and physician awareness. Although epidemiologic trends have been documented for pancreatic cystic lesions[9-11], the true incidence and epidemiologic trend for SPTP are less clear. An understanding of its epidemiology has been hampered by the pervasive tendency to report the incidence along with other pancreatic tumors.
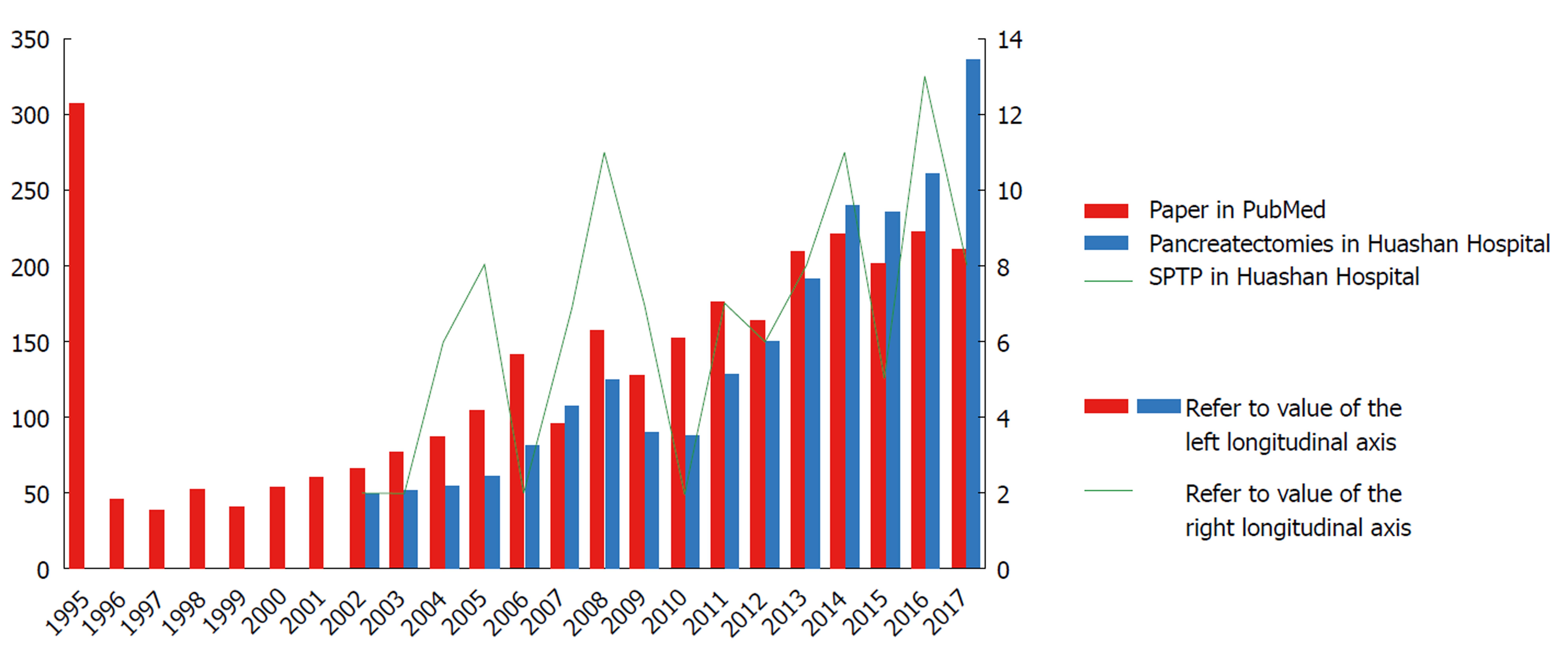
As the incidence of pancreatic tumors in China increases year by year[12], the number of patients with pancreatic diseases admitted to Huashan Hospital affiliated to Fudan University, Shanghai, China has continued to grow, so has the number of surgical procedures performed during the last decade. The number of patients with SPTP also increased during these years, with an average of more than six patients with this disease having been confirmed each year. Literature related to SPTP and the number of patients reported have rapidly grown since 1996 (Figure 1). A total of 390 cases were described in a previous systematic review of SPTP cases reported in China between 1996 and 2006[1]. Law et al[13] conducted a systematic review of English literature concerning SPTP published up to 2012, and identified 2744 cases of SPTP. A nationwide survey from South Korea showed that SPTP ranked as the third most common pancreatic cystic tumors (18.3%)[14]. These figures indicate that SPTP is not uncommon now worldwide. Given the population trend and the paucity of studies available to guide management of patients with SPTP, further research is imperative.
The origin, biological behavior and nature history of SPTP are not fully understood until now, leaving it as an enigmatic entity. SPTP was regarded as a borderline malignant tumor initially due to lack of evidence-based demonstration of true benign tumor. The WHO used the term “low-grade malignant” instead of “benign” in 2010. SPTP has a wide variability of tumor features from completely solid to almost completely cystic. Imaging studies have shown that smaller SPTPs usually appear as completely or mostly solid, while larger SPTPs typically appear as a large well-encapsulated heterogeneous mass with varying solid-cystic components due to necrosis, hemorrhage and degeneration[15]. A recent report revealed that evolution of liver metastasis from SPTP was relatively slow, with the metastatic lesions having a similar growth pattern of primary tumor characterized by a solid-cystic mass with pseudopapillary structures[16].
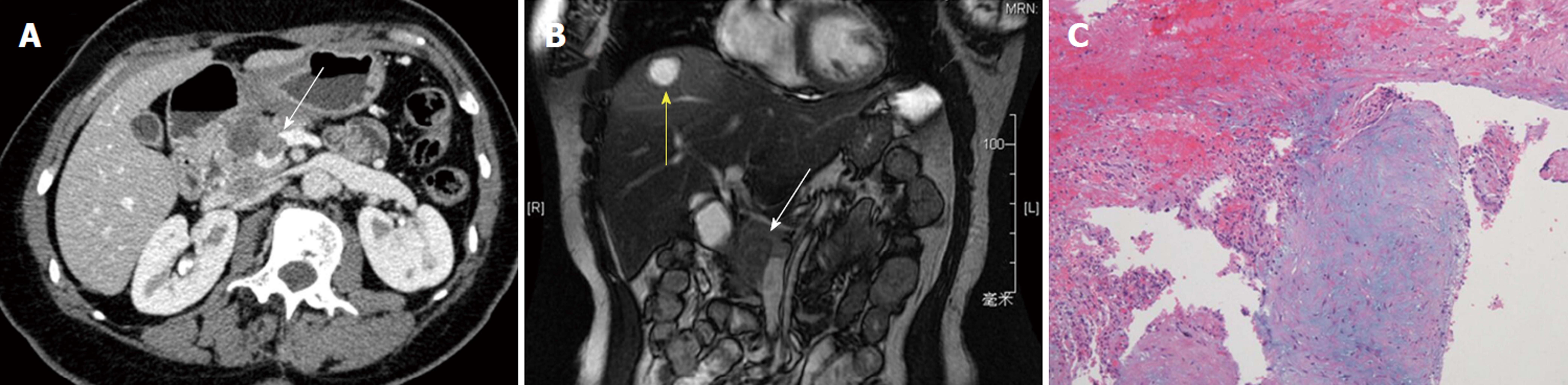
Parallel to the controversy regarding its histogenetic derivation, assessment of the malignant potential of SPTP remained a major controversial issue for decades. Although SPTP is considered as a tumor of low-grade malignancy, patients with this disease occasionally present with invasion into the portal/splenic vein (Figure 2) and/or adjacent organs or liver metastasis, mimicking pancreatic ductal adenecarcinoma. The prognosis of SPTP is generally good, with cases having a chance for long-term survival even with recurrence and/or metastasis after surgical resection. Up to 10% of patients experienced a recurrence and/or metastasis of the disease after years of follow-up, and only a small subset of patients eventually died of this disease[6-8,17-37] (Table 1).
| Ref. | Country | Centers | F/M | Age (yr) | Size (cm) | Malignant, n (%) | Follow up (mo) | R/M (n) | Alive (n) |
| Peng et al[17], 2006 | China | Single | 25/0 | 33 (11-65) | 9.3 (2.5-25) | 3 (12) | 3-111 | 0 | 25 |
| Yu et al[18], 2007 | China | Single | 25/1 | 25.2 (13-57) | 7.5 (3.8-15) | 9 (34.6) | 66 (10-237) | 2 | 24 |
| Machado et al[19], 2008 | Brazil | Single | 27/7 | 23 (10-72) | 7 (1.5-15) | 13 (38.2) | 84 (3-170) | 2 | 33 |
| Lee et al[20], 2008 | South Korea | Multi | 57/5 | 30 (8-63) | 6.5 (1.5-14) | 9 (14.5) | 47.5 (5.1-240.4) | 2 | 62 |
| Matos et al[21], 2009 | United States/Germany | Multi | 20/1 | 33 (13-60) | 5.5 (2.5-19.3) | 3 (14.3) | 55 (7-176) | 0 | 21 |
| Nguyen et al[22], 2011 | Australia | Multi | 30/4 | 33.3 (19.6-42.3) | 6 (4.5-9) | 9 (26.5) | 70 (48-178) | 2 | 32 |
| Kim et al[23], 2011 | South Korea | Single | 98/16 | 36 (11-75) | 4.2 (1.2-15) | 26 (22.8) | 57 (11-177) | 4 | 114 |
| Butte et al[24], 2011 | United States | Single | 38/7 | 38 (10-63) | 4.9 (1.4-20) | 9 (20) | 44 (1-250) | 5 | 381 |
| Cai et al[25], 2013 | China | Single | 30/3 | 29.2 (12-59) | 4.9 (2-15) | 17 (51.5) | 45 (4-118) | 1 | 32 |
| El Nakeeb et al[26], 2013 | Egypt | Single | 22/2 | 24.83 (12-52) | 9.2 (3-25) | 6 (25) | 71.6 (1-180) | 2 | 22 |
| Raman et al[27], 2013 | United States | Single | 43/8 | 29.3 (12.2-74.8) | 5.3 (1.7-11.1) | 11 (21.6) | 37 (0-122) | 1 | 50 |
| Serrano et al[28], 2014 | Canada | Single | 26/6 | 36 (13-64) | 4.7 (1.5-14) | 15 (46.9) | 43 (3-207) | 3 | 31 |
| Suzuki et al[29], 2014 | Japan | Single | 29/5 | 37.1 (15-68) | 4.3 (1-11) | 3 (8.8) | 67 (3-326) | 0 | 34 |
| Kim et al[30], 2014 | South Korea | Single | 85/21 | 36 (10-65) | 4.5 (1-15) | 17 (16) | 56.9 (37-93.4) | 2 | 105 |
| Kang et al[6], 2014 | South Korea | Multi | 317/34 | 36.8 ± 12.4 | 5.7 ± 3.3 | 98 (27.9) | > 6 | 9 | 3162 |
| Estrella et al[7], 2014 | United States | Single | 54/10 | 33 (9-62) | 5 (1.4-20) | 49 (76.6) | 76 (2-203) | 10 | 533 |
| Yu et al[31], 2015 | China | Multi | 93/4 | 31.2 (16-57) | 5.9 (1.5-14) | 16 (16.5) | 70.2 (3.5-221.5) | 3 | 96 |
| Zhang et al[32], 2015 | China | Single | 56/6 | 26 (8-66) | 7.2 (3-15) | 3 (4.8) | 46 (2-135) | 0 | 62 |
| Yang et al[8], 2016 | China | Single | 58/13 | 31 (12-64) | 5 (1-13) | 13 (18.3) | 45 (3-118) | 3 | 70 |
| Irtan et al[33], 2016 | France | Multi | 41/10 | 13.1 (8.7-17.9) | 7 (2-12) | 22 (43.1) | 65 (0.3-221) | 7 | 51 |
| Marchegiani et al[34], 2016 | Italy/United States | Multi | 113/18 | 33 (7-68) | 4 (0.7-20) | 16 (12.2) | 62 (12-304) | 2 | 1054 |
| Xu et al[35], 2017 | China | Single | 93/28 | 33.7 (11-68) | 5 (1-13) | 35 (28.9) | 42.7 (6-97) | 3 | 1005 |
| Song et al[36], 2017 | China | Single | 46/7 | 35.4 (14-67) | 6.4 (2-14) | 10 (18.9) | 48 (3-123) | 2 | 456 |
| Lubezky et al[37], 2017 | Israel | Single | 29/3 | 28.4 ± 12.2 | 5.9 (0.9-14) | 13 (40.6) | 49.2 (1-228) | 4 | 31 |
Studies showed that tumor markers such as carbohydrate antigen 19-9 (CA19-9) and carcinoembryonic antigen (CEA) were usually within normal ranges in patients with this disease. Thus, routine tumor markers are of no value to predict malignant SPTP[1]. Radiologically, SPTP typically appears as a well-capsulated heterogeneous mass with solid and cystic components, while small SPTP commonly represents a solid mass. Capsule and intratumoral hemorrhage are important clues to the diagnosis as they are rarely detected in other pancreatic neoplasms. In some cases, calcification may be present, whereas pancreatic duct dilatation is rarely found. Yang et al[16] reported that the liver metastatic lesions from SPTP increased in sizes gradually with cystic change. The relatively slow evolution of liver metastasis indicates its classic growth pattern. Although the proportion of solid component[38] and incomplete capsule[39,40] were shown to be associated with malignancy by a few reports, no consistent results were demonstrated. Rastogi et al[41] reported that tumors with greater enhancement assessed by contrast-enhanced computed tomography (CT) had aggressive characteristics. However, no correlations between malignancy and other radiological features including calcification were found. These findings indicate that diagnosis of malignant SPTP is difficult with imaging studies. Endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) has been shown to be useful for preoperative definite diagnosis[42]. However, it may cause rupture of tumor and seeding of the needle tract by tumor cells during the procedure[43,44]. Although EUS-FNA has been used more frequently than ever in SPTP[13], its malignant nature is still difficult to confirm because of lack of specific markers.
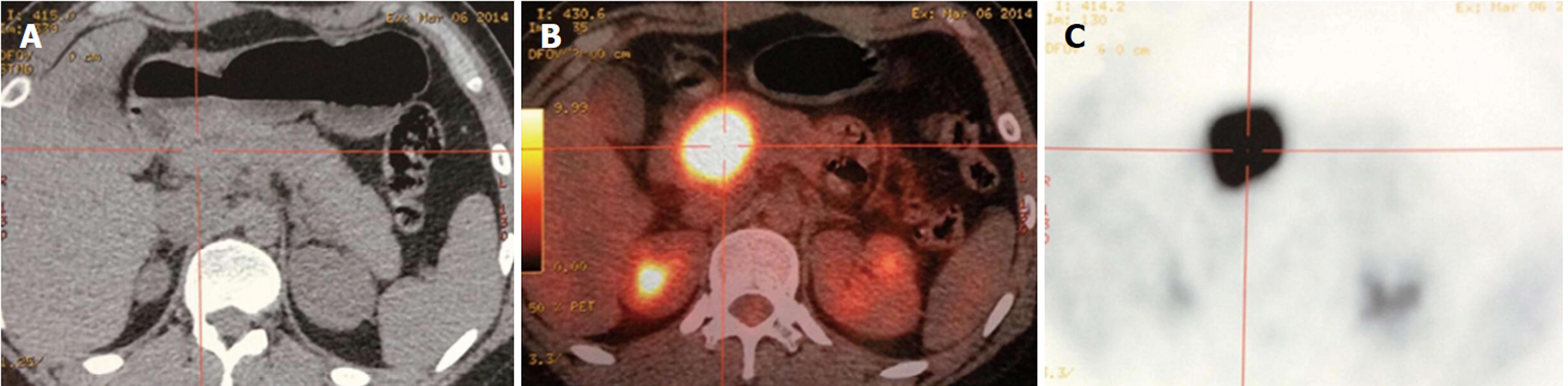
Position emission tomography/computed tomography (PET/CT) is a useful modality in the detection of malignant tumors and has been widely used in patients with pancreatic disease[45]. Limited data are available on PET/CT characteristics of SPTP, making the value of this modality controversial. It has been reported that SPTP has significantly higher tumor size-adjusted metabolic tumor volume and total lesion glycolysis compared with pancreatic ductal adenocarcinoma[46], which leads to a high rate of false positivity in F-18-fluorodeoxyglucose PET/CT when diagnosing this disease (Figure 3). However, this feature suggests that PET/CT may be helpful in detecting metastases of SPTP. Kang et al[47] categorized SPTP into five types according to the PET images and found no association between the fluorodeoxyglucose uptake and malignant potential. Until now, no definitive conclusions can be drawn about the clinical significance of PET/CT in SPTP due to limited cases reported. Thus, the clinical application value of PET scan in SPTP needs further investigation.
Surgical resection is curative in most of the patients with SPTP resulting in a five-year disease-specific survival rate of 98.5%[8]. Long-term survival can be achieved even in those with advanced or metastatic disease. It is interesting to note that patients who underwent limited resection with microscopically positive margins had similar outcomes as those who underwent extensive surgery with R0 resection[7]. The generally good prognosis of SPTP attributes to its relatively low malignant biological behavior. Therefore, aggressive surgical intervention is the optimal therapy for patients with advanced SPTP, even with metastasis. Wang et al[48] reported four patients with liver metastases undergoing aggressive surgery. All the patients received surgical resections for both the primary and metastatic lesions as completely as possible, and had good clinical outcomes during follow-up.
Adjuvant therapies such as chemotherapy (5-fluorouracil and gemcitabine as the main chemotherapeutic drugs) and radiotherapy have been reported in a few patients with a mean survival of 51.1 mo[13]. Sporadic reports found that neoadjvant chemotherapy or radiation therapy could benefit some patients with unresectable tumors[49-51]. Other therapeutic methods including radiofrequency ablation[52], transcatheter arterial chemoembolization[53], selective internal radiotherapy (SIRT)[54] and liver transplantation[55] have also been reported to achieve good results for patients with liver metastasis from SPTP. However, despite a better understanding of this disease, individual treatment of unresectable or metastatic SPTP requires further study.
Malignant SPTP occurs in 18.3% of adult patients and in 43.1% of pediatric patients[8,33]. Preoperative differential diagnosis between benign and malignant SPTP is usually very difficult except in patients with tumor invasion to adjacent organs or with distant metastasis. There has been no consistency about the diagnostic criteria of malignant SPTP until today. Criteria for malignancy in SPTP have not been well established. Many researchers used the WHO-defined criteria for classification of solid pseudopapillary carcinoma, such as angioinvasion, perineural invasion, or deep infiltration into the surrounding tissue or metastasis to confirm the diagnosis of malignant SPTP[30]. Butte et al[24] defined malignant SPTP as locally unresectable tumor with macrovascular invasion, metastatic disease to regional or distant sites, or recurrence of disease after surgery. Ye et al[56] considered SPTP with incomplete capsules as malignant.
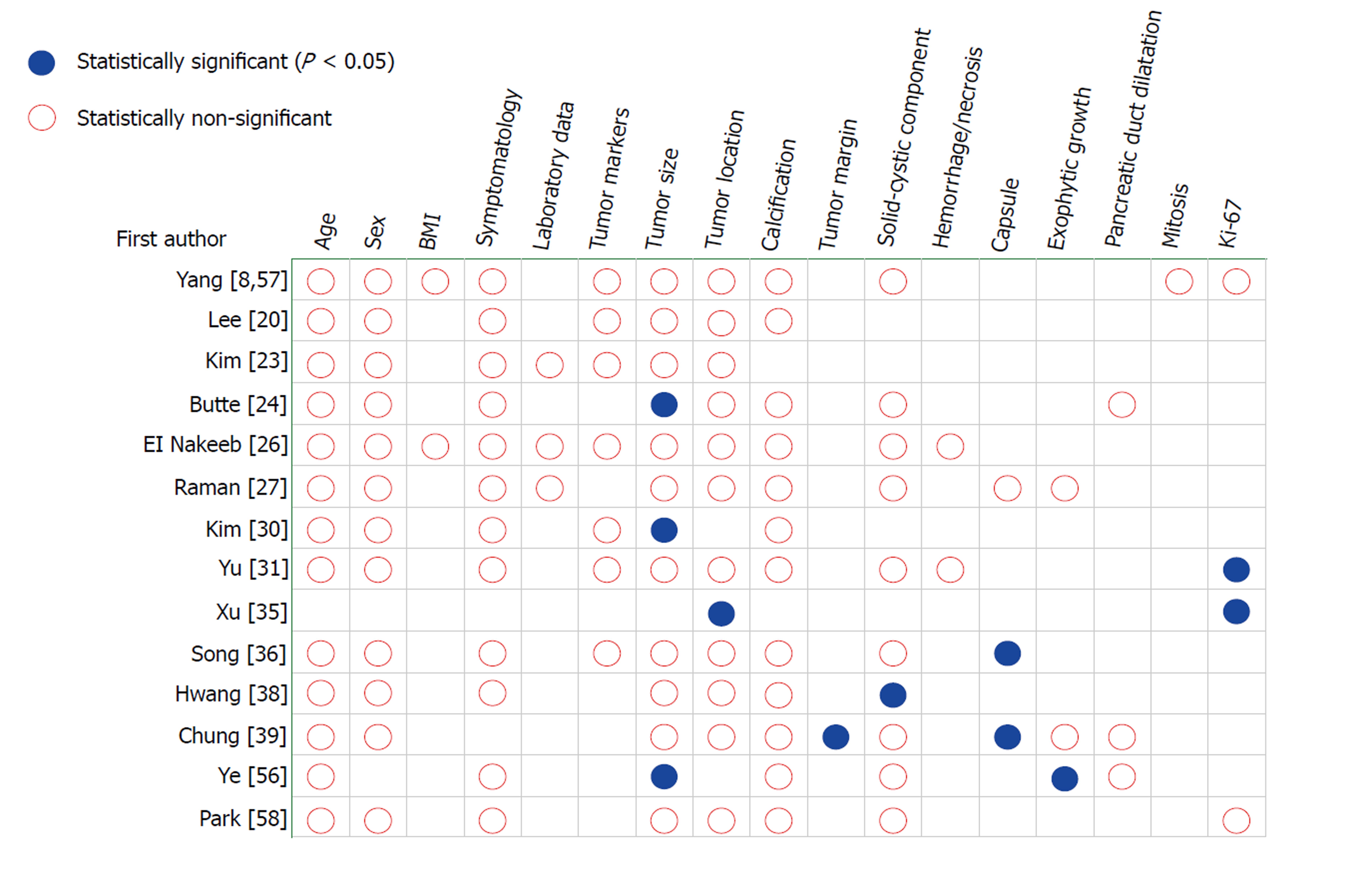
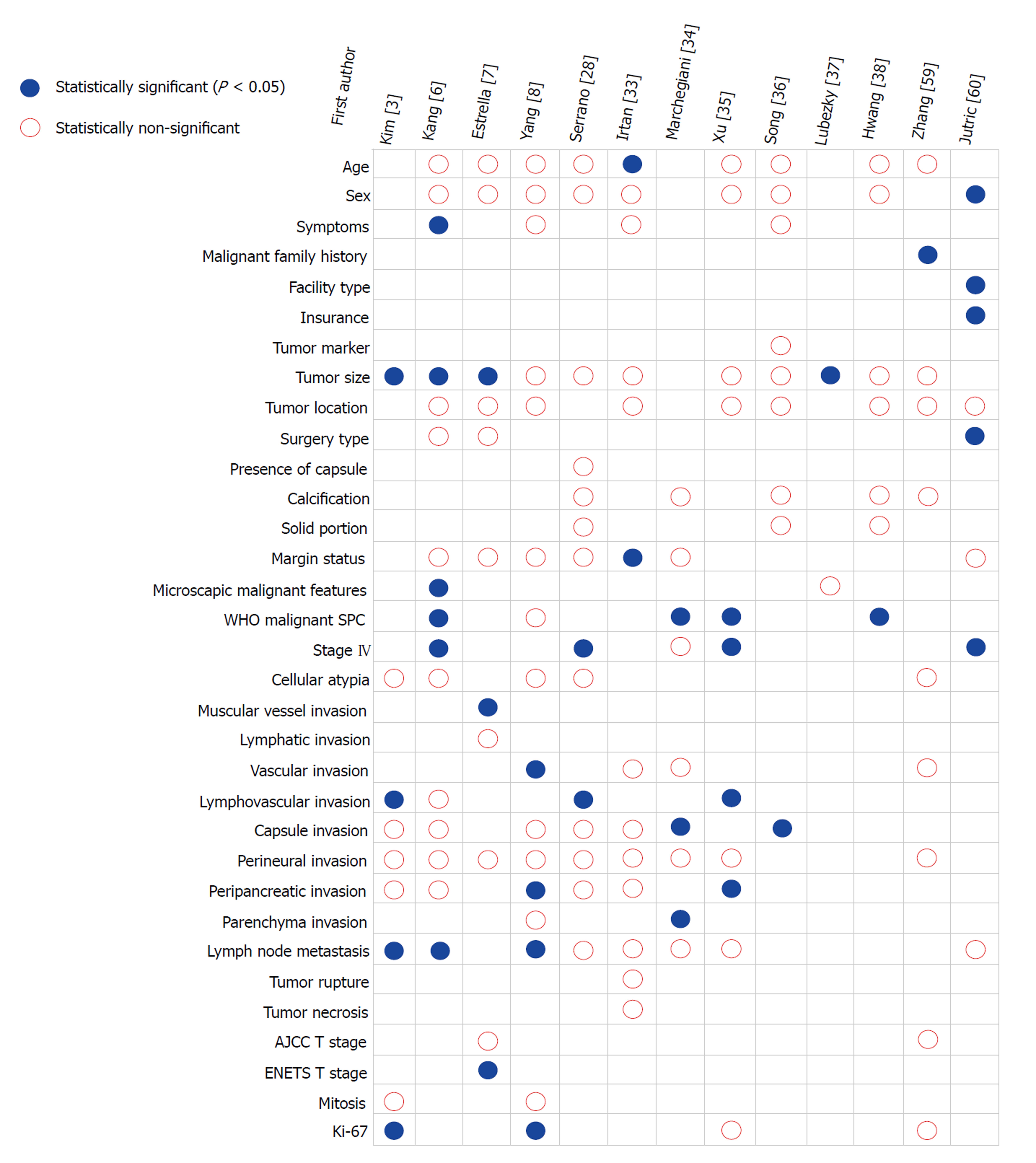
Due to the arbitrary criteria of malignancy used, and rarity of the disease with small proportion of malignancy, conflicting results have been reported about factors associated with malignant potential across institutions[8,20,23,24,26,27,30,31,35,36,38,39,56-58] (Figure 4). Butte et al[24] found that patients with malignant SPTP presented with larger tumor size (P < 0.005). Chung et al[39] explored differential imaging features between malignant and benign SPTP, and found that malignant SPTP more frequently had focal lobulated margins (P = 0.027) and focal discontinuity of capsule (P = 0.005). The study by Ye et al[56] revealed that SPTP with incomplete capsule had larger tumor size (P=0.0015) and mainly exophytic growth pattern (P = 0.0194). Yu et al[31] and Xu et al[35] showed that positive status for Ki-67 correlated with malignancy of SPTP, while Yang et al[8] did not demonstrate any association between the Ki-67 index and malignant SPTP. Most other studies[20,23,26,27,57,58] found no significant differences between benign and malignant SPTP, including age, sex, symptomatology, laboratory data, tumor marker, tumor size and location, tumor composition, growth pattern, and histopathology. Thus, malignancy cannot be easily predicted on the basis of preoperative findings and immunohistochemical patterns.
Most of the patients with SPTP have a good prognosis, while some have a less favorable prognosis because of recurrence and/or metastases. Studies on SPTP were characterized by case reports and small case series lacking of long-term follow-up. Kang et al[6] reported a low recurrence rate (2.8%) and excellent disease free survival and overall survival for SPTP after surgical resection in South Korea. The patients had a 5-year disease free survival of 95.4% and an overall survival of 98.8%. In a recent systematic review, the 5-year and 10-year recurrence free survival was 89.5% and 86.3%, respectively, with the 5-year and 10-year disease specific survival of 92.3% and 86.5%, respectively[8]. It is unclear whether factors associated with malignant potential are statistically significant predictors of adverse outcomes. Although a few recent studies have gathered significant series of SPTP, results are inconclusive with regard to predictors of prognosis[3,6-8,28,33-38,59,60] (Figure 5).
Estrella et al[7] showed that recurrent/metastatic SPTP was significantly associated with larger tumor size, invasion of muscular vessels, and the European Neuroendocrine Tumour Society (ENETS) tumor stage, but not with other clinicopathologic factors. In addition, muscular vessel invasion, ENETS T4 disease, and stage IV were important predictors of poor disease-specific survival after surgical resection. Kang et al[6] demonstrated that tumor size larger than 8 cm, microscopic malignant features, and stage IV were significant prognostic factors for tumor recurrence by multivariate analysis. Irtan et al[33] confirmed that the significant risk factors for recurrence in pediatric cases of SPTP were age < 13.5 years at diagnosis and positive surgical margins at initial tumor resection. It is interesting to note that many other studies[6-8,22,28,34,60] have shown that patients who underwent limited resection and those with R1 resection had the same clinical outcomes as those who received more extensive resection with negative margin. Serrano et al[28] clarified that patients with stage IV or lymphovascular invasion more commonly developed recurrence. Both studies of Marchegiani et al[34] and Hwang et al[38] revealed that recurrence was more common in patients with malignant SPTP which fulfilled the WHO criteria. The study by Zhang et al[59] indicated that recurrence in malignant SPTP correlated with family malignant tumor history.
Several studies[3,57,61,62] have proposed Ki-67 as an additional support to histology for predicting tumor outcome, but conflicting results do exist. Yang et al[8] identified the most discriminating value of Ki-67 index using receiver operating characteristic curve analysis and demonstrated that the prognostic value of Ki-67 was maintained in both the Huashan cohort and the new historical cohort from literature. The result was consistent with a latest study by Kim et al[3]. However, similar to most studies, multivariate analysis could not be performed due to the small number of events. Nevertheless, a much larger number of patients is needed to validate the prognostic relevance of Ki-67.
Recent studies have analyzed the biological behavior of SPTP, however reliable data on long-term follow-up are still needed. Case reports, small retrospective case series, and subjective views rather than facts dominate the available data. These studies have limitations including a small number of cases or events, no uniform parameters studied, lack of a gold standard for judging malignancy, and short length of follow-up. Although some studies adopted the WHO definition of malignancy, a considerable number of studies did not specify the proportion of malignant patients. The excellent prognosis makes overall survival difficult to be assessed, even if several studies have evaluated disease/recurrence free survival. In the light of these limitations, multicenter large-scale studies with long-term follow-up are needed to determine prognostic factors.
To date, no staging systems have been used to stratify patients in any international guidelines for management and follow-up of SPTP[63-65]. The American Joint Committee on Cancer (AJCC)/Union for International Cancer Control (UICC) tumor, node, metastasis (TNM) staging system is a generally accepted standard for cancer staging with the principal aim of facilitating a uniform and standardized analysis of malignant tumors. While the current TNM staging system applies well to pancreatic carcinoma, it is not specific to SPTP. Tumors considered for the TNM system have potentials of local invasiveness (T-categorization) and spread via the lymphatic and blood vessels (N- and M-categories). In view of the rarity of lymphatic and hematogenous metastasis from SPTP, its usefulness in this condition was evidently limited.
The relative rarity of SPTP has delayed the development of evidence-based treatment guidelines. Patients with benign SPTP are still at risk of tumor recurrence or metastasis after surgical resection. Contemporary evidence supports surgery as the primary treatment for patients with operable metastatic SPTP[48,66]. One obstacle to better management of patients is the lack of a predictive classification that accurately describes the complexity and heterogeneity of this disease. In order to provide proper information to predict prognosis, a more specific and standardized histopathological evaluation of SPTP is needed. It is obvious that we urgently need an international consensus for collecting standardized data on SPTP. Better understanding of molecular mechanisms involved in SPTP tumorigenesis is important for improved management. It is probable that novel molecular prognostic variables for SPTP, which may be incorporated into classification systems, will emerge in future.
Manuscript source: Invited manuscript
Specialty type: Oncology
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Sugita R S- Editor: Ji FF L- Editor: Ma JY E- Editor: Tan WW
| 1. | Yang F, Fu DL, Jin C, Long J, Yu XJ, Xu J, Ni QX. Clinical experiences of solid pseudopapillary tumors of the pancreas in China. J Gastroenterol Hepatol. 2008;23:1847-1851. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 37] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 2. | Leraas HJ, Kim J, Sun Z, Ezekian B, Gulack BC, Reed CR, Tracy ET. Solid Pseudopapillary Neoplasm of the Pancreas in Children and Adults: A National Study of 369 Patients. J Pediatr Hematol Oncol. 2018;40:e233-e236. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 3. | Kim EK, Jang M, Park M, Kim H. LEF1, TFE3, and AR are putative diagnostic markers of solid pseudopapillary neoplasms. Oncotarget. 2017;8:93404-93413. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 29] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 4. | Park M, Kim M, Hwang D, Park M, Kim WK, Kim SK, Shin J, Park ES, Kang CM, Paik YK. Characterization of gene expression and activated signaling pathways in solid-pseudopapillary neoplasm of pancreas. Mod Pathol. 2014;27:580-593. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 66] [Cited by in F6Publishing: 71] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 5. | Park M, Lim JS, Lee HJ, Na K, Lee MJ, Kang CM, Paik YK, Kim H. Distinct Protein Expression Profiles of Solid-Pseudopapillary Neoplasms of the Pancreas. J Proteome Res. 2015;14:3007-3014. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 19] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 6. | Kang CM, Choi SH, Kim SC, Lee WJ, Choi DW, Kim SW; Korean Pancreatic Surgery Club. Predicting recurrence of pancreatic solid pseudopapillary tumors after surgical resection: a multicenter analysis in Korea. Ann Surg. 2014;260:348-355. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 87] [Cited by in F6Publishing: 90] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 7. | Estrella JS, Li L, Rashid A, Wang H, Katz MH, Fleming JB, Abbruzzese JL, Wang H. Solid pseudopapillary neoplasm of the pancreas: clinicopathologic and survival analyses of 64 cases from a single institution. Am J Surg Pathol. 2014;38:147-157. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 75] [Cited by in F6Publishing: 71] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 8. | Yang F, Yu X, Bao Y, Du Z, Jin C, Fu D. Prognostic value of Ki-67 in solid pseudopapillary tumor of the pancreas: Huashan experience and systematic review of the literature. Surgery. 2016;159:1023-1031. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 57] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 9. | Soroida Y, Sato M, Hikita H, Hagiwara S, Sato M, Gotoh H, Kato S, Iwai T, Yamazaki T, Yatomi Y. Pancreatic cysts in general population on ultrasonography: Prevalence and development of risk score. J Gastroenterol. 2016;51:1133-1140. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 10. | Zanini N, Giordano M, Smerieri E, Cipolla d’Abruzzo G, Guidi M, Pazzaglini G, De Luca F, Chiaruzzi G, Vitullo G, Piva P. Estimation of the prevalence of asymptomatic pancreatic cysts in the population of San Marino. Pancreatology. 2015;15:417-422. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 11. | Moris M, Bridges MD, Pooley RA, Raimondo M, Woodward TA, Stauffer JA, Asbun HJ, Wallace MB. Association Between Advances in High-Resolution Cross-Section Imaging Technologies and Increase in Prevalence of Pancreatic Cysts From 2005 to 2014. Clin Gastroenterol Hepatol. 2016;14:585-593.e3. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 90] [Cited by in F6Publishing: 95] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 12. | Lin QJ, Yang F, Jin C, Fu DL. Current status and progress of pancreatic cancer in China. World J Gastroenterol. 2015;21:7988-8003. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 181] [Cited by in F6Publishing: 192] [Article Influence: 21.3] [Reference Citation Analysis (1)] |
| 13. | Law JK, Ahmed A, Singh VK, Akshintala VS, Olson MT, Raman SP, Ali SZ, Fishman EK, Kamel I, Canto MI. A systematic review of solid-pseudopapillary neoplasms: are these rare lesions? Pancreas. 2014;43:331-337. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 212] [Cited by in F6Publishing: 216] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 14. | Yoon WJ, Lee JK, Lee KH, Ryu JK, Kim YT, Yoon YB. Cystic neoplasms of the exocrine pancreas: an update of a nationwide survey in Korea. Pancreas. 2008;37:254-258. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 45] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 15. | Baek JH, Lee JM, Kim SH, Kim SJ, Kim SH, Lee JY, Han JK, Choi BI. Small (<or=3 cm) solid pseudopapillary tumors of the pancreas at multiphasic multidetector CT. Radiology. 2010;257:97-106. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 81] [Cited by in F6Publishing: 88] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 16. | Yang F, Jin C, Fu D. Evolution of liver metastasis from solid pseudopapillary tumor of the pancreas. Surgery. 2017;161:1739-1740. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 4] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 17. | Peng CH, Chen DF, Zhou GW, Yang WP, Tao ZY, Lei RQ, Zhange SD, Li HW. The solid-pseudopapillary tumor of pancreas: the clinical characteristics and surgical treatment. J Surg Res. 2006;131:276-282. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 36] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 18. | Yu CC, Tseng JH, Yeh CN, Hwang TL, Jan YY. Clinicopathological study of solid and pseudopapillary tumor of pancreas: emphasis on magnetic resonance imaging findings. World J Gastroenterol. 2007;13:1811-1815. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 32] [Cited by in F6Publishing: 30] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 19. | Machado MC, Machado MA, Bacchella T, Jukemura J, Almeida JL, Cunha JE. Solid pseudopapillary neoplasm of the pancreas: distinct patterns of onset, diagnosis, and prognosis for male versus female patients. Surgery. 2008;143:29-34. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 142] [Cited by in F6Publishing: 154] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 20. | Lee SE, Jang JY, Hwang DW, Park KW, Kim SW. Clinical features and outcome of solid pseudopapillary neoplasm: differences between adults and children. Arch Surg. 2008;143:1218-1221. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 95] [Cited by in F6Publishing: 99] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 21. | Matos JM, Grützmann R, Agaram NP, Saeger HD, Kumar HR, Lillemoe KD, Schmidt CM. Solid pseudopapillary neoplasms of the pancreas: a multi-institutional study of 21 patients. J Surg Res. 2009;157:e137-e142. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 40] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 22. | Nguyen NQ, Johns AL, Gill AJ, Ring N, Chang DK, Clarkson A, Merrett ND, Kench JG, Colvin EK, Scarlett CJ. Clinical and immunohistochemical features of 34 solid pseudopapillary tumors of the pancreas. J Gastroenterol Hepatol. 2011;26:267-274. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 42] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 23. | Kim CW, Han DJ, Kim J, Kim YH, Park JB, Kim SC. Solid pseudopapillary tumor of the pancreas: can malignancy be predicted? Surgery. 2011;149:625-634. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 66] [Cited by in F6Publishing: 75] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 24. | Butte JM, Brennan MF, Gönen M, Tang LH, D’Angelica MI, Fong Y, Dematteo RP, Jarnagin WR, Allen PJ. Solid pseudopapillary tumors of the pancreas. Clinical features, surgical outcomes, and long-term survival in 45 consecutive patients from a single center. J Gastrointest Surg. 2011;15:350-357. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 120] [Cited by in F6Publishing: 109] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 25. | Cai H, Zhou M, Hu Y, He H, Chen J, Tian W, Deng Y. Solid-pseudopapillary neoplasms of the pancreas: clinical and pathological features of 33 cases. Surg Today. 2013;43:148-154. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 26. | El Nakeeb A, Abdel Wahab M, Elkashef WF, Azer M, Kandil T. Solid pseudopapillary tumour of the pancreas: Incidence, prognosis and outcome of surgery (single center experience). Int J Surg. 2013;11:447-457. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 27. | Raman SP, Kawamoto S, Law JK, Blackford A, Lennon AM, Wolfgang CL, Hruban RH, Cameron JL, Fishman EK. Institutional experience with solid pseudopapillary neoplasms: focus on computed tomography, magnetic resonance imaging, conventional ultrasound, endoscopic ultrasound, and predictors of aggressive histology. J Comput Assist Tomogr. 2013;37:824-833. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 28. | Serrano PE, Serra S, Al-Ali H, Gallinger S, Greig PD, McGilvray ID, Moulton CA, Wei AC, Cleary SP. Risk factors associated with recurrence in patients with solid pseudopapillary tumors of the pancreas. JOP. 2014;15:561-568. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 12] [Reference Citation Analysis (0)] |
| 29. | Suzuki S, Hatori T, Furukawa T, Shiratori K, Yamamoto M. Clinical and pathological features of solid pseudopapillary neoplasms of the pancreas at a single institution. Dig Surg. 2014;31:143-150. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 30. | Kim MJ, Choi DW, Choi SH, Heo JS, Sung JY. Surgical treatment of solid pseudopapillary neoplasms of the pancreas and risk factors for malignancy. Br J Surg. 2014;101:1266-1271. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 74] [Cited by in F6Publishing: 83] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 31. | Yu P, Cheng X, Du Y, Yang L, Xu Z, Yin W, Zhong Z, Wang X, Xu H, Hu C. Solid Pseudopapillary Neoplasms of the Pancreas: a 19-Year Multicenter Experience in China. J Gastrointest Surg. 2015;19:1433-1440. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 43] [Cited by in F6Publishing: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 32. | Zhang C, Liu F, Chang H, Li H, Zhou X, Lu J, Qin C, Sun Y, Sun H, Lin J. Less Aggressive Surgical Procedure for Treatment of Solid Pseudopapillary Tumor: Limited Experience from a Single Institute. PLoS One. 2015;10:e0143452. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 33. | Irtan S, Galmiche-Rolland L, Elie C, Orbach D, Sauvanet A, Elias D, Guérin F, Coze C, Faure-Conter C, Becmeur F. Recurrence of Solid Pseudopapillary Neoplasms of the Pancreas: Results of a Nationwide Study of Risk Factors and Treatment Modalities. Pediatr Blood Cancer. 2016;63:1515-1521. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 33] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 34. | Marchegiani G, Andrianello S, Massignani M, Malleo G, Maggino L, Paiella S, Ferrone CR, Luchini C, Scarpa A, Capelli P. Solid pseudopapillary tumors of the pancreas: Specific pathological features predict the likelihood of postoperative recurrence. J Surg Oncol. 2016;114:597-601. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 57] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 35. | Xu Y, Zhao G, Pu N, Nuerxiati A, Ji Y, Zhang L, Rong Y, Lou W, Wang D, Kuang T. One Hundred Twenty-One Resected Solid Pseudopapillary Tumors of the Pancreas: An 8-Year Single-Institution Experience at Zhongshan Hospital, Shanghai, China. Pancreas. 2017;46:1023-1028. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 36. | Song H, Dong M, Zhou J, Sheng W, Zhong B, Gao W. Solid Pseudopapillary Neoplasm of the Pancreas: Clinicopathologic Feature, Risk Factors of Malignancy, and Survival Analysis of 53 Cases from a Single Center. Biomed Res Int. 2017;2017:5465261. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 29] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 37. | Lubezky N, Papoulas M, Lessing Y, Gitstein G, Brazowski E, Nachmany I, Lahat G, Goykhman Y, Ben-Yehuda A, Nakache R. Solid pseudopapillary neoplasm of the pancreas: Management and long-term outcome. Eur J Surg Oncol. 2017;43:1056-1060. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 51] [Cited by in F6Publishing: 47] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 38. | Hwang J, Kim DY, Kim SC, Namgoong JM, Hong SM. Solid-pseudopapillary neoplasm of the pancreas in children: can we predict malignancy? J Pediatr Surg. 2014;49:1730-1733. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 39] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 39. | Chung YE, Kim MJ, Choi JY, Lim JS, Hong HS, Kim YC, Cho HJ, Kim KA, Choi SY. Differentiation of benign and malignant solid pseudopapillary neoplasms of the pancreas. J Comput Assist Tomogr. 2009;33:689-694. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 39] [Cited by in F6Publishing: 41] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 40. | Barat M, Dohan A, Dautry R, Barral M, Pocard M, Soyer P. Solid pseudopapillary adenocarcinoma of the pancreas: CT presentation of a rare malignant variant. Diagn Interv Imaging. 2017;98:823-824. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2] [Cited by in F6Publishing: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 41. | Rastogi A, Assing M, Taggart M, Rao B, Sun J, Elsayes K, Tamm E, Bhosale P. Does Computed Tomography Have the Ability to Differentiate Aggressive From Nonaggressive Solid Pseudopapillary Neoplasm? J Comput Assist Tomogr. 2018;42:405-411. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 42. | Law JK, Stoita A, Wever W, Gleeson FC, Dries AM, Blackford A, Kiswani V, Shin EJ, Khashab MA, Canto MI. Endoscopic ultrasound-guided fine needle aspiration improves the pre-operative diagnostic yield of solid-pseudopapillary neoplasm of the pancreas: an international multicenter case series (with video). Surg Endosc. 2014;28:2592-2598. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 42] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 43. | Virgilio E, Mercantini P, Ferri M, Cunsolo G, Tarantino G, Cavallini M, Ziparo V. Is EUS-FNA of solid-pseudopapillary neoplasms of the pancreas as a preoperative procedure really necessary and free of acceptable risks? Pancreatology. 2014;14:536-538. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 44. | Lévy P, Hammel P, Ruszniewski P. [Do not biopsy pseudopapillary and solid tumors of the pancreas!]. Gastroenterol Clin Biol. 2009;33:501-502. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 45. | Wang XY, Yang F, Jin C, Fu DL. Utility of PET/CT in diagnosis, staging, assessment of resectability and metabolic response of pancreatic cancer. World J Gastroenterol. 2014;20:15580-15589. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 57] [Cited by in F6Publishing: 44] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 46. | Kim YI, Kim SK, Paeng JC, Lee HY. Comparison of F-18-FDG PET/CT findings between pancreatic solid pseudopapillary tumor and pancreatic ductal adenocarcinoma. Eur J Radiol. 2014;83:231-235. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 47. | Kang CM, Cho A, Kim H, Chung YE, Hwang HK, Choi SH, Lee WJ. Clinical correlations with (18)FDG PET scan patterns in solid pseudopapillary tumors of the pancreas: still a surgical enigma? Pancreatology. 2014;14:515-523. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 48. | Wang WB, Zhang TP, Sun MQ, Peng Z, Chen G, Zhao YP. Solid pseudopapillary tumor of the pancreas with liver metastasis: Clinical features and management. Eur J Surg Oncol. 2014;40:1572-1577. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 49. | Maffuz A, Bustamante Fde T, Silva JA, Torres-Vargas S. Preoperative gemcitabine for unresectable, solid pseudopapillary tumour of the pancreas. Lancet Oncol. 2005;6:185-186. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 75] [Cited by in F6Publishing: 67] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 50. | Kanter J, Wilson DB, Strasberg S. Downsizing to resectability of a large solid and cystic papillary tumor of the pancreas by single-agent chemotherapy. J Pediatr Surg. 2009;44:e23-e25. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 51. | Naar L, Spanomichou DA, Mastoraki A, Smyrniotis V, Arkadopoulos N. Solid Pseudopapillary Neoplasms of the Pancreas: A Surgical and Genetic Enigma. World J Surg. 2017;41:1871-1881. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 52. | Li JX, Wu H, Huang JW, Prasoon P, Zeng Y. Synchronous intraoperative radiofrequency ablation for multiple liver metastasis and resection of giant solid pseudopapillary tumors of the pancreas. Chin Med J (Engl). 2012;125:1661-1663. [PubMed] [Cited in This Article: ] |
| 53. | Liu T, He J, Cao D, Huang Y. Successful treatment of liver metastasis from solid pseudopapillary tumor of the pancreas: a case report. Contemp Oncol (Pozn). 2013;17:400-403. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2] [Cited by in F6Publishing: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 54. | Krug S, Bartsch DK, Schober M, Librizzi D, Pfestroff A, Burbelko M, Moll R, Michl P, Gress TM. Successful selective internal radiotherapy (SIRT) in a patient with a malignant solid pseudopapillary pancreatic neoplasm (SPN). Pancreatology. 2012;12:423-427. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 55. | Dovigo AG, Díaz MB, Gütierrez MG, Selles CF, Grobas JP, Valladares M, Suárez F, Marini M. Liver transplantation as treatment in a massive metastasis from Gruber-Frantz pancreatic tumor: a case report. Transplant Proc. 2011;43:2272-2273. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 56. | Ye J, Ma M, Cheng D, Yuan F, Deng X, Zhan Q, Shen B, Peng C. Solid-pseudopapillary tumor of the pancreas: clinical features, pathological characteristics, and origin. J Surg Oncol. 2012;106:728-735. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 57. | Yang F, Jin C, Long J, Yu XJ, Xu J, Di Y, Li J, Fu de L, Ni QX. Solid pseudopapillary tumor of the pancreas: a case series of 26 consecutive patients. Am J Surg. 2009;198:210-215. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 71] [Cited by in F6Publishing: 80] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 58. | Park JK, Cho EJ, Ryu JK, Kim YT, Yoon YB. Natural history and malignant risk factors of solid pseudopapillary tumors of the pancreas. Postgrad Med. 2013;125:92-99. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 59. | Zhang H, Wang W, Yu S, Xiao Y, Chen J. The prognosis and clinical characteristics of advanced (malignant) solid pseudopapillary neoplasm of the pancreas. Tumour Biol. 2016;37:5347-5353. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 60. | Jutric Z, Rozenfeld Y, Grendar J, Hammill CW, Cassera MA, Newell PH, Hansen PD, Wolf RF. Analysis of 340 Patients with Solid Pseudopapillary Tumors of the Pancreas: A Closer Look at Patients with Metastatic Disease. Ann Surg Oncol. 2017;24:2015-2022. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 28] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 61. | Adamthwaite JA, Verbeke CS, Stringer MD, Guillou PJ, Menon KV. Solid pseudopapillary tumour of the pancreas: diverse presentation, outcome and histology. JOP. 2006;7:635-642. [PubMed] [Cited in This Article: ] |
| 62. | Yagcı A, Yakan S, Coskun A, Erkan N, Yıldırım M, Yalcın E, Postacı H. Diagnosis and treatment of solid pseudopapillary tumor of the pancreas: experience of one single institution from Turkey. World J Surg Oncol. 2013;11:308. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 44] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 63. | Del Chiaro M, Verbeke C, Salvia R, Klöppel G, Werner J, McKay C, Friess H, Manfredi R, Van Cutsem E, Löhr M. European experts consensus statement on cystic tumours of the pancreas. Dig Liver Dis. 2013;45:703-711. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 324] [Cited by in F6Publishing: 301] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 64. | Italian Association of Hospital Gastroenterologists and Endoscopists; Italian Association for the Study of the Pancreas, Buscarini E, Pezzilli R, Cannizzaro R, De Angelis C, Gion M, Morana G, Zamboni G, Arcidiacono P, Balzano G, Barresi L, Basso D, Bocus P, Calculli L, Capurso G, Canzonieri V, Casadei R, Crippa S, D’Onofrio M, Frulloni L, Fusaroli P, Manfredi G, Pacchioni D, Pasquali C, Rocca R, Ventrucci M, Venturini S, Villanacci V, Zerbi A, Falconi M; Cystic Pancreatic Neoplasm Study Group. Italian consensus guidelines for the diagnostic work-up and follow-up of cystic pancreatic neoplasms. Dig Liver Dis. 2014;46:479-493. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 81] [Cited by in F6Publishing: 83] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 65. | Vege SS, Ziring B, Jain R, Moayyedi P; Clinical Guidelines Committee; American Gastroenterology Association. American gastroenterological association institute guideline on the diagnosis and management of asymptomatic neoplastic pancreatic cysts. Gastroenterology. 2015;148:819-822; quize12-13. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 629] [Cited by in F6Publishing: 663] [Article Influence: 73.7] [Reference Citation Analysis (0)] |
| 66. | Tajima H, Takamura H, Kitagawa H, Nakayama A, Shoji M, Watanabe T, Tsukada T, Nakanuma S, Okamoto K, Sakai S. Multiple liver metastases of pancreatic solid pseudopapillary tumor treated with resection following chemotherapy and transcatheter arterial embolization: A case report. Oncol Lett. 2015;9:1733-1738. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |