Published online Oct 21, 2009. doi: 10.3748/wjg.15.4980
Revised: September 11, 2009
Accepted: September 18, 2009
Published online: October 21, 2009
Cystic Brunner’s gland hamartoma in the duodenum is exceedingly rare, although microscopic examination may sometimes reveal a Brunner’s gland hamartoma containing dilated ducts in the duodenum. We present a case of large cystic Brunner’s gland hamartoma in the duodenum with a long stalk, which is described in light of multidetector-row computed tomography, magnetic resonance imaging, and a modified small bowel series, together with pathologic correlation and differential diagnosis.
- Citation: Park BJ, Kim MJ, Lee JH, Park SS, Sung DJ, Cho SB. Cystic Brunner’s gland hamartoma in the duodenum: A case report. World J Gastroenterol 2009; 15(39): 4980-4983
- URL: https://www.wjgnet.com/1007-9327/full/v15/i39/4980.htm
- DOI: https://dx.doi.org/10.3748/wjg.15.4980
Brunner’s gland hamartoma is an infrequently encountered benign tumor in the proximal duodenum, accounting for approximately 5% of all duodenal masses[1,2]. Fifty-seven percent of the tumors originate from the duodenal bulb and the incidence of the tumor decreases as the distance from the pyloric ring grows[3].
Cystic lesions of the Brunner’s glands are exceedingly rare. Only 12 cases have been documented in the literature under various terms such as Brunner’s gland cyst, cystadenoma, mucocele, and cystic Brunner’s gland hamartoma[4]. There has been no literature depicting predominant cystic lesions of Brunner’s gland hamartoma in imaging modalities, except for two cases that were found at endoscopic ultrasonography. Although mild dilatation of the ducts may be present in Brunner’s gland lesions, it is only detected in histologic test[5,6].
Herein, we introduce a rare cystic Brunner’s gland hamartoma with a long stalk arising from the duodenal third portion found on multidetector-row computed tomography (MDCT), magnetic resonance imaging (MRI), and a modified small bowel series, together with pathologic correlation.
A 30-year-old man with a 3-d history of aggravated episodes of nausea, vomiting, and epigastric pain was referred by a private clinic. He was healthy with no specific family or past medical history. Physical examination and laboratory evaluation revealed no abnormalities.
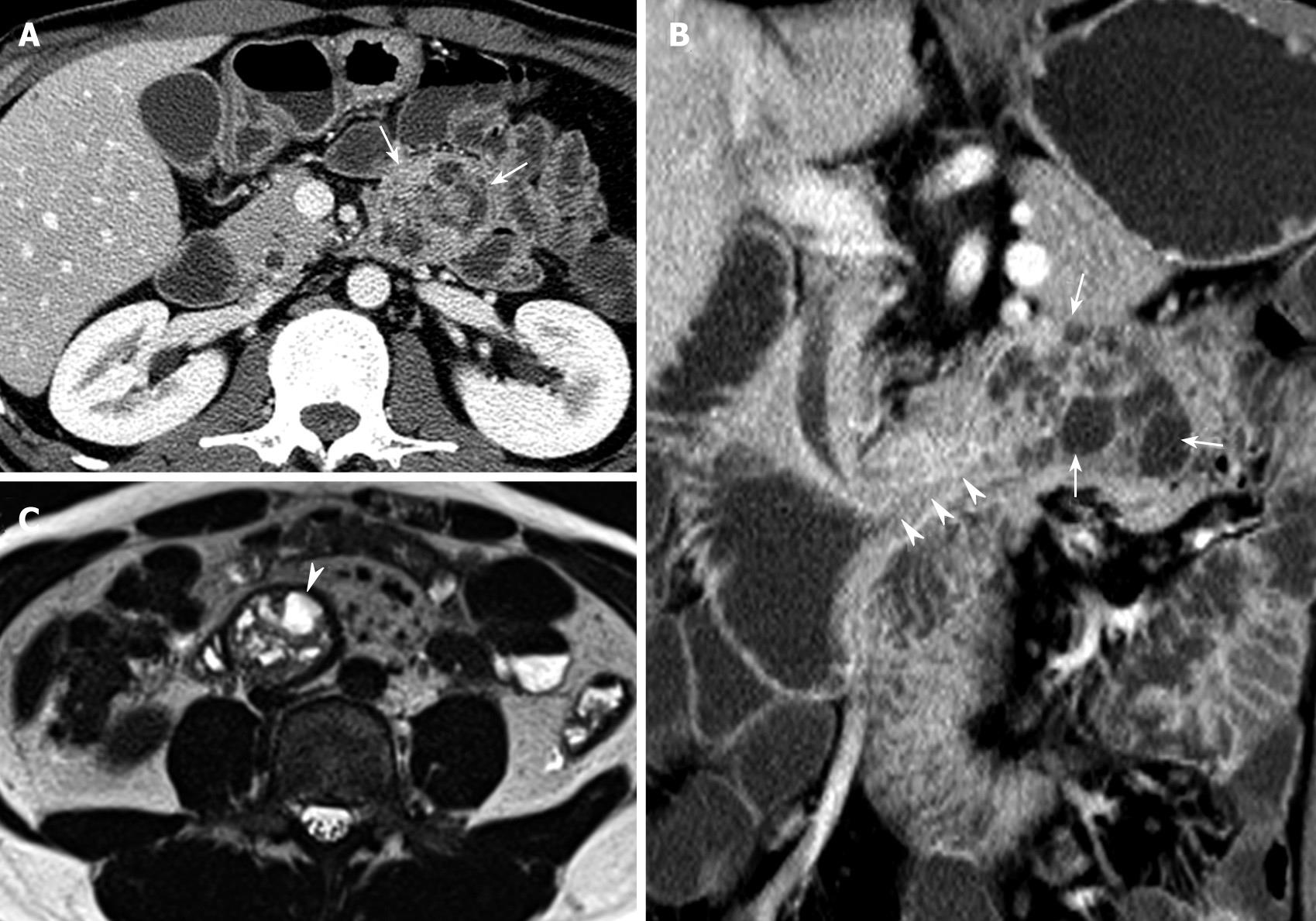
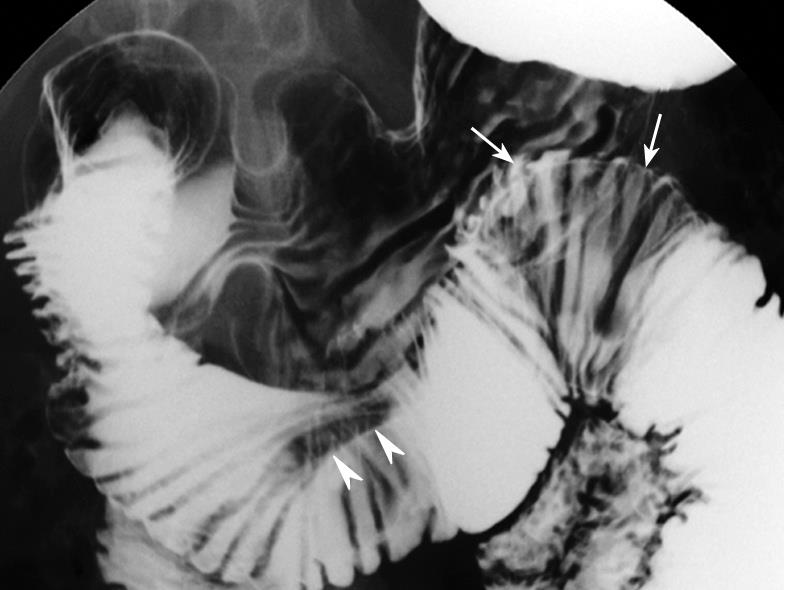
Upper tract endoscopy was not performed because endoscopic results reported by the private clinic were within the normal limits. MDCT showed a 4 cm × 4 cm polypoid mass with multifocal internal low densities in the proximal jejunum just below the Treitz ligament (Figure 1A), and the lesion’s long stalk arising from the third portion of the duodenum (Figure 1B). Multifocal high-signal intensities on T2-weighted MRI reflected cystic components (Figure 1C), which were identical to the low densities on MDCT (Figure 1A and B). The lesion’s location moved to the third portion of the duodenum on MRI (Figure 1C). On the modified small bowel series, a mobile smooth lobular filling defect with a long stalk was visible in the proximal jejunum (Figure 2).
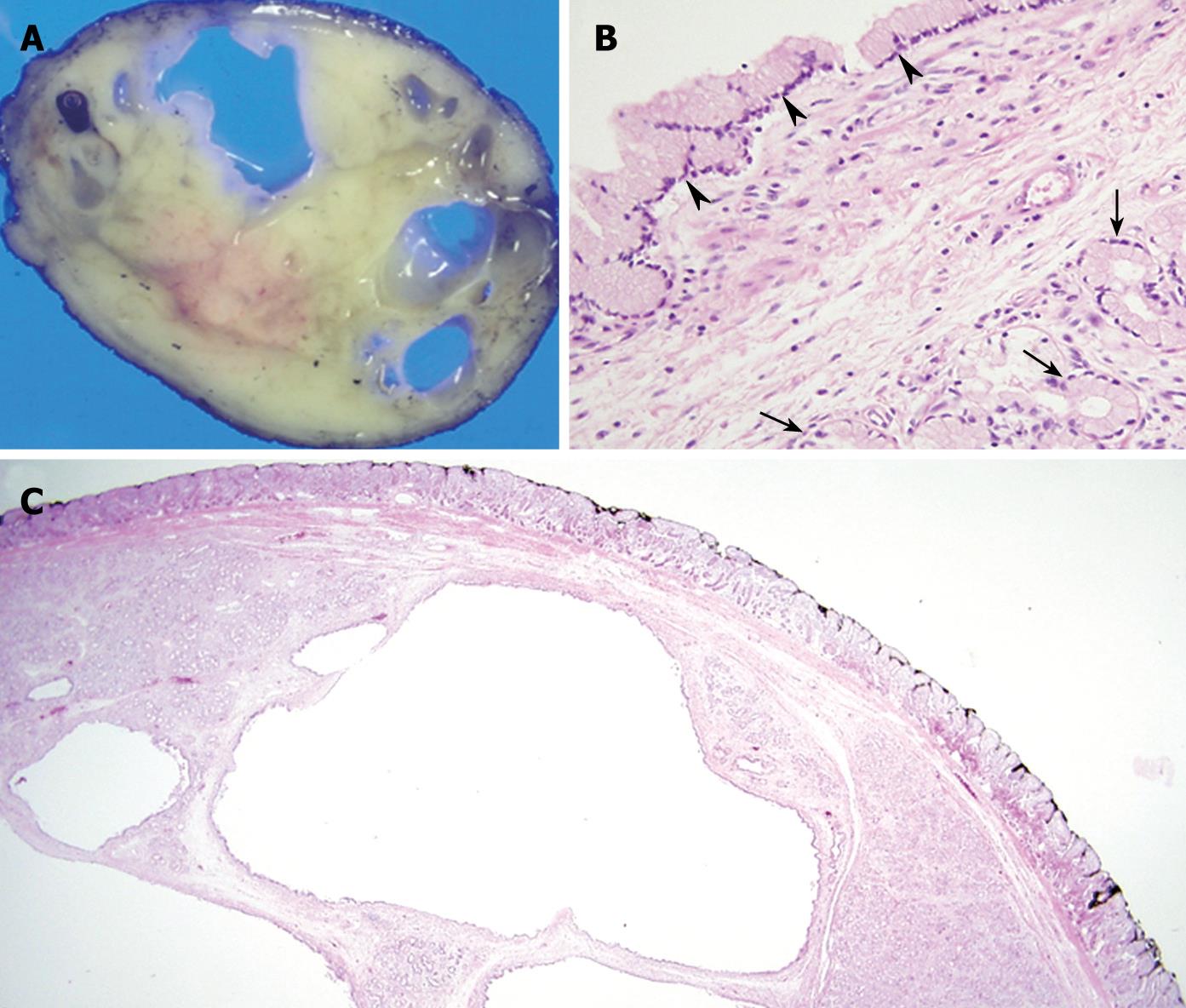
A pedunculated, well circumscribed mass was removed at laparoscopic enterotomy of the proximal jejunum, and the residual long remnant stalk was removed by post-operative endoscopy. The cut surface of the resected specimen revealed an approximately 4 cm × 3 cm whitish ovoid mass with multiple internal cystic spaces (Figure 3A). Histological examination of the specimen found that the main body of the polyp contained a lobular collection of mature Brunner’s glands with large multifocal areas of glandular cystic dilation (Figure 3B and C). There was no evidence of malignancy, and the lesion was diagnosed as a pedunculated cystic Brunner’s gland hamartoma.
The etiology of Brunner’s gland hamartoma has not been clearly elucidated. Brunner’s gland hamartoma represents 5%-10% of all small bowel tumors and occurs most commonly in the fifth and sixth decades of life, with no gender or race predominance[7]. Brunner’s gland hamartoma is most commonly located on the posterior wall of the duodenum near the junction of the first and second portions. In a series of 27 patients described by Levine et al[8], Brunner’s gland hamartoma was located in the duodenal bulb, the second and third portions of the duodenum of 70%, 26%, and 4% of the cases, respectively. In the present case, the long stalk base was located in the proximal third portion of the duodenum.
Although Brunner’s gland hamartoma is incidentally found, Levine et al[8] reported that the most common presentations in symptomatic patients are gastrointestinal hemorrhage (37%) and obstructive symptoms (37%). Rarer presentations have been found in conjunction with recurrent pancreatitis[9], obstructive jaundice[10], and biliary fistula[11]. Brunner’s gland hamartoma is usually non-dysplastic and entirely benign. However, at least one recent report is available on a Brunner’s gland hamartoma with malignant potential[12]. Brunner’s gland hamartoma of the duodenum can be removed endoscopically because it is usually benign. However, we performed laparoscopic enterotomy because endoscopic results reported by the private clinic were within the normal limits. It has been suggested that endoscopic or surgical removal of Brunner’s gland hamartoma can prevent the development of complications and endoscopic polypectomy represents the ideal approach depending on the site and size of Brunner’s gland hamartoma and the presence of a peduncle[13]. In the present case, considering its size and distal location, the endoscopist was reluctant to perform the procedure, hence laparoscopic enterotomy was performed as our next treatment option.
Patel et al[14] have reported seven cases of Brunner’s gland hamartoma on CT. In that report, two lesions with homogenous enhancement correlated with histologic findings of solid glandular proliferation, and the remainder were found with heterogeneous enhancement and hypoattenuation. The latter cases revealed smooth muscle proliferation and small cystic dilatation of the Brunner’s glands. Up to now, to our knowledge, no study has described predominant cystic lesions of Brunner’s gland hamartoma found on imaging modalities such as MDCT and MRI, except for two cases that were found on endoscopic ultrasonography[5,6]. Endoscopic ultrasonography provides additional information about the internal components of the duodenal mass. Imaging modalities such as MDCT, MRI, and modified small bowel series were useful for tissue characterization and localization of the mass in our case, because the large pedunculated mass of the jejunum was difficult to approach by endoscopic ultrasonography.
In the present case, large cyst lesions in a pedunculated mass were revealed by characteristic multifocal high-signal intensities on T2-weighted MRI images, and large hypoattenuating areas on enhanced MDCT correlated with large cystic dilatation of the glands. Mobility due to its long stalk was confirmed by a modified small bowel series and positional change during serial imaging studies.
Diagnosis of Brunner’s gland hamartoma is not always easy at present because radiological findings are often nonspecific[15]. CT is useful only for confirming the absence of extra-luminal extension of a tumor[16]. Radiologic findings of Brunner’s gland hamartoma are extremely similar to those of several other lesions, including adenomatous polyps, lymphoma, carcinoid tumors, and mesenchymal tumors such as gastrointestinal stromal tumor, leiomyoma, and neurogenic tumor. Since duodenal tumors depicted in a cystic configuration through radiologic modalities are rare, the recognition of a cystic component in a solitary duodenal mass may be useful for preoperative diagnosis of Brunner’s gland hamartoma.
In conclusion, cystic configuration of a solitary duodenal mass should be generally considered the clue to the diagnosis of Brunner’s gland hamartoma.
Peer reviewers: Dr. Serdar Karakose, Professor, Department of Radiology, Meram Medical Faculty, Selcuk University, Konya 42080, Turkey; Kazuo Chijiiwa, Profesor, Department of Surgery 1, Miyazaki University School of Medicine, Miyazaki, 889-1692, Japan
S- Editor Wang YR L- Editor Wang XL E- Editor Ma WH
| 1. | Botsford TW, Crowe P, Crocker DW. Tumors of the small intestine. A review of experience with 115 cases including a report of a rare case of malignant hemangio-endothelioma. Am J Surg. 1962;103:358-365. [Cited in This Article: ] |
| 2. | Ghazi A, Ferstenberg H, Shinya H. Endoscopic gastroduodenal polypectomy. Ann Surg. 1984;200:175-180. [Cited in This Article: ] |
| 3. | Silverman L, Waugh JM, Huizenga KA, Harrison EG, Jr . Large adenomatous polyp of Brunner's glands. Am J Clin Pathol. 1961;36:438-443. [Cited in This Article: ] |
| 4. | Silverman L, Waugh JM, Huizenga KA, Harrison EG Jr. Large adenomatous polyp of Brunner’s glands. Am J Clin Pathol. 1961;36:438-443. [Cited in This Article: ] |
| 5. | Yamakawa M, Murata I, Yamao T, Kawai K, Kohno S. Cystic Brunner's gland hamartoma. Gastrointest Endosc. 2003;57:919. [Cited in This Article: ] |
| 6. | Changchien CS, Hsu CC, Hu TH. Endosonographic appearances of Brunner's gland hamartomas. J Clin Ultrasound. 2001;29:243-246. [Cited in This Article: ] |
| 7. | Peison B, Benisch B. Brunner's gland adenoma of the duodenal bulb. Am J Gastroenterol. 1982;77:276-278. [Cited in This Article: ] |
| 8. | Levine JA, Burgart LJ, Batts KP, Wang KK. Brunner's gland hamartomas: clinical presentation and pathological features of 27 cases. Am J Gastroenterol. 1995;90:290-294. [Cited in This Article: ] |
| 9. | Scholz HG. [Recurrent acute pancreatitis, a complication of brunneromas (authors transl)]. Leber Magen Darm. 1976;6:300-302. [Cited in This Article: ] |
| 10. | Skellenger ME, Kinner BM, Jordan PH Jr. Brunner's gland hamartomas can mimic carcinoma of the head of the pancreas. Surg Gynecol Obstet. 1983;156:774-776. [Cited in This Article: ] |
| 11. | Hedges AR. Hamartoma of Brunner's gland causing pyloric obstruction and a biliary fistula. Case report. Acta Chir Scand. 1988;154:475-476. [Cited in This Article: ] |
| 12. | Brookes MJ, Manjunatha S, Allen CA, Cox M. Malignant potential in a Brunner’s gland hamartoma. Postgrad Med J. 2003;79:416-417. [Cited in This Article: ] |
| 13. | Rocco A, Borriello P, Compare D, De Colibus P, Pica L, Iacono A, Nardone G. Large Brunner's gland adenoma: case report and literature review. World J Gastroenterol. 2006;12:1966-1968. [Cited in This Article: ] |
| 14. | Patel ND, Levy AD, Mehrotra AK, Sobin LH. Brunner's gland hyperplasia and hamartoma: imaging features with clinicopathologic correlation. AJR Am J Roentgenol. 2006;187:715-722. [Cited in This Article: ] |
| 15. | Gao YP, Zhu JS, Zheng WJ. Brunner's gland adenoma of duodenum: a case report and literature review. World J Gastroenterol. 2004;10:2616-2617. [Cited in This Article: ] |
| 16. | Merine D, Jones B, Ghahremani GG, Hamilton SR, Bayless TM. Hyperplasia of Brunner glands: the spectrum of its radiographic manifestations. Gastrointest Radiol. 1991;16:104-108. [Cited in This Article: ] |