Published online Feb 7, 2019. doi: 10.3748/wjg.v25.i5.539
Peer-review started: November 22, 2018
First decision: December 12, 2018
Revised: December 20, 2018
Accepted: January 14, 2019
Article in press: January 14, 2019
Published online: February 7, 2019
Functional gastrointestinal disorders (FGID) are heterogeneous disorders with a variety of clinical manifestations, primarily defined by signs and symptoms rather than a definite underlying cause. Their pathophysiology remains obscure and, although it is expected to differ according to the specific FGID, disruptions in the brain-gut axis are now thought to be a common denominator in their pathogenesis. The hormone ghrelin is an important component of this axis, exerting a wide repertoire of physiological actions, including regulation of gastrointestinal motility and protection of mucosal tissue. Ghrelin’s gene shows genetic polymorphism, while its protein product undergoes complex regulation and metabolism in the human body. Numerous studies have studied ghrelin’s relation to the emergence of FGIDs, its potential value as an index of disease severity and as a predictive marker for symptom relief during attempted treatment. Despite the mixed results currently available in scientific literature, the plethora of statistically significant findings shows that disruptions in ghrelin genetics and expression are plausibly related to FGID pathogenesis. The aim of this paper is to review current literature studying these associations, in an effort to uncover certain patterns of alterations in both genetics and expression, which could delineate its true contribution to FGID emergence, either as a causative agent or as a pathogenetic intermediate.
Core tip: Functional gastrointestinal disorders are diverse clinical entities whose pathogenesis and phenotype are thought to stem from both genetic and environmental factors. Many reviews have attempted to summarize general pathogenetic mechanisms related to functional gastrointestinal disorders (FGIDs), but more specific knowledge is currently limited. Studies on the brain-gut axis peptide ghrelin have chiefly concentrated on its association with obesity and eating disorders. This review focuses on the possible role of ghrelin in FGID pathogenesis, in an attempt to elucidate the contribution of certain genetic alterations to the emergence of disease.
- Citation: Koutouratsas T, Kalli T, Karamanolis G, Gazouli M. Contribution of ghrelin to functional gastrointestinal disorders’ pathogenesis. World J Gastroenterol 2019; 25(5): 539-551
- URL: https://www.wjgnet.com/1007-9327/full/v25/i5/539.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i5.539
Functional gastrointestinal disorders (FGIDs) comprise a variety of disorders stemming from disruptions of the brain-gut axis. These disorders are classified by gastrointestinal symptoms linked to any combination of motility disruption, visceral hypersensitivity, malformed mucosal and immune function, altered gut microbiota and altered central nervous system processing[1,2]. Some FGIDs are quite prevalent, for instance, functional dyspepsia (FD) and irritable bowel syndrome (IBS), which affect roughly 20% and 10% of the general population[3]. It should be noted that the prevalence of FGIDs varies significantly among different countries worldwide; for instance, IBS has lowest estimated prevalence of 3% in the United States and 6.1% in Japan whereas FD has a prevalence of 26% in the United States and 18.4% in Hong Kong[4,5]. It is postulated that many differences in genetic background, diet, physical environment, socioeconomic status, and microbiome may contribute to this observed variation[4,6-8]; thus, for these mainly symptom-defined disorders, the need for a deeper understanding of their pathogenesis and potential treatment strategies is clinically crucial[2].
The Rome Committee for the Classification of Functional Gastrointestinal Disorders has developed diagnostic criteria for FGIDs, and as of 2018, the Rome IV classification recognizes 33 adult and 20 pediatric FGIDs grouped into 8 categories: esophageal disorders, gastroduodenal disorders, bowel disorders, centrally mediated disorders of gastrointestinal pain, gallbladder and sphincter of Oddi disorders, anorectal disorders, childhood functional gastrointestinal (GI) disorders of neonates/toddlers and childhood functional GI disorders of children/adolescents[2,9].
Many pathophysiological mechanisms have been proposed in search of FGID etiologic factors, including genetics, immune contribution, disorders of the GI serotoninergic (5-hydroxytryptamine, 5-HT) innervation, infections, inflammation, increased intestinal permeability, disrupted bile salt metabolism, changes in the microbiota, the impact of diet, and disturbances in the brain-gut axis.
Ghrelin is a brain-gut axis peptide that was isolated from stomach cells and was found to be the endogenous ligand for growth hormone (GH) secretagogue receptor[10,11]. It is known to have growth hormone inducing and appetite-stimulating effects, while also being of importance in gut motility regulation[12].
As noted earlier, brain-gut axis dysfunctions, namely disruptions of enteric nervous system (ENS) and CNS interconnections, are considered to be critical in FGID pathogenesis[13]. This review explores the role of the brain-gut peptide ghrelin in certain of these disorders, based on current scientific data.
Scientific literature search was conducted using the PubMed and Google Scholar databases with the keywords “ghrelin”, “obestatin”, “ghrelin opposite strand”, “GHSR”, “GOAT”, “functional GI disorders”, “IBS”, “irritable bowel syndrome”, “esophageal disorders”, “gastroduodenal disorders”, “functional dyspepsia”, “bowel disorders”, “gastrointestinal pain”, “gallbladder disorders”, “sphincter of Oddi disorders”, “anorectal disorders”, “CVS” and combinations of the aforementioned.
Ghrelin is a 28 amino acid peptide hormone secreted mainly by the stomach endocrine cells[14]. In humans, the ghrelin gene (GHRL) is located on the short arm of chromosome 3 and comprises five exons coding for the precursor preproghrelin, the first two coding for the functional 28-amino acid region[14,15]. Opposite to the coding strand is an antisense gene called GHRLOS (ghrelin opposite strand) spanning 44kb, whose RNA products undergo alternative splicing and are likely to serve as non-coding regulatory RNAs in the ghrelin axis[16]. The GHRL gene is greatly polymorphic, having more than 300 coding or non-coding region single nucleotide polymorphisms (SNPs) currently validated by 1000 genomes on dbSNP, 4 of which are listed as pathogenic[17].
Ghrelin is found in plasma in two major forms: one that has undergone n-octanoylation at Ser3, called n-octanoyl- or acyl- ghrelin, and one that lacks this modification, referred to as des-acyl ghrelin[14]. The enzyme ghrelin-O-acyltransferase acts on des-acyl ghrelin to produce n-octanoyl ghrelin in ghrelin-producing cells[14,18]. Des-acyl ghrelin is the predominant circulating form of ghrelin[19].
The n-octanoylation at the N-terminal serine is necessary for ghrelin to exert its whole range of physiological actions, such as GH induction, although des-acyl ghrelin can also have certain physiologic effects, for instance, anti-apoptotic and hypotensive actions on the cardiovascular system[14,18,20]. Des-acyl ghrelin and acyl-ghrelin can have both similar and opposing physiological actions (Table 1)[19].
| Parameter | Acyl-ghrelin | Des-acyl ghrelin | Ref. |
| Appetite1 | Increase | Decrease | [87] |
| Metabolism1 | Positive energy balance | Negative energy balance | [87] |
| Adipogenesis | Increase | Increase | [88] |
| Cardiomyocyte and endothelial cell death | Inhibition | Inhibition | [20] |
| Papillary muscle contractility | Negative inotropy | Negative inotropy | [89] |
| Blood pressure | Decrease | Decrease | [90] |
| Insulin release1 | Decrease | Increase | [91] |
| Insulin sensitivity1 | Decrease | Increase | [92] |
| Muscle wasting | Decrease | Decrease | [93] |
| Gastric emptying1 | Increase | Decrease | [94] |
| Gastric motility1 | Increase | Decrease | [95] |
Ghrelin is mainly produced by P/D1 oxyntic cells (similar to mouse X/A cells) mainly in the fundus of the stomach[18,21,22]. Its mRNA is also found in small and large intestine cells, in pancreatic alpha and beta islet cells, in kidney glomeruli and, to a certain degree, in most human tissues[14,19,21-23]. In secretory granules of endocrine cells in the upper small intestine, ghrelin is colocalized with motilin, a peptide hormone involved in phase III of the migrating motor complex[24,25].
Ghrelin’s receptor is known as growth hormone secretagogue receptor (GHSR) and is a rhodopsin-like G-protein coupled receptor (GPCR) with seven membrane-spanning segments[26,27]. The receptor’s gene is located on the large arm of chromosome 3, and its mRNA product can be alternatively spliced to two different transcripts, 1a and 1b; however, it is only the 1a mRNA protein product, GHSR1a, which can bind ghrelin[26]. GHSR1a also displays constitutive activity[24]. Type 1a mRNA has been found in the arcuate and ventromedial nuclei of the hypothalamus, in the pituitary, thyroid gland, and adrenals, whereas the type 1b mRNA is ubiquitously present in human tissue[19,22]. It has been argued that GHSR1b, the 1b type mRNA protein product, may modulate the constitutive activity displayed by GHSR1a[26]. When bound to ligand, be that ghrelin or the 36% homologous protein motilin, the GHSR1a receptor has been found to activate many downstream signaling cascades, including the MAPK, mTOR, AMPK, PI3K/Akt and PLC pathways, while displaying functional agonism[14,18,24,26].
Many factors are thought to regulate ghrelin secretion by the stomach P/D1 like cells. These include positive regulators such as fasting, muscarinic stimulation by the vagus nerve, beta-adrenergic stimulation, estrogen, cholecystokinin, glucagon, and deep sleep as well as inhibitory regulators, including alpha-adrenergic stimulation, insulin, glucose, leptin, long chain fatty acids and somatostatin[18,24]. Total plasma ghrelin concentration peaks before meals and drops to a minimum within an hour postprandially[18,21]. Protein and fatty meals have been found to reduce total and acyl-ghrelin plasma levels, whereas carbohydrate meals cause an initial elevation of both levels followed by suppression, thought to be mediated by insulin[28]. Women have been found to have higher levels of total plasma ghrelin[29].
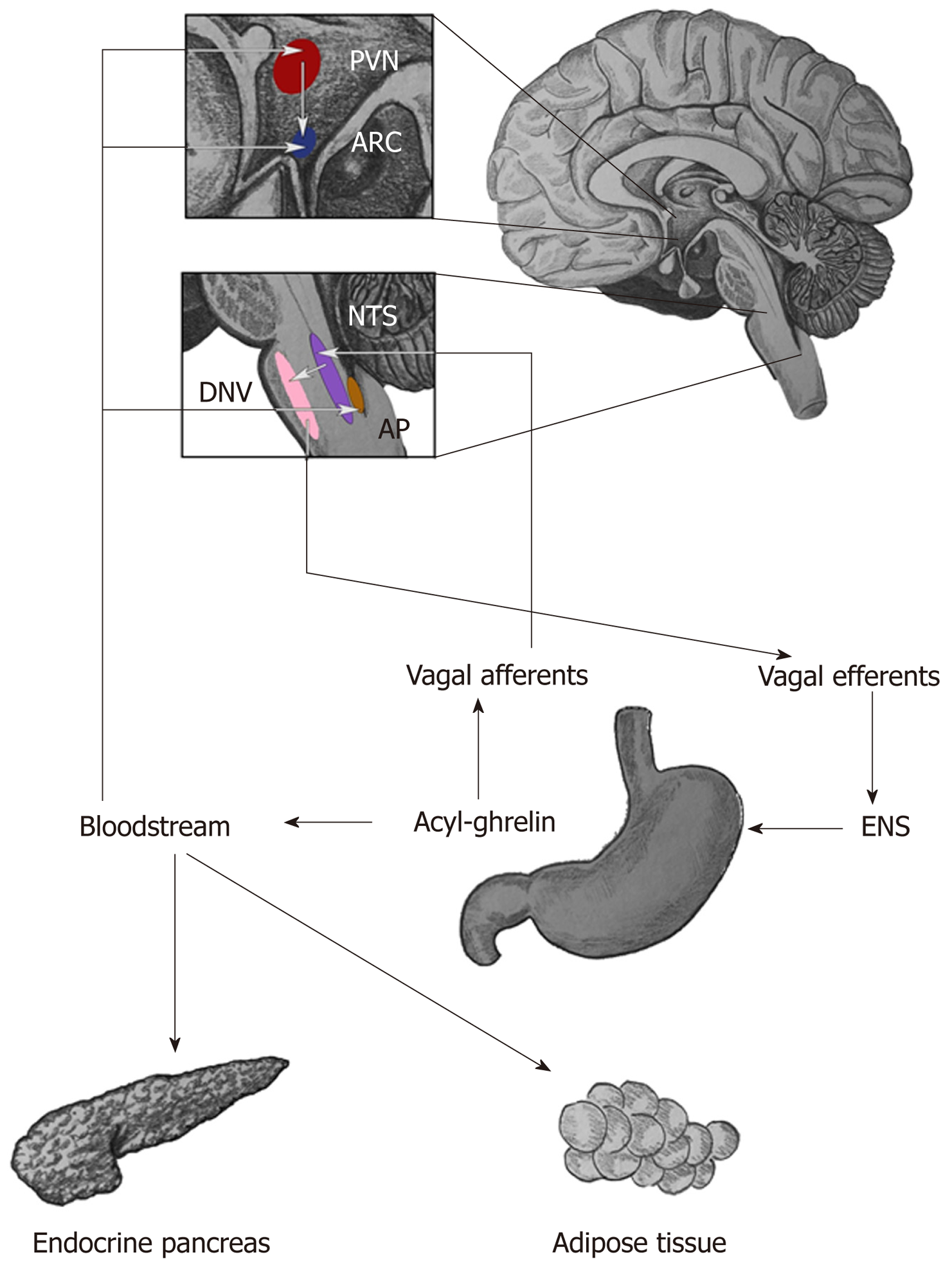
Ghrelin displays a large repertoire of physiological actions (Figure 1). It directly acts on pituitary somatotrophs to increase growth hormone (GH) secretion synergistically with growth hormone-releasing hormone (GHRH), being the more potent GH secretagogue[19,27,30]. Regarding appetite stimulation, ghrelin acts on vagal afferent neurons in the stomach which project to the nucleus tractus solitarius (NTS) of the brainstem, a CNS region which participates in visceral reflexes and connects to hypothalamic feeding centers[24,31,32]. Acting on arcuate nucleus cells of the hypothalamus, it upregulates production of the orexigenic peptides neuropeptide Y (NPY) and agouti-related peptide (AgRP)[19,33,34]; therefore, it increases appetite. The same effect could also be explained by ghrelin’s action in the area postrema (AP), a brainstem region poorly shielded by the blood-brain barrier that is responsible for humoral emesis initiation[35]. Acting on AP neurons, ghrelin could decrease nausea and increase feeding[24]. In general metabolism, ghrelin decreases fat utilization[36].
In the gastrointestinal system, ghrelin is considered to be a component of the brain-gut axis[37]. Its effects include gastric acid secretion and generation of nitric oxide and prostaglandins, vasodilators which protect the gastric mucosa from ischemic stress and alcoholic damage[18,21]. Similar to the structurally related protein motilin, it can induce migrating motor complexes, causing premature phase III activity in the stomach and duodenum[18] and, moreover, accelerates gastric emptying[14,21,37,38]. It also has been found to increase muscle tone in the proximal stomach[38]; furthermore, ghrelin decreases the small intestine transit time but has no such effect on the colon[21]; however, ghrelin agonists which, unlike ghrelin, penetrate the blood-spinal cord barrier have been found to increase colonic contractility and propulsion, activating the defecation reflex[39]. It should be noted that, low ghrelin levels have been detected in obese individuals or individuals after bariatric surgery, and administration of physiological doses of exogenous ghrelin to these individuals does not appreciably affect gastric motility[40].
Pathophysiologically, ghrelin serves as a marker for chronic gastritis and its levels are indicative of the histopathological severity of H. pylori infection; furthermore, it has been reported that in gastric adenocarcinoma and advanced grade colorectal cancer, plasma ghrelin levels are significantly lower, possibly due to inadequate ghrelin production by the affected tissue[41]. Ghrelin receptor type 1b (GSHR1b), although formerly considered physiologically inert, has been found to promote proliferative and invasive activity in colorectal malignancies[26,41]. Regarding inflammatory GI disorders, ulcerative colitis and ileal Crohn disease patients have significantly elevated plasma ghrelin levels. Ghrelin is also positively correlated with serum inflammatory markers in active inflammatory bowel disease patients[42]. Ghrelin could exert antinociceptive effects in GI disease, acting on opioid receptors and downregulating TRPV1, an ion channel expressed chiefly on nociceptive sensory neurons, as found in patients with IBS[43].
FD is a clinical disorder whose symptoms originate in the gastroduodenal region[44]. According to Rome criteria, the symptom complex is often related to ingestion of a meal and includes epigastric pain, postprandial fullness, bloating, early satiety, belching, nausea, vomiting and epigastric burning. Attempts have been made to simplify the intricate heterogeneity of the dyspepsia symptom complex and a subdivision based on the predominant symptoms of either pain or discomfort has been proposed; thus, there are two clinically distinct FD syndromes, although these often overlap clinically: postprandial distress syndrome (PDS; comprising early satiety or meal-related fullness) and epigastric pain syndrome[44]. In a recent meta-analysis, the overall pooled prevalence of uninvestigated dyspepsia was 20.8% (95%CI: 17.8% to 23.9%)[45,46]; over 75% of those patients will not have a structrural cause for their symptoms after a thorough work-up and are thought to have FD[45,46].
Although multiple mechanisms of FD pathogenesis (including abnormal gastric motility, visceral hypersensitivity, low grade mucosal inflammation and cellular changes in enteric nerves, muscle or interstitial cells of Cajal) have been suggested, its underlying etiology and pharmacological therapy remain unclear. As we have already mentioned, ghrelin, a gut-derived peptide found in the stomach, plays a role in the regulation of gastric motility[42,47].
Several papers have been published studying associations between plasma ghrelin levels and FD (Table 2 includes a summary of the literature on changing serum ghrelin levels in patients with FD, which remains controversial).
| Ref. | Participants | Parameters | FD patients compared to Controls | P value |
| Shindo et al[57] | PDS (n = 76)/Controls ( n = 20) | Acyl-ghrelin, plasma levels | Lower | < 0.05a |
| Shinomiya et al[54] | FD (n = 18)/Controls (n = 18) | Acyl-ghrelin, plasma levels | Higher | > 0.05 |
| Des-acyl ghrelin, plasma levels | Lower | > 0.05 | ||
| Nishizawa et al[49] | FD (n = 47)/Controls (n = 17) | Total ghrelin, plasma levels | Higher | < 0.05a |
| Acyl-ghrelin, plasma levels | Higher | < 0.05a | ||
| Lanzini et al[48] | FD (n = 39)/Controls ( n = 53) | Total ghrelin, plasma levels | Higher | < 0.01a |
| Takamori et al[51] | FD (n = 16)/Controls (n = 19) | Des-acyl ghrelin, fasting state, plasma levels | Lower | 0.0019a |
| Acyl-ghrelin, fasting state, plasma levels | Lower | 0.4191 | ||
| Des-acyl ghrelin to acyl-ghrelin ratio, fasting state, plasma levels | Lower | 0.0154a | ||
| Lee et al[50] | FD (n = 42)/Controls (n = 14) | Total ghrelin, fasting state, plasma levels | Lower | < 0.05a |
| Choi et al[56] | FD (n = 39)/Controls (n = 38) | Male patients, acyl-ghrelin, plasma levels | Lower | 0.017a |
| FD (n = 65)/Controls (n = 49) | Female patients, acyl-ghrelin, plasma levels | Lower | 0.348 | |
| Lee et al[65] | FD (n = 167)/Controls (n = 434) | rs42451, A allele | More frequent (PDS subgroup) | < 0.05a |
| rs42451, AA genotype | More frequent (PDS subgroup) | < 0.05a |
Studies on the relationship between FD and ghrelin reported that circulating levels of total ghrelin were significantly higher in patients with FD than in controls[48,49]. The higher levels of ghrelin occurred at 30 min. after the meal which is the time when most of the dyspeptic symptoms usually occur, supporting the idea of a putative role for ghrelin in the etiology of the dyspeptic symptoms in FD patients.
In contrast, Lee et al[50] found that dysmotility-like FD patients had lower total ghrelin levels in the fasting state than controls and that these patients displayed no postprandial drop in total ghrelin levels. In addition, in the same group of FD patients, Takamori et al[51] showed lower fasting total ghrelin levels than in controls and no difference between fasting and postprandial levels in FD patients. Interestingly, a study on a rat model for chronic stress showed that stress initially decreases gastric emptying, due to sympathetic nervous system activation, but eventually gastric emptying accelerates and plasma ghrelin levels are found elevated. This was considered an indication that alterations in ghrelin physiology may in part be responsible for gastric motility disorders which are triggered by stress, such as FD[52,53].
Keeping in mind that the acylated form of ghrelin exhibits physiologic activity (i.e. appetite, adipogenesis etc.), many studies have focused on this subtype (Table 1). Shinomiya et al[54] found that although fasting levels of acylated ghrelin were not significantly different between healthy controls and FD patients, its plasma levels correlated with the subjective symptom score in patients with FD, especially females. In accordance to this study, Takamori et al[51] reported that fasting and postprandial levels of acylated ghrelin were similar in FD patients and healthy controls. Authors suggested that in those patients with dysmotility-like FD, secretion and metabolism of ghrelin are the pathologically affected factors and not the step of acylation. A recent study by Kim et al[55], confirmed the lack of difference in plasma acylated ghrelin levels between healthy controls and FD patients; however, abnormal plasma acylated ghrelin levels before or after a meal related to specific symptoms seen in patients with FD; remarkably, FD patients with higher fasting plasma levels of acylated ghrelin suffered from less epigastric pain, whereas post prandial decrease of the high fasting levels of acylated ghrelin were associated with higher early satiety scores. Apparently, higher fasting plasma levels of acylated ghrelin yield a gastroprotective effect on the gastric mucosa, while postprandial levels interfere with the gastric accommodation reflex.
In contrast, other studies measured lower acylated ghrelin levels in samples from FD patients. Significantly lower plasma acylated ghrelin levels in FD patients compared to controls were reported by Choi et al[56], however only for male patients, suggesting that FD pathogenesis in females may employ additional mechanisms. Shindo et al[57] also reported that acylated ghrelin levels were significantly lower in PDS patients than in controls, while des-acyl ghrelin levels did not differ significantly among EPS, PDS patients, and healthy volunteers. They suggested that lower acylated ghrelin levels may reflect the gastric atrophy and ghrelin-producing cell loss in PDS patients; furthermore, they found that in PDS patients, plasma acyl-ghrelin levels are inversely correlated with the diagnostic marker Tmax, the time of maximal gastric excretion, to a significant degree. They suggested that the study of gastric emptying and plasma ghrelin levels could be useful in the diagnosis of PDS[57,58]; indeed, plasma acylated ghrelin levels were lower in the PDS group than in controls, EPS, and combination of PDS and EPS groups[59]. Interestingly, H. pylori eradication increased plasma acylated ghrelin levels, in contrast to a systematic review which reported that H. pylori eradication had no influence in circulating ghrelin levels[60]. Evidently, it was not just the presence of H. pylori, but also the histological changes associated with it, such as intestinal metaplasia, that affected plasma ghrelin levels[61].
There have also been studies linking certain genetic polymorphisms of the GHRL gene with certain parameters of the FD phenotype (Table 3). Ando et al[62] found that the Leu72Met genotype that is a SNP outside the region coding for mature ghrelin was significantly and independently correlated with low plasma acylated ghrelin levels; however, this correlation, as well as a correlation with the Gln90Leu genotype, could not be confirmed in a later study on H. pylori negative FD patients[63]. In contrast, this study found a significant correlation between the preproghrelin 3056TT genotype and the plasma levels of acylated ghrelin, although it did not find a correlation between any of the Arg51Gln, Preproghrelin3056TC, Leu72Met and Gln90Leu polymorphisms and FD phenotypes[63]. Trying to clarify the putative associations of Gln90Leu and Leu72Met genotypes with ghrelin activity, Yamawaki et al[64] showed that Leu72Met SNP was associated with early phase of gastric emptying but not with entire gastric emptying in FD patients, while there was no significant association between this SNP and early phase of gastric emptying in healthy volunteers; moreover, the Gln90Leu SNP has been associated with depression severity in FD patients[64]. The GHRL rs42451 AA genotype, as well as the A allele, have been reported to be more frequent among PDS patients, compared to controls (Table 2)[65].
| Ref. | Participants | Genotype | Phenotype | P value |
| Ando et al[62] | Healthy (n = 264) | Leu72Met, AA+CA compared to CC | Higher acyl-ghrelin plasma levels | 0.015a |
| 3056T C, CC+TC compared to TT | Higher acyl-ghrelin plasma levels | 0.021a | ||
| Futagami et al[63] | FD (n = 74)/PDS (n = 51)/EPS (n = 23)/Controls | 3056TC, TC+CC compared to TT | Higher acyl-ghrelin plasma levels | 0.025a |
| Leu72Met, CA+AA compared to CC | Higher acyl-ghrelin plasma levels | 0.347 | ||
| Yamawaki et al[64] | FD (n = 74) | Leu72Met, GG compared to GT+TT | Early phase of gastric emptying, 10 minutes after meal | 0.038a |
| FD (n = 74) | Leu72Met, GT+TT compared to GG | Higher SRQ-D scores (GT+TT compared to GG genotype) | 0.0097a | |
| Controls (n = 64) | Leu72Met, GG compared to GT+TT | Early phase of gastric emptying, 10 minutes after meal | > 0.05 |
Regarding treatment outlooks, FD has been considered as a possible indication for ghrelin and ghrelin agonists, such as the synthetic pentapeptide relamorelin, given ghrelin’s prokinetic effects on the stomach[39,53]. Ghrelin’s use is limited due to short plasma half-life and bioavailability, so mainly ghrelin agonists are currently being tested in clinical trials for the treatment of diabetic gastroparesis and chronic constipation[39,53,66,67]. Akamizu et al[47] postulated the possible usefulness of ghrelin in the treatment of weight loss in FD patients, after a 2009 trial in which intravenous ghrelin was administered to patients with FD, causing increased appetite (approx. by 30% before and after treatment) in 4 out of 5. This trial did not, however, yield statistically significant results[47].
Recently, rikkunshito, a standardized Japanese herbal medicine with ghrelin signal-enhancing actions, has been studied as a candidate for the treatment of FD[68,69]. A 2014 double-blind randomized control trial showed that treatment with rikkunshito significantly reduced epigastric pain in patients with the PDS subtype of FD, an effect which was not accompanied by an elevation in plasma ghrelin levels[69]. A later study reported that a low baseline level of plasma des-acyl ghrelin was significantly and independently associated with stronger symptom relief following treatment with rikkunshito in H. pylori negative patients, which was also not accompanied by elevated post-treatment plasma ghrelin levels[68]. These findings suggest that the clinical value of ghrelin, ghrelin agonists and ghrelin-enhancing drugs in treating FD is probably greater in patients with certain patterns of alteration in ghrelin physiology.
IBS is characterized by the presence of recurrent abdominal pain that is associated with defecation or a change in bowel habits (i.e. constipation, diarrhea, or an alteration between constipation and diarrhea). Bloating could often be present, but it is not mandatory for diagnosis. Symptom onset should occur at least 6 mo before diagnosis and symptoms should be present during the last 3 mo[70]. Diagnosis focuses on symptom assessment, as biochemical, histopathological and radiological tests are currently of little clinical value[71]. Based on its clinical features, IBS can be subdivided into constipation predominant (IBS-C), diarrhea predominant (IBS-D) and alternating stool pattern (IBS-A) IBS. IBS is the most common gastrointestinal disorder encountered in primary care with a world-wide prevalence of around 11.2% (95%CI: 9.8%-12.8%) based on a recent meta-analysis[45,72].
IBS is a disorder whose pathophysiology has yet to be completely elucidated. Many pathophysiological mechanisms have been correlated with the emergence of IBS symptoms, including visceral hypersensitivity, abnormal gut motility, dysregulated intestinal secretions, autonomous dysfunction, increased intestinal permeability, neurotransmitters, behavioral changes, immune activation, endocrine dysfunction and altered gut microbiota, and previous infection[73-75]; moreover, up to 33% patients with IBS had a positive for IBS family history compared to 2% of the controls, a finding which suggests the contribution of a strong genetic component[71,76].
Focusing on endocrine dysfunction, ghrelin is one of the gut hormones which are thought to be involved in IBS pathogenesis; therefore, its expression patterns have been studied in IBS patients compared to controls (Table 4). In a 2008 study, El-Salhy et al[77] reported that the density of ghrelin-immunoreactive cells in stomach oxyntic mucosa was significantly lower in IBS-C and significantly higher in IBS-D patients than in healthy controls, with the plasma and tissue extract concentrations of total and acyl ghrelin showing no significant difference. They argued that the latter finding may suggest the existence of compensation in ghrelin secretion, and it is the occasional instability of this mechanism that may result in intermittent IBS symptoms[77]. Sjӧlund et al[78] were later able to confirm that circulating ghrelin levels covaried with motilin in IBS patients but not in healthy volunteers. Authors suggested that the two peptides’ synergy together with low vagal activity may result in typical IBS dysmotility[78]. Şahin-Eryılmaz et al[79] later reported higher plasma ghrelin levels in IBS-D patients and higher ghrelin staining in the antral mucosal glands of IBS-C patients, compared to controls. No studies have reported different patterns of ghrelin alterations among IBS patients according to gender, in spite of IBS predominantly affecting women, which more often complain of abdominal pain, compared to men who primarily complain of diarrhea, findings which indicate different pathophysiological basis for the disorder between genders[80].
| Ref. | Participants | Parameters | IBS patients compared to Controls | P value |
| Salhy et al[77] | IBS-C (n = 19)/Controls (n = 10) | Ghrelin cell density in gastric mucosa | Higher | < 0.0001a |
| IBS-D (n = 18)/Controls (n = 10) | Ghrelin cell density in gastric mucosa | Lower | < 0.0001a | |
| Şahin-Eryılmaz et al[79] | IBS-C (n = 30)/Controls (n = 30) | Ghrelin staining, antral mucosal glands | Higher | 0.038a |
| IBS-D (n = 30)/Controls (n = 30) | Plasma ghrelin levels | Higher | 0.001a | |
| Sjölund et al[78] | IBS (n = 9)/Controls (n = 9) | Acyl-ghrelin and motilin plasma levels covariation | Covariation present in IBS patients only | < 0.02a |
| Des-acyl ghrelin and motilin plasma levels covariation | Covariation present in IBS patients only | < 0.04a | ||
| Total ghrelin and motilin plasma levels covariation | Covariation present in IBS patients only | < 0.004a | ||
| Lee et al[65] | IBS (n = 60)/Controls (n = 434) | rs3755777, G allele | Less frequent (IBS-D subgroup) | < 0.05a |
| rs3755777, GG+CG genotypes | Less frequent (IBS-D subgroup) | < 0.05a | ||
| Russo et al[36] | IBS-D (n = 28)/Controls (n = 19) | Leu72Met, T allele | Less frequent | 0.027a |
| Leu72Met, GT genotype | Less frequent | 0.041a | ||
| Videlock et al[81] | IBS (n = 20)/Controls (n = 10) | GHRLOS lncRNA levels | Down-regulated, IBS | < 0.05a |
| Down-regulated, IBS-D | < 0.05a |
Regarding ghrelin polymorphisms (Table 4), Lee et al[65] found that the G allele of rs3755777 was significantly associated with IBS-D, as the GG and CG genotypes were significantly less frequent in IBS-D patients than in controls. Russo et al[36] later found the Leu72Met T allele and GT genotype to be significantly reduced in IBS-D patients compared to controls.
Considering other alterations in ghrelin expression, a 2017 study found that GHRLOS, the GHRL opposite strand transcript assumed to have a regulatory function in the ghrelin expression (see above), was downregulated in sigmoid biopsy samples from IBS patients compared to controls[81]. This finding underlines the contribution of DNA epigenetics - regulatory RNA, DNA methylation, histone covalent modification- in the emergence of many pathophysiologically obscure diseases, possibly guiding future IBS research towards new directions[82].
Unlike FD, ghrelin and ghrelin receptor agonists are not thought to be of as great therapeutic value in IBS, because ghrelin weakly affects the distal GI tract, which is more involved in IBS pathophysiology[53].
Cyclic vomiting syndrome (CVS) is a functional gastrointestinal disorder characterized by recurrent episodes of severe nausea and vomiting, separated by relatively asymptomatic periods. It was previously thought to be a pediatric disorder, but current knowledge recognizes its incidence in adults. However, it is still underdiagnosed[83]. CVS pathophysiology includes brain-gut axis dysfunction, mitochondrial mutations, autonomic involvement, cannabinoid, and opioid receptor mutations and dysregulation of the hypothalamic-pituitary-adrenal axis[83].
A 2018 study by Hejazi et al[84] found that in adult patients with CVS, fasting serum ghrelin levels were significantly elevated in comparison with controls. It was speculated that elevated ghrelin levels could be related to the rapid gastric emptying found during the recovery phase of CVS[84].
Infant colic is a functional gastrointestinal disorder of early infancy defined as “paroxysms of irritability, fussing or crying lasting more than 3 h per day and occurring more than 3 d each week”[9,85]. Many organic factors are thought to participate in its pathogenesis, including diet, gas, intestinal hypermotility and hormones, although it has been argued that these explain only a few of infant colic cases[85].
Savino et al[86] reported in a 2006 paper that infants with colic have significantly higher serum ghrelin levels compared to controls; however, they could not clarify whether this was a cause or effect of the condition. One proposed hypothesis was that ghrelin induces hypermotility in the physiologically immature infant intestine[86].
It is evident that no clear pattern of ghrelin genetic alterations has been established which can be integrated into a model for FGID pathogenesis. Even for certain types of FGIDs, statistically significant results often contradict previously published data or later studies fail to reproduce previous significant findings. The role of ghrelin in the emergence, progress, or treatment of FGIDs could be important, depending on certain disease subtype, patient gender, age, or other related physiological factors. Ghrelin’s wide range of physiological effects, although, shows that a pathophysiological involvement is certainly plausible.
The complicated nature of these clinical entities suggests that ghrelin disruptions could account for a small percentage of cases or that they only weakly affect the disease phenotype. This indicates that further studies should employ more specific strategies in order to elucidate its role in pathogenesis, to control for confounding factors and to clarify whether ghrelin is an independent causal factor or an intermediate in disease mechanisms
The plethora of statistically significant data, despite displaying occasional contradiction, does encourage further research into the genetic or epigenetic alterations of the GHRL gene, as these may possibly serve as substrate for the different pathophysiological pathways which give rise to the variety of FGID phenotypes. Further studies should focus on thoroughly applying official diagnostic criteria among patient groups, on recognizing FGID subtypes as distinct pathophysiological entities, on controlling for factors (e.g. BMI and gender) which also affect ghrelin physiology, on studying specific clinical parameters (e.g. gastric emptying times) in relation to certain ghrelin alterations, and on exploring other polymorphisms related to the hormone’s physiology, such as those referring to the ghrelin receptor gene or regulatory lncRNAs and miRNAs. Comparable results between studies are also imperative for meta-analysis, and few currently exist as such.
We thank Dr. Dimitris G Pollalis for helping us with the figure design.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Greece
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Capasso R, Kalaitzakis E, Ro S S- Editor: Gong ZM L- Editor: A E- Editor: Huang Y
| 1. | Schmulson MJ, Drossman DA. What Is New in Rome IV. J Neurogastroenterol Motil. 2017;23:151-163. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 419] [Cited by in F6Publishing: 371] [Article Influence: 53.0] [Reference Citation Analysis (0)] |
| 2. | Drossman DA. Functional Gastrointestinal Disorders: History, Pathophysiology, Clinical Features and Rome IV. Gastroenterology. 2016; Epub ahead of print. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1366] [Cited by in F6Publishing: 1153] [Article Influence: 144.1] [Reference Citation Analysis (1)] |
| 3. | Talley NJ. Scope of the problem of functional digestive disorders. Eur J Surg Suppl. 1998;35-41. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 4] [Cited by in F6Publishing: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 4. | Canavan C, West J, Card T. The epidemiology of irritable bowel syndrome. Clin Epidemiol. 2014;6:71-80. [PubMed] [Cited in This Article: ] |
| 5. | Mahadeva S, Goh KL. Epidemiology of functional dyspepsia: a global perspective. World J Gastroenterol. 2006;12:2661-2666. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 256] [Cited by in F6Publishing: 249] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 6. | Scarpato E, Kolacek S, Jojkic-Pavkov D, Konjik V, Živković N, Roman E, Kostovski A, Zdraveska N, Altamimi E, Papadopoulou A, Karagiozoglou-Lampoudi T, Shamir R, Bar Lev MR, Koleilat A, Mneimneh S, Bruzzese D, Leis R, Staiano A; MEAP Group. Prevalence of Functional Gastrointestinal Disorders in Children and Adolescents in the Mediterranean Region of Europe. Clin Gastroenterol Hepatol. 2018;16:870-876. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 45] [Cited by in F6Publishing: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 7. | Aziz I, Palsson OS, Törnblom H, Sperber AD, Whitehead WE, Simrén M. The Prevalence and Impact of Overlapping Rome IV-Diagnosed Functional Gastrointestinal Disorders on Somatization, Quality of Life, and Healthcare Utilization: A Cross-Sectional General Population Study in Three Countries. Am J Gastroenterol. 2018;113:86-96. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 98] [Cited by in F6Publishing: 113] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 8. | Boronat AC, Ferreira-Maia AP, Matijasevich A, Wang YP. Epidemiology of functional gastrointestinal disorders in children and adolescents: A systematic review. World J Gastroenterol. 2017;23:3915-3927. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 67] [Cited by in F6Publishing: 72] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 9. | Hyman PE, Milla PJ, Benninga MA, Davidson GP, Fleisher DF, Taminiau J. Childhood functional gastrointestinal disorders: neonate/toddler. Gastroenterology. 2006;130:1519-1526. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 389] [Cited by in F6Publishing: 319] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 10. | Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature. 1999;402:656-660. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5961] [Cited by in F6Publishing: 5707] [Article Influence: 228.3] [Reference Citation Analysis (0)] |
| 11. | Howard AD, Feighner SD, Cully DF, Arena JP, Liberator PA, Rosenblum CI, Hamelin M, Hreniuk DL, Palyha OC, Anderson J, Paress PS, Diaz C, Chou M, Liu KK, McKee KK, Pong SS, Chaung LY, Elbrecht A, Dashkevicz M, Heavens R, Rigby M, Sirinathsinghji DJ, Dean DC, Melillo DG, Patchett AA, Nargund R, Griffin PR, DeMartino JA, Gupta SK, Schaeffer JM, Smith RG, Van der Ploeg LH. A receptor in pituitary and hypothalamus that functions in growth hormone release. Science. 1996;273:974-977. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1445] [Cited by in F6Publishing: 1343] [Article Influence: 48.0] [Reference Citation Analysis (0)] |
| 12. | Ogiso K, Asakawa A, Amitani H, Inui A. Ghrelin: a gut hormonal basis of motility regulation and functional dyspepsia. J Gastroenterol Hepatol. 2011;26 Suppl 3:67-72. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 13. | Jones MP, Dilley JB, Drossman D, Crowell MD. Brain-gut connections in functional GI disorders: anatomic and physiologic relationships. Neurogastroenterol Motil. 2006;18:91-103. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 224] [Cited by in F6Publishing: 214] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 14. | Sato T, Nakamura Y, Shiimura Y, Ohgusu H, Kangawa K, Kojima M. Structure, regulation and function of ghrelin. J Biochem. 2012;151:119-128. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 141] [Cited by in F6Publishing: 143] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 15. | Higgins SC, Gueorguiev M, Korbonits M. Ghrelin, the peripheral hunger hormone. Ann Med. 2007;39:116-136. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 108] [Cited by in F6Publishing: 83] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 16. | Seim I, Carter SL, Herington AC, Chopin LK. Complex organisation and structure of the ghrelin antisense strand gene GHRLOS, a candidate non-coding RNA gene. BMC Mol Biol. 2008;9:95. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 17. | ghrl. SNP - NCBI [Internet]. Cited 2018-10-10. Available from: https://www.ncbi.nlm.nih.gov/snp/?term=ghrl. [Cited in This Article: ] |
| 18. | Veldhuis JD, Bowers CY. Integrating GHS into the Ghrelin System. Int J Pept. 2010;2010. [PubMed] [Cited in This Article: ] |
| 19. | Kojima M, Kangawa K. Ghrelin: structure and function. Physiol Rev. 2005;85:495-522. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1111] [Cited by in F6Publishing: 1096] [Article Influence: 57.7] [Reference Citation Analysis (0)] |
| 20. | Baldanzi G, Filigheddu N, Cutrupi S, Catapano F, Bonissoni S, Fubini A, Malan D, Baj G, Granata R, Broglio F, Papotti M, Surico N, Bussolino F, Isgaard J, Deghenghi R, Sinigaglia F, Prat M, Muccioli G, Ghigo E, Graziani A. Ghrelin and des-acyl ghrelin inhibit cell death in cardiomyocytes and endothelial cells through ERK1/2 and PI 3-kinase/AKT. J Cell Biol. 2002;159:1029-1037. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 526] [Cited by in F6Publishing: 530] [Article Influence: 24.1] [Reference Citation Analysis (0)] |
| 21. | Inui A, Asakawa A, Bowers CY, Mantovani G, Laviano A, Meguid MM, Fujimiya M. Ghrelin, appetite, and gastric motility: the emerging role of the stomach as an endocrine organ. FASEB J. 2004;18:439-456. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 293] [Cited by in F6Publishing: 264] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 22. | Gnanapavan S, Kola B, Bustin SA, Morris DG, McGee P, Fairclough P, Bhattacharya S, Carpenter R, Grossman AB, Korbonits M. The tissue distribution of the mRNA of ghrelin and subtypes of its receptor, GHS-R, in humans. J Clin Endocrinol Metab. 2002;87:2988. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 687] [Cited by in F6Publishing: 761] [Article Influence: 34.6] [Reference Citation Analysis (0)] |
| 23. | Date Y, Nakazato M, Hashiguchi S, Dezaki K, Mondal MS, Hosoda H, Kojima M, Kangawa K, Arima T, Matsuo H, Yada T, Matsukura S. Ghrelin is present in pancreatic alpha-cells of humans and rats and stimulates insulin secretion. Diabetes. 2002;51:124-129. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 380] [Cited by in F6Publishing: 396] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 24. | Sanger GJ, Broad J, Callaghan B, Furness JB. Ghrelin and Motilin Control Systems in GI Physiology and Therapeutics. Handb Exp Pharmacol. 2017;239:379-416. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 25. | Wierup N, Björkqvist M, Weström B, Pierzynowski S, Sundler F, Sjölund K. Ghrelin and motilin are cosecreted from a prominent endocrine cell population in the small intestine. J Clin Endocrinol Metab. 2007;92:3573-3581. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 70] [Cited by in F6Publishing: 67] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 26. | Yin Y, Li Y, Zhang W. The growth hormone secretagogue receptor: its intracellular signaling and regulation. Int J Mol Sci. 2014;15:4837-4855. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 85] [Cited by in F6Publishing: 84] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 27. | Davenport AP, Bonner TI, Foord SM, Harmar AJ, Neubig RR, Pin JP, Spedding M, Kojima M, Kangawa K. International Union of Pharmacology. LVI. Ghrelin receptor nomenclature, distribution, and function. Pharmacol Rev. 2005;57:541-546. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 156] [Cited by in F6Publishing: 176] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 28. | Foster-Schubert KE, Overduin J, Prudom CE, Liu J, Callahan HS, Gaylinn BD, Thorner MO, Cummings DE. Acyl and total ghrelin are suppressed strongly by ingested proteins, weakly by lipids, and biphasically by carbohydrates. J Clin Endocrinol Metab. 2008;93:1971-1979. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 200] [Cited by in F6Publishing: 200] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 29. | Makovey J, Naganathan V, Seibel M, Sambrook P. Gender differences in plasma ghrelin and its relations to body composition and bone - an opposite-sex twin study. Clin Endocrinol (Oxf). 2007;66:530-537. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 40] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 30. | Takaya K, Ariyasu H, Kanamoto N, Iwakura H, Yoshimoto A, Harada M, Mori K, Komatsu Y, Usui T, Shimatsu A, Ogawa Y, Hosoda K, Akamizu T, Kojima M, Kangawa K, Nakao K. Ghrelin strongly stimulates growth hormone release in humans. J Clin Endocrinol Metab. 2000;85:4908-4911. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 382] [Cited by in F6Publishing: 395] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 31. | Andresen MC, Kunze DL. Nucleus tractus solitarius--gateway to neural circulatory control. Annu Rev Physiol. 1994;56:93-116. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 272] [Cited by in F6Publishing: 314] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 32. | Wren AM, Seal LJ, Cohen MA, Brynes AE, Frost GS, Murphy KG, Dhillo WS, Ghatei MA, Bloom SR. Ghrelin enhances appetite and increases food intake in humans. J Clin Endocrinol Metab. 2001;86:5992. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1222] [Cited by in F6Publishing: 1242] [Article Influence: 54.0] [Reference Citation Analysis (0)] |
| 33. | Morton GJ, Schwartz MW. The NPY/AgRP neuron and energy homeostasis. Int J Obes Relat Metab Disord. 2001;25 Suppl 5:S56-S62. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 171] [Cited by in F6Publishing: 180] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 34. | Briggs DI, Andrews ZB. Metabolic status regulates ghrelin function on energy homeostasis. Neuroendocrinology. 2011;93:48-57. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 82] [Cited by in F6Publishing: 83] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 35. | Fry M, Ferguson AV. Ghrelin: central nervous system sites of action in regulation of energy balance. Int J Pept. 2010;2010. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 31] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 36. | Russo F, Chimienti G, Linsalata M, Clemente C, Orlando A, Riezzo G. The obestatin/ghrelin ratio and ghrelin genetics in adult celiac patients before and after a gluten-free diet, in irritable bowel syndrome patients and healthy individuals. Eur J Gastroenterol Hepatol. 2017;29:160-168. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 37. | Peeters TL. Ghrelin: a new player in the control of gastrointestinal functions. Gut. 2005;54:1638-1649. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 129] [Cited by in F6Publishing: 130] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 38. | Tack J, Depoortere I, Bisschops R, Delporte C, Coulie B, Meulemans A, Janssens J, Peeters T. Influence of ghrelin on interdigestive gastrointestinal motility in humans. Gut. 2006;55:327-333. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 259] [Cited by in F6Publishing: 276] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 39. | Sanger GJ, Furness JB. Ghrelin and motilin receptors as drug targets for gastrointestinal disorders. Nat Rev Gastroenterol Hepatol. 2016;13:38-48. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 80] [Cited by in F6Publishing: 90] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 40. | Camilleri M, Papathanasopoulos A, Odunsi ST. Actions and therapeutic pathways of ghrelin for gastrointestinal disorders. Nat Rev Gastroenterol Hepatol. 2009;6:343-352. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 89] [Cited by in F6Publishing: 77] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 41. | Waseem T, Javaid-Ur-Rehman, Ahmad F, Azam M, Qureshi MA. Role of ghrelin axis in colorectal cancer: a novel association. Peptides. 2008;29:1369-1376. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 59] [Cited by in F6Publishing: 63] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 42. | Cheung CK, Wu JC. Role of ghrelin in the pathophysiology of gastrointestinal disease. Gut Liver. 2013;7:505-512. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 43. | Mao Y, Li Z, Chen K, Yu H, Zhang S, Jiang M, Ma Y, Liang C, Liu H, Li H, Hua Q, Zhou H, Sun Y, Fan X. Antinociceptive Effect of Ghrelin in a Rat Model of Irritable Bowel Syndrome Involves TRPV1/Opioid Systems. Cell Physiol Biochem. 2017;43:518-530. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 44. | Tack J, Talley NJ, Camilleri M, Holtmann G, Hu P, Malagelada JR, Stanghellini V. Functional gastroduodenal disorders. Gastroenterology. 2006;130:1466-1479. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1243] [Cited by in F6Publishing: 1154] [Article Influence: 64.1] [Reference Citation Analysis (0)] |
| 45. | Ford AC, Marwaha A, Sood R, Moayyedi P. Global prevalence of, and risk factors for, uninvestigated dyspepsia: a meta-analysis. Gut. 2015;64:1049-1057. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 279] [Cited by in F6Publishing: 271] [Article Influence: 30.1] [Reference Citation Analysis (0)] |
| 46. | Ford AC, Marwaha A, Lim A, Moayyedi P. What is the prevalence of clinically significant endoscopic findings in subjects with dyspepsia? Systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2010;8:830-837, 837.e1-837.e2. [PubMed] [Cited in This Article: ] |
| 47. | Akamizu T, Iwakura H, Ariyasu H, Kangawa K. Ghrelin and functional dyspepsia. Int J Pept. 2010;2010. [PubMed] [Cited in This Article: ] |
| 48. | Lanzini A, Magni P, Petroni ML, Motta M, Lanzarotto F, Villanacci V, Amato M, Mora A, Bertolazzi S, Benini F, Ricci C. Circulating ghrelin level is increased in coeliac disease as in functional dyspepsia and reverts to normal during gluten-free diet. Aliment Pharmacol Ther. 2006;23:907-913. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 32] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 49. | Kazemi M, Eshraghian A, Hamidpour L, Taghavi S. Changes in serum ghrelin level in relation to meal-time in patients with functional dyspepsia. United European Gastroenterol J. 2015;3:11-16. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 50. | Lee KJ, Cha DY, Cheon SJ, Yeo M, Cho SW. Plasma ghrelin levels and their relationship with gastric emptying in patients with dysmotility-like functional dyspepsia. Digestion. 2009;80:58-63. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 44] [Cited by in F6Publishing: 47] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 51. | Takamori K, Mizuta Y, Takeshima F, Akazawa Y, Isomoto H, Ohnita K, Ohba K, Omagari K, Shikuwa S, Kohno S. Relation among plasma ghrelin level, gastric emptying, and psychologic condition in patients with functional dyspepsia. J Clin Gastroenterol. 2007;41:477-483. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 41] [Cited by in F6Publishing: 38] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 52. | Ochi M, Tominaga K, Tanaka F, Tanigawa T, Shiba M, Watanabe T, Fujiwara Y, Oshitani N, Higuchi K, Arakawa T. Effect of chronic stress on gastric emptying and plasma ghrelin levels in rats. Life Sci. 2008;82:862-868. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 98] [Cited by in F6Publishing: 99] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 53. | Greenwood-Van Meerveld B, Kriegsman M, Nelson R. Ghrelin as a target for gastrointestinal motility disorders. Peptides. 2011;32:2352-2356. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 54. | Shinomiya T, Fukunaga M, Akamizu T, Irako T, Yokode M, Kangawa K, Nakai Y, Nakai Y. Plasma acylated ghrelin levels correlate with subjective symptoms of functional dyspepsia in female patients. Scand J Gastroenterol. 2005;40:648-653. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 54] [Cited by in F6Publishing: 56] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 55. | Kim YS, Lee JS, Lee TH, Cho JY, Kim JO, Kim WJ, Kim HG, Jeon SR, Jeong HS. Plasma levels of acylated ghrelin in patients with functional dyspepsia. World J Gastroenterol. 2012;18:2231-2237. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 12] [Cited by in F6Publishing: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 56. | Choi YJ, Park YS, Kim N, Kim YS, Lee SM, Lee DH, Jung HC. Gender differences in ghrelin, nociception genes, psychological factors and quality of life in functional dyspepsia. World J Gastroenterol. 2017;23:8053-8061. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 24] [Cited by in F6Publishing: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 57. | Shindo T, Futagami S, Hiratsuka T, Horie A, Hamamoto T, Ueki N, Kusunoki M, Miyake K, Gudis K, Tsukui T, Iwakiri K, Sakamoto C. Comparison of gastric emptying and plasma ghrelin levels in patients with functional dyspepsia and non-erosive reflux disease. Digestion. 2009;79:65-72. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 94] [Cited by in F6Publishing: 91] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 58. | Yamawaki H, Futagami S, Wakabayashi M, Sakasegawa N, Agawa S, Higuchi K, Kodaka Y, Iwakiri K. Management of functional dyspepsia: state of the art and emerging therapies. Ther Adv Chronic Dis. 2018;9:23-32. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 28] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 59. | Choi YJ, Kim N, Yoon H, Shin CM, Park YS, Park JH, Nam RH, Lee DH, Jung HC. Increase in plasma acyl ghrelin levels is associated with abatement of dyspepsia following Helicobacter pylori eradication. J Gastroenterol. 2016;51:548-559. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 60. | Nweneka CV, Prentice AM. Helicobacter pylori infection and circulating ghrelin levels - a systematic review. BMC Gastroenterol. 2011;11:7. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 54] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 61. | Kim SH, Kim JW, Byun J, Jeong JB, Kim BG, Lee KL. Plasma ghrelin level and plasma ghrelin/obestatin ratio are related to intestinal metaplasia in elderly patients with functional dyspepsia. PLoS One. 2017;12:e0175231. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 62. | Ando T, Ichimaru Y, Konjiki F, Shoji M, Komaki G. Variations in the preproghrelin gene correlate with higher body mass index, fat mass, and body dissatisfaction in young Japanese women. Am J Clin Nutr. 2007;86:25-32. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 39] [Cited by in F6Publishing: 41] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 63. | Futagami S, Shimpuku M, Kawagoe T, Izumi N, Ohishi N, Yamawaki H, Shindo T, Nagoya H, Horie A, Kodaka Y, Gudis K, Itoh T, Sakamoto C. The preproghrelin 3056 TT genotype is associated with the feeling of hunger and low acylated ghrelin levels in Japanese patients with Helicobacter pylori-negative functional dyspepsia. Intern Med. 2013;52:1155-1163. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 64. | Yamawaki H, Futagami S, Shimpuku M, Shindo T, Maruki Y, Nagoya H, Kodaka Y, Sato H, Gudis K, Kawagoe T, Sakamoto C. Leu72Met408 Polymorphism of the Ghrelin Gene Is Associated With Early Phase of Gastric Emptying in the Patients With Functional Dyspepsia in Japan. J Neurogastroenterol Motil. 2015;21:93-102. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 65. | Lee KJ, Cho SW. M2019 Polymorphisms of Ghrelin Preprohormone Gene are Associated with the Susceptibility to Postprandial Distress Syndrome and Irritable Bowel Syndrome with Diarrhea. Gastroenterology. 2010;138:S-460. [DOI] [Cited in This Article: ] |
| 66. | Zatorski H, Mosinska P, Storr M, Fichna J. Relamorelin and other ghrelin receptor agonists - future options for gastroparesis, functional dyspepsia and proton pump inhibitors-resistant non-erosive reflux disease. J Physiol Pharmacol. 2017;68:797-805. [PubMed] [Cited in This Article: ] |
| 67. | Tack J, Camilleri M. New developments in the treatment of gastroparesis and functional dyspepsia. Curr Opin Pharmacol. 2018;43:111-117. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 61] [Cited by in F6Publishing: 66] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 68. | Togawa K, Matsuzaki J, Kobayakawa M, Fukushima Y, Suzaki F, Kasugai K, Nishizawa T, Naito Y, Hayakawa T, Kamiya T, Andoh T, Yoshida H, Tokura Y, Nagata H, Mori M, Kato K, Hosoda H, Takebayashi T, Miura S, Uemura N, Joh T, Hibi T, Suzuki H. Association of baseline plasma des-acyl ghrelin level with the response to rikkunshito in patients with functional dyspepsia. J Gastroenterol Hepatol. 2016;31:334-341. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 69. | Suzuki H, Matsuzaki J, Fukushima Y, Suzaki F, Kasugai K, Nishizawa T, Naito Y, Hayakawa T, Kamiya T, Andoh T, Yoshida H, Tokura Y, Nagata H, Kobayakawa M, Mori M, Kato K, Hosoda H, Takebayashi T, Miura S, Uemura N, Joh T, Hibi T, Tack J; Rikkunshito study group. Randomized clinical trial: rikkunshito in the treatment of functional dyspepsia--a multicenter, double-blind, randomized, placebo-controlled study. Neurogastroenterol Motil. 2014;26:950-961. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 95] [Cited by in F6Publishing: 93] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 70. | Mearin F, Lacy BE, Chang L, Chey WD, Lembo AJ, Simren M, Spiller R. Bowel Disorders. Gastroenterology. 2016; Epub ahead of print. [PubMed] [Cited in This Article: ] |
| 71. | El-Salhy M. Irritable bowel syndrome: diagnosis and pathogenesis. World J Gastroenterol. 2012;18:5151-5163. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 77] [Cited by in F6Publishing: 109] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 72. | Posserud I, Ersryd A, Simrén M. Functional findings in irritable bowel syndrome. World J Gastroenterol. 2006;12:2830-2838. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 46] [Cited by in F6Publishing: 47] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 73. | Clarke G, Quigley EM, Cryan JF, Dinan TG. Irritable bowel syndrome: towards biomarker identification. Trends Mol Med. 2009;15:478-489. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 125] [Cited by in F6Publishing: 147] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 74. | Chey WD, Kurlander J, Eswaran S. Irritable bowel syndrome: a clinical review. JAMA. 2015;313:949-958. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 629] [Cited by in F6Publishing: 637] [Article Influence: 70.8] [Reference Citation Analysis (0)] |
| 75. | Karantanos T, Markoutsaki T, Gazouli M, Anagnou NP, Karamanolis DG. Current insights in to the pathophysiology of Irritable Bowel Syndrome. Gut Pathog. 2010;2:3. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 53] [Cited by in F6Publishing: 55] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 76. | Whorwell PJ, McCallum M, Creed FH, Roberts CT. Non-colonic features of irritable bowel syndrome. Gut. 1986;27:37-40. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 355] [Cited by in F6Publishing: 381] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 77. | El-Salhy M, Lillebø E, Reinemo A, Salmelid L. Ghrelin in patients with irritable bowel syndrome. Int J Mol Med. 2009;23:703-707. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 39] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 78. | Sjölund K, Ekman R, Wierup N. Covariation of plasma ghrelin and motilin in irritable bowel syndrome. Peptides. 2010;31:1109-1112. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 24] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 79. | Şahin-Eryılmaz G, Başak K, Çakır-Madenci Ö, Koç H, Tüzün S, Dolapçıoğlu C, Ahıshalı E, Dabak MR. Relationship between irritable bowel syndrome and plasma and tissue ghrelin levels. Turk J Gastroenterol. 2018;29:631-635. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5] [Cited by in F6Publishing: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 80. | Kim YS, Kim N. Sex-Gender Differences in Irritable Bowel Syndrome. J Neurogastroenterol Motil. 2018;24:544-558. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 100] [Cited by in F6Publishing: 131] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 81. | Videlock EJ, Mahurkar-Joshi S, Iliopoulos D, Pothoulakis C, Mayer EA, Chang L. Dysregulation of the Long-Noncoding RNA, Ghrlos, in Irritable Bowel Syndrome. Gastroenterology. 2017;152:S722. [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 82. | Holliday R. Epigenetics: a historical overview. Epigenetics. 2006;1:76-80. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 437] [Cited by in F6Publishing: 356] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 83. | Bhandari S, Jha P, Thakur A, Kar A, Gerdes H, Venkatesan T. Cyclic vomiting syndrome: epidemiology, diagnosis, and treatment. Clin Auton Res. 2018;28:203-209. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 84. | Hejazi RA, Lavenbarg TH, McCallum RW. Elevated serum ghrelin levels in adult patients with cyclic vomiting syndrome. Am J Gastroenterol. 2011;106:1858-1859. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7] [Cited by in F6Publishing: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 85. | Miller AR, Barr RG. Infantile colic. Is it a gut issue? Pediatr Clin North Am. 1991;38:1407-1423. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 66] [Cited by in F6Publishing: 66] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 86. | Savino F, Grassino EC, Guidi C, Oggero R, Silvestro L, Miniero R. Ghrelin and motilin concentration in colicky infants. Acta Paediatr. 2006;95:738-741. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 87. | Chen CY, Asakawa A, Fujimiya M, Lee SD, Inui A. Ghrelin gene products and the regulation of food intake and gut motility. Pharmacol Rev. 2009;61:430-481. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 159] [Cited by in F6Publishing: 161] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 88. | Thompson NM, Gill DA, Davies R, Loveridge N, Houston PA, Robinson IC, Wells T. Ghrelin and des-octanoyl ghrelin promote adipogenesis directly in vivo by a mechanism independent of the type 1a growth hormone secretagogue receptor. Endocrinology. 2004;145:234-242. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 299] [Cited by in F6Publishing: 313] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 89. | Bedendi I, Alloatti G, Marcantoni A, Malan D, Catapano F, Ghé C, Deghenghi R, Ghigo E, Muccioli G. Cardiac effects of ghrelin and its endogenous derivatives des-octanoyl ghrelin and des-Gln14-ghrelin. Eur J Pharmacol. 2003;476:87-95. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 127] [Cited by in F6Publishing: 131] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 90. | Tsubota Y, Owada-Makabe K, Yukawa K, Maeda M. Hypotensive effect of des-acyl ghrelin at nucleus tractus solitarii of rat. Neuroreport. 2005;16:163-166. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 91. | Broglio F, Gottero C, Prodam F, Gauna C, Muccioli G, Papotti M, Abribat T, Van Der Lely AJ, Ghigo E. Non-acylated ghrelin counteracts the metabolic but not the neuroendocrine response to acylated ghrelin in humans. J Clin Endocrinol Metab. 2004;89:3062-3065. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 241] [Cited by in F6Publishing: 243] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 92. | Gauna C, Meyler FM, Janssen JA, Delhanty PJ, Abribat T, van Koetsveld P, Hofland LJ, Broglio F, Ghigo E, van der Lely AJ. Administration of acylated ghrelin reduces insulin sensitivity, whereas the combination of acylated plus unacylated ghrelin strongly improves insulin sensitivity. J Clin Endocrinol Metab. 2004;89:5035-5042. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 179] [Cited by in F6Publishing: 185] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 93. | Reano S, Graziani A, Filigheddu N. Acylated and unacylated ghrelin administration to blunt muscle wasting. Curr Opin Clin Nutr Metab Care. 2014;17:236-240. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 26] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 94. | Asakawa A, Inui A, Fujimiya M, Sakamaki R, Shinfuku N, Ueta Y, Meguid MM, Kasuga M. Stomach regulates energy balance via acylated ghrelin and desacyl ghrelin. Gut. 2005;54:18-24. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 350] [Cited by in F6Publishing: 362] [Article Influence: 19.1] [Reference Citation Analysis (0)] |
| 95. | Chen CY, Inui A, Asakawa A, Fujino K, Kato I, Chen CC, Ueno N, Fujimiya M. Des-acyl ghrelin acts by CRF type 2 receptors to disrupt fasted stomach motility in conscious rats. Gastroenterology. 2005;129:8-25. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 175] [Cited by in F6Publishing: 182] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 96. | Yagi T, Asakawa A, Ueda H, Miyawaki S, Inui A. The role of ghrelin in patients with functional dyspepsia and its potential clinical relevance (Review). Int J Mol Med. 2013;32:523-531. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |