Published online Jan 21, 2013. doi: 10.3748/wjg.v19.i3.347
Revised: December 12, 2012
Accepted: December 20, 2012
Published online: January 21, 2013
AIM: To investigate the epidemiological trends in incidence and mortality of perforated peptic ulcer (PPU) in a well-defined Norwegian population.
METHODS: A retrospective, population-based, single-center, consecutive cohort study of all patients diagnosed with benign perforated peptic ulcer. Included were both gastric and duodenal ulcer patients admitted to Stavanger University Hospital between January 2001 and December 2010. Ulcers with a malignant neoplasia diagnosis, verified by histology after biopsy or resection, were excluded. Patients were identified from the hospitals administrative electronic database using pertinent ICD-9 and ICD-10 codes (K25.1, K25.2, K25.5, K25.6, K26.1, K26.2, K26.5, K26.6). Additional searches using appropriate codes for relevant laparoscopic and open surgical procedures (e.g., JDA 60, JDA 61, JDH 70 and JDH 71) were performed to enable a complete identification of all patients. Patient demographics, presentation patterns and clinical data were retrieved from hospital records and surgical notes. Crude and adjusted incidence and mortality rates were estimated by using national population demographics data.
RESULTS: In the study period, a total of 172 patients with PPU were identified. The adjusted incidence rate for the overall 10-year period was 6.5 per 100 000 per year (95%CI: 5.6-7.6) and the adjusted mortality rate for the overall 10-year period was 1.1 per 100 000 per year (95%CI: 0.7-1.6). A non-significant decline in adjusted incidence rate from 9.7 to 5.6 occurred during the decade. The standardized mortality ratio for the whole study period was 5.7 (95%CI: 3.9-8.2), while the total 30-d mortality was 16.3%. No difference in incidence or mortality was found between genders. However, for patients ≥ 60 years, the incidence increased over 10-fold, and mortality more than 50-fold, compared to younger ages. The admission rates outside office hours were high with almost two out of three (63%) admissions seen at evening/night time shifts and/or during weekends. The observed seasonal variations in admissions were not statistically significant.
CONCLUSION: The adjusted incidence rate, seasonal distribution and mortality rate was stable. PPU frequently presents outside regular work-hours. Increase in incidence and mortality occurs with older age.
- Citation: Thorsen K, Søreide JA, Kvaløy JT, Glomsaker T, Søreide K. Epidemiology of perforated peptic ulcer: Age- and gender-adjusted analysis of incidence and mortality. World J Gastroenterol 2013; 19(3): 347-354
- URL: https://www.wjgnet.com/1007-9327/full/v19/i3/347.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i3.347
Each year peptic ulcer disease (PUD) affects 4 million people around the world[1]. Complications are encountered in 10%-20% of these patients and 2%-14% of the ulcers will perforate[2,3]. Perforated peptic ulcer (PPU) is a quite rare, but life threatening disease and the mortality varies from 10%-40%[2,4-6]. Females account for more than half the cases, they are older and have more comorbidity than their male counterparts[6]. Main etiologic factors include use of non-stereoidal anti-inflammatory drugs (NSAIDs), steroids, smoking, Helicobacter pylori (H. pylori) and a diet high in salt[3,7]. All these factors have in common that they affect acid secretion in the gastric mucosa. Defining the exact etiological factor in any given patient may often be difficult, as more than one risk factor may be present and they tend to interact[8]. While previous reports have shown a seasonal variation in the incidence of PPU, others have failed to find such a pattern[9-11].
The incidence rate of PPU has, with some fluctuations, been fairly stable in Northern Europe for decades with reported annual incidence rates of about 4-11 per 100 000 per year[12,13]. Several studies have investigated this up to the beginning of the 21st century, however there is hardly any data on incidence reports from this area the last decade. Due to the high mortality and morbidity rates from PPU, it is of importance to understand the epidemiology to, if possible, enable preventive measures.
The aim of this study was to investigate the recent epidemiological incidence trends and presentation of benign perforated gastroduodenal peptic ulcer in a well-defined Norwegian population.
In this retrospective study all patients diagnosed with benign perforated peptic ulcer (either gastric ulcer or duodenal ulcer) admitted to Stavanger University Hospital (SUH) between January 2001 and December 2010 were included. Ulcers with a malign neoplasia diagnosis, verified by histology after biopsy or resection, were excluded.
SUH serves as the only hospital in the greater Stavanger area and has a primary mixed urban and rural catchment area of about 330 000 (per January, 2012). The “twin cities” of Stavanger and Sandnes are together the third most populated area in Norway, and Stavanger is the densest populated city in Norway.
Patients were identified from the hospitals administrative electronic database using pertinent ICD-9 and ICD-10 codes (K25.1, K25.2, K25.5, K25.6, K26.1, K26.2, K26.5, K26.6). Additional searches using appropriate codes for various surgical procedures (JDA 60 gastroraphy, JDA 61 laparoscopic gastroraphy, JDH 70 duodenoraphy, JDH 71 laparoscopic duodenoraphy) were done to enable a complete identification of all patients. Patient demographics and clinical data were retrieved from hospital records and surgical notes. Crude and adjusted mortality rates were estimated.
Incidence calculations: The crude incidence is the number of new cases of perforated peptic ulcer in our region per 100 000 persons per year. The adjusted incidencerates are the crude rates adjusted for age and gender vs the total Norwegian population using Statistics Norway. The crude death rate is the total number of deaths in the study population per 100 000 persons per year. The adjusted mortality rate is the crude death rate adjusted for age and gender vs the total Norwegian population using Statistics Norway.
The standardized mortality ratio is the number of observed deaths within 30 d of hospital admission in the study group divided by the number of expected deaths in a general population with the same age and sex distribution.
Ulcer localisation was regarded as gastric when present anywhere in the stomach, including prepyloric and pyloric ulcers. Postpyloric ulcers were classified as duodenal. One ulcer located in an anastomosis after a gastrectomy was regarded as a gastric ulcer. The diagnosis and exact localisation was made at operation in most cases, while some were verified at postoperative endoscopy.
Comorbidity was defined as any concomitant disease at the time of admittance for PPU, including cardiovascular disease, pulmonary disease, autoimmune disorders and known or previous cancer disease in patient history.
NSAIDs included acetylic acid as part of the NSAID group in this study.
Seasons were classified as spring (March, April and May); late spring/summer (May, June, July and August); summer (June, July and August); autumn (September, October and November); and winter (December, January and February).
Evening/night time was defined as the hours between 16:30 and 07:30 all days of the week, including Saturday and Sunday.
The study was approved as a quality control assurance according to general guidelines provided by the Regional Ethics Committee Vest (REK Vest).
PASW Statistics 19.0 for Mac (SPSS Inc., Chicago, IL) was used for statistical analysis. Descriptive analyses were performed using χ2 or Fisher’s exact test where appropriate for dichotomous data, and Mann-Whitney U test for continuous data, where applicable. Poisson regression analysis was used to test for differences in crude rates between different periods and between different population groups. Time-trends were also tested by a Poisson time series model. Logistic regression was used to test for association with gender and comorbidity. All tests are two sided and P values < 0.050 were regarded as statistically significant.
A total of 172 patients with a perforated peptic ulcer were identified between 2001 and 2010. Median age was 68 years ranging from 18 to 100 years. There was an equal gender distribution (52% were women), but women were significantly older than men (median age of 73 years vs 62 years, respectively, P < 0.001). The number of women affected increased significantly with age across age groups, with only one in four patients aged < 50 years of age being female, compared to two-thirds of those > 70 years of age being female (26% vs 65% women, respectively, P = 0.002). Further patient characteristics are given in Table 1.
| Duodenal ulcer | Gastric ulcer | Total | P value | |
| n = 60 | n = 112 | n = 172 | ||
| Age | ||||
| < 60 yr | 18 (30) | 37 (33) | 55 (32) | 0.68 |
| ≥ 60 yr | 42 (70) | 75 (67) | 117 (68) | |
| Gender | ||||
| Female | 27 (45) | 62 (55) | 89 (52) | 0.20 |
| Male | 33 (55) | 50 (45) | 83 (48) | |
| ASA status | 0.99 | |||
| 1 | 0 | 0 | 0 | |
| 2 | 2 (3) | 3 (3) | 5 (3) | |
| 3 | 34 (57) | 63 (56) | 97 (56) | |
| 4 | 20 (33) | 39 (35) | 59 (34) | |
| 5 | 4 (7) | 7 (6) | 11 (6) | |
| Social status | 0.32 | |||
| Married | 37 (62) | 58 (52) | 95 (55) | |
| Divorced | 3 (5) | 8 (7) | 11 (6) | |
| Widowed | 9 (15) | 28 (25) | 37 (22) | |
| Single | 6 (10) | 7 (6) | 13 (8) | |
| Unknown | 5 (8) | 11 (10) | 16 (9) | |
| Smokers1 | 26 (54) | 60 (69) | 86 (64) | 0.09 |
| NSAID-use | 26 (43) | 50 (45) | 76 (44) | 0.87 |
| Steroid-use | 5 (8) | 11 (10) | 16 (9) | 0.73 |
| Comorbidity | 45 (75) | 97 (87) | 142 (82) | 0.06 |
| Cardiovascular disease | 27 (45) | 50 (45) | 77 (45) | 0.89 |
| Pulmonary disease | 8 (13) | 19 (17) | 27 (16) | 0.53 |
| Autoimmune disease | 13 (22) | 18 (16) | 31 (18) | 0.36 |
| Malignancy (past or current) | 6 (10) | 23 (21) | 29 (17) | 0.08 |
| Past ulcer history | 8 (13) | 18 (16) | 26 (15) | 0.36 |
| 30-d mortality | 14 (23) | 14 (13) | 28 (16) | 0.07 |
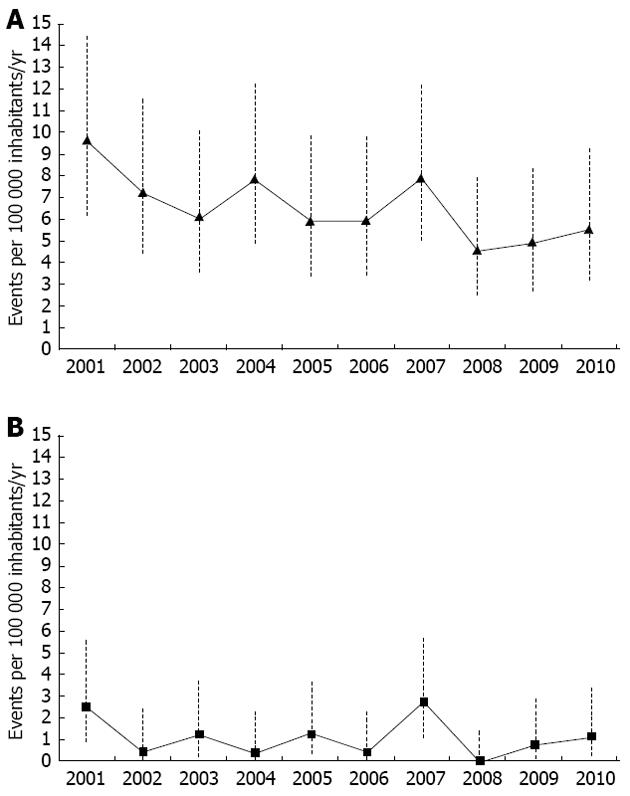
The crude incidence rate of PPU during the study period was 5.6 per 100 000 per year and declined from 8.5 in 2001 to 4.6 in 2010. The adjusted incidence rate for the overall 10-year period was 6.5 per 100 000 per year (95%CI: 5.6-7.6) and declined from 9.7 to 5.6 over the decade, but neither crude nor adjusted incidence rate decline were statistically significant (Figure 1A). Patients ≥ 60 years of age accounted for 117 of 172 (68%), and remained the dominant age group throughout the decade.
When considering the incidence in specific age- and gender-groups, the adjusted incidence rate for men demonstrated considerable differences, with men < 60 years at 2.7 per 100 000 per year (95%CI: 1.9-3.8) compared to 22.1 (95%CI: 16.3-29.4) for men ≥ 60 years of age. A similar pattern was seen in women, with women < 60 years at 1.5 per 100 000 per year (95%CI: 0.9-2.4), while it was 26.1 for those aged ≥ 60 (95%CI: 20.3-32.9) for the whole period. This corresponds to an almost 10-fold increase for men and a corresponding 17-fold increase for women in incidence for those aged ≥ 60 years.
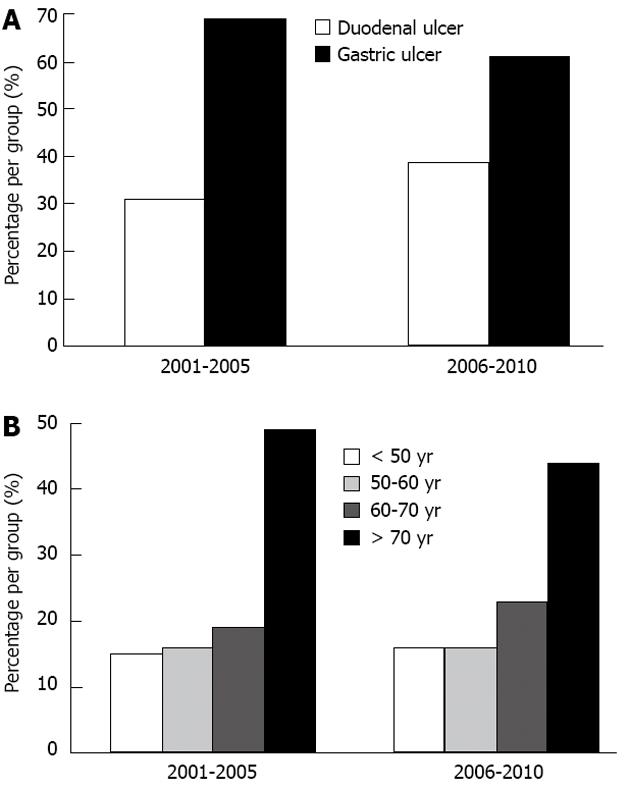
An indicated decline in PPU numbers seen in Figure 1A is evident (but not statistically significant) in those aged > 70 years when the decade was divided in two periods, as illustrated in Figure 2.
The crude mortality rate for the study period was 0.9 and the adjusted mortality rate for the overall 10-year period was 1.1 per 100 000 per year (95%CI: 0.7-1.6) and was stable during the whole period (Figure 1B). The standardized mortality ratio for the whole study period was 5.7 (95%CI: 3.9-8.2), while the total 30-d mortality was 16.3%.
The adjusted mortality rate for men < 60 years was 0.1 per 100 000 per year (95%CI: 0.0-0.43) and 4.6 (95%CI: 2.21-8.48) for men aged ≥ 60 years. For women the adjusted mortality rate for the whole decade for those aged < 60 years was 0.1 (95%CI: 0.0-0.45) while it was 5.6 (95%CI: 3.13-9.22) for those aged ≥ 60 years. This demonstrated negligible differences between genders, but an almost 50 to 60 times increase in mortality among the elderly age group.
Gastric ulcers predominated (Figure 2A) and accounted for 112 of 172 (65%) patients in this study, but declined during the latter years of the period, while the frequency of duodenal ulcers remained stable, but increased somewhat the latter period (Figure 2). Prepyloric ulcers represented 61 of 112 (54%) of gastric ulcers and 21 of 112 (19%) were located in the pylorus. In the corpus/fundus area 12 of 112 (11%) ulcers were observed, while 8 of 112 (7%) were located in the antrum. One ulcer was located in an anastomosis and 9 of 112 (8%) ulcers were missing exact localisation in the stomach, but being classified as gastric ulcers at operation.
Of those aged > 60 years of age 105 of 117 (90%) had comorbidity compared to 37 of 55 (67%) of those aged ≤ 60 years (P < 0.001). Women also had significantly higher rate of comorbidity compared to men (91% vs 74%, respectively, P = 0.002), also when adjusted for age (P = 0.036).
Data on smoking was obtained in 135 of 172 patients. Of the 135 with confirmed information on smoking habits, 86 (64%) were smokers. Consequently, at least 50% of the total 172 patients were smokers, but likely this is an underestimate. Of those aged < 60 years of age 37 of 44 (84%) were smokers, while 49 of 91 (54%) of those aged ≥ 60 were smokers (P = 0.001). The number of smokers registered was stable during the decade.
The number of NSAID users was stable during the decade studied and were used by 47 of 89 women and 29 of 83 men (53% vs 35%, respectively, P = 0.018). Also, NSAID-use was more common in those aged ≥ 60 years (55 of 117; 47%), compared to the younger patients aged < 60 years (20 of 55; 36%), but this was not statistically significant.
Patients were admitted at all hours and days during the week. While 70 of 172 patients (41%) were admitted during weekends (Friday, Saturday and Sunday), more than half (91 of 172; 53%) of the patients were admitted during evening/night time shifts and 109 of 172 (63%) patients were admitted either during evening/night time and/or on Saturday/Sunday. Notably, during this time a lower staff: patient ratio than during regular office hours is present.
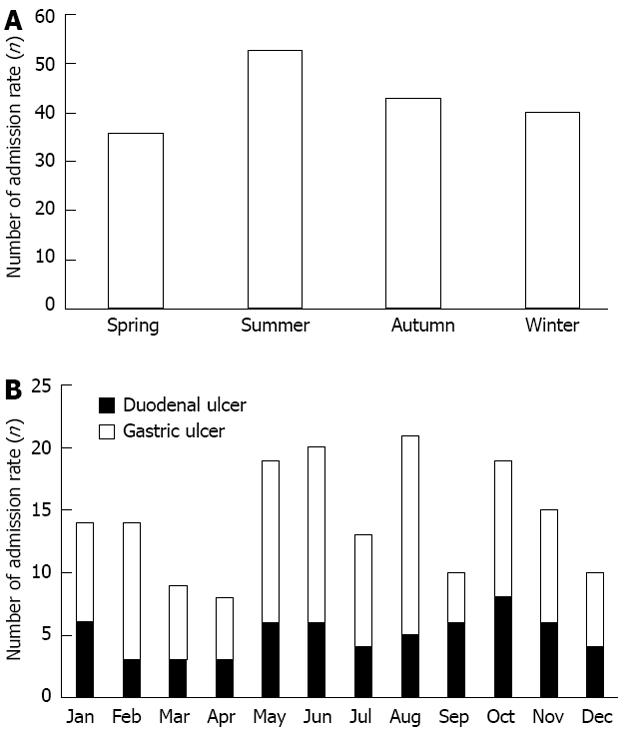
PPU was more frequent in patients ≥ 60 years of age (117 of 172; 68%) than < 60 years of age (55 of 172; 32%). This was particularly evident during the four months of late spring/summer. Poisson regression and time series analyses revealed no statistical significant differences regarding monthly variation or seasonal variation for PPU admittance patterns (Figure 3).
In this single-institution, population based study we found an adjusted incidence rate of 6.5 (95%CI: 5.5-7.5) per 100 000/year during this period. This is in concordance to several Northern European studies, most of them reporting an incidence between 4-11 per 100 000/year[13-17]. Several factors may influence the incidence rates of PPU. For one, epidemiological studies have demonstrated that birth-cohorts born up to the 1930s were at higher risk of acquiring PPU than later birth cohorts[15,18]. The reason for this is not known, but speculation of an overriding influence of H. pylori infection on the population, has been postulated as the main cause[18]. However, H. pylori infection has been deemed of less importance in perforated peptic ulcer disease compared to that of uncomplicated peptic ulcer disease[1,7]. However, the association between specific birth-cohorts and mortality from PUD has been quite convincing, and a decline in the incidence of PPU may thus be expected when the cohorts at risk disappear with time[4,18]. This may have affected the incidence rates also in this study, which are rates comparable to other epidemiological studies from the turn of the century[19], and which may reflect a stable scenario with a relatively low population at risk for PPU in the current century.
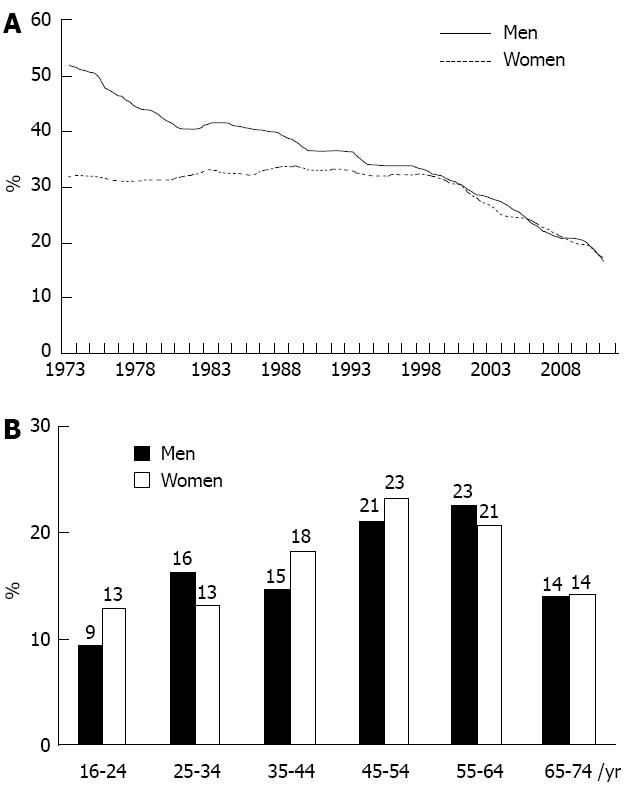
Another etiologic factor for perforated peptic ulcer is smoking[20,21]. Prospective data from SUH between 1987 and 1993 observed that 58 of 104 (56%) of all PPU patients were smokers[22], reflecting the current data. The percentage of smokers in the PPU population may reflect this as a known risk for ulcer disease and perforation. Notably, in 2011 the number of daily smokers in Norway (Figure 4) is reduced to half the number compared to 2001 data[23], and consequently this risk factor has decreased dramatically over just a decade. The decrease in smoking habits is seen in all the age groups from 16-64 years, but most evident in the age group from 16-44 years[23]. As previously stated, PPU is much more common in older than younger people, and the declining number of smokers in the population should influence incidence rates of PPU with time. However, a decade of decline in smoking in the population may not be enough time to reveal this. Also, an increasing age-population and preserved smoking habits in the older may contribute to a stable incidence still.
Medications may influence PPU incidence and occurrence in several ways, both as risk factors and as preventive means. NSAID-use is associated to peptic ulcers and to peptic ulcer complications, however, the majority of PPUs are not related to NSAIDs[24]. In recent years an increase of NSAID-use has been seen and according to numbers from the Norwegian pharmaceutical association: both acetylic acid (14% increase), diclofenac (35% increase) and ibuprofen (26% increase) usage increased from 2006 to 2010 (over the counter sales included), while the usage of the most common PPI (Esomeprazol) was stable[25]. Previous data from Australia has also shown increasing use of NSAIDs in the population without increasing incidence rates of PPU[26]. A study of peptic ulcer complications in the Swedish population over three decades up to 2002, found declining incidence rates after 1988, when PPI were introduced in Sweden. The authors concluded that the reasons for this most likely were multifactorial, but then including an effect of PPI[14]. However, few studies have found an effect of PPI or H2 blockers on the PPU incidence or mortality rates[13].
The overall mortality at 16.3% is in comparison to other contemporary European studies from Denmark, the United Kingdom and the Netherlands ranging from 12% to 27%[27-29], but differs from a recent South Korean study reporting mortality at 3%[30]. The difference may be explained by methodology to retrieve data, as the European studies are hospital-based cohorts, and the South Korean study is based on a national health insurance claims database. In one Danish national cohort[31], the overall mortality was 27%, and reduced to 17% for patients entered in a pre-specified perioperative protocol to reduce mortality[31]. In the current population under study with an overall mortality of 16%, there was no pre-specified protocol in use, but institutional trend towards an increased use of pre-operative computed tomography for diagnosis and laparoscopy as mode of intervention during the study period[6].
Ulcer site definition may be confusing, since some classify prepyloric and pyloric ulcers as duodenal[10], while others classify those ulcers as part of the stomach and hence gastric ulcers[13]. Further, extensive perioperative and inflammatory tissue changes may make it difficult to distinguish between the duodenum and the pyloric area during the operation, hence clinical misclassification can obviously occur. Nevertheless, gastric ulcers predominated in this study and this is in accordance to similar reports from Norway, The Netherlands and Iceland[32-34]. However, a non-significant decline of perforated gastric ulcers was seen in the latter years of the decade, while the frequency of duodenal ulcers was stable. Decreasing incidence of gastric ulcers in those aged > 70 years have also been shown in a large study from England and Wales[17]. We did not identify any associations according to ulcer site, but a trend towards younger men having more duodenal ulcers and older women having more gastric ulcers were seen. This has been shown in similar studies before and may represent a somewhat different aetiology, with older, female patients more often having NSAIDs exposure prior to PPU episodes[17,35]. The latter was also seen in this study.
The off-hour admission rates were high with almost two of three patients admitted at either evening/night time shifts or during weekends (i.e., Saturday/Sunday). Consequently, a high proportion of potentially very sick patients requiring an acute operation will be seen when senior staff may not be immediately available or in-house, which may potentially delay a correct work-up and timely diagnosis and consequently operative intervention, than if admitted at office hours. However, we do not have data to substantiate this potential association in the current study.
Several studies have addressed seasonal variation of perforated peptic ulcer[9,11,36,37]. In this study a peak of perforated peptic ulcers was observed during the four months of late spring/summer, but this pattern faded when seasons where divided in three months. However a previous study from Western Norway over 5.5 decades found similar seasonal variations and those variations were consistent over time[38]. However, no statistical significance was found for variations over time, and this is in line with previous reports that failed to find such patterns[9,10].
The retrospective nature of this study limits the accuracy of data quality, which would have been obtained by a prospective manner for some variables, such as exposure to risk factors (i.e., smoking status; medications used, etc.) which may be subject to bias by underreporting of past history. However, with a population-based catchment area and a well-developed health system of general physicians and a universal health insurance program for all citizens in Norway, patients are usually admitted with admission notes containing past history, although this may obviously be more prone to failure outside office hours and for emergency referrals. Due to restrictions of the number of patients, we are cautious of performing unwarranted subgroup analysis, and may thus not have been able to confirm significant trends found in other studies.
In conclusion, the adjusted incidence rates in the first decade of the 21st century was stable and reflected the decline seen towards the end of the 20th century. Smoking cessation in the general population and a reduction in the population represented by birth cohorts at higher risk are two important factors that may influence the current low incidence of PPU, compared to that in past decades. Perforated peptic ulcer continues to present outside regular work-hours in over half the time and frequently during weekends, with little difference in seasonal distribution. Mortality is unchanged and stable and is most considerable in the aged population. This may be further subject to change with an increasingly elderly population and should be followed by population-based monitoring for this disease over time.
Perforated peptic ulcer (PPU) is a life threatening disease with historically reported high morbidity and mortality rates. Disease epidemiology has changed during the last century, but current data on epidemiological trends in PPU is lacking. The aim of this study is to investigate epidemiological trends in a well-defined Norwegian population over a decade.
Central research in perforated peptic ulcer the latter years has focused on safety of laparoscopy as primary operation and regarding short term mortality after operation. Few studies have presented data regarding epidemiology over the last decade. This study demonstrates a stable incidence and mortality rate over a decade.
A change in peptic ulcer disease epidemiology the last decades came with the discovery of Helicobacter pylori with the subsequent eradication therapy with antibiotica, and the introduction of proton pump inhibitors. For perforated peptic ulcers little has changed except the use of laparoscopy as a surgical alternative for repair. The mortality remains high and the incidence has been stable.
The epidemiology of perforated peptic ulcer appears to be stable with few changes compared to the very recent past. A better understanding of the aetiology may be warranted for better prevention and reduction of incidence. Understanding factors contributing to mortality will be important to further improve outcomes.
The study is an interesting, well designed and well written one. It will help to shed light on the current situation regarding peptic ulcer disease.
P- Reviewer Abdel-Salam OMElS S- Editor Song XX L- Editor A E- Editor Li JY
| 1. | Zelickson MS, Bronder CM, Johnson BL, Camunas JA, Smith DE, Rawlinson D, Von S, Stone HH, Taylor SM. Helicobacter pylori is not the predominant etiology for peptic ulcers requiring operation. Am Surg. 2011;77:1054-1060. [PubMed] [Cited in This Article: ] |
| 2. | Bertleff MJ, Lange JF. Perforated peptic ulcer disease: a review of history and treatment. Dig Surg. 2010;27:161-169. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 131] [Cited by in F6Publishing: 136] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 3. | Lau JY, Sung J, Hill C, Henderson C, Howden CW, Metz DC. Systematic review of the epidemiology of complicated peptic ulcer disease: incidence, recurrence, risk factors and mortality. Digestion. 2011;84:102-113. [PubMed] [Cited in This Article: ] |
| 4. | Svanes C. Trends in perforated peptic ulcer: incidence, etiology, treatment, and prognosis. World J Surg. 2000;24:277-283. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 140] [Cited by in F6Publishing: 153] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 5. | Møller MH, Adamsen S, Wøjdemann M, Møller AM. Perforated peptic ulcer: how to improve outcome? Scand J Gastroenterol. 2009;44:15-22. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 47] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 6. | Thorsen K, Glomsaker TB, von Meer A, Søreide K, Søreide JA. Trends in diagnosis and surgical management of patients with perforated peptic ulcer. J Gastrointest Surg. 2011;15:1329-1335. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 86] [Cited by in F6Publishing: 97] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 7. | Gisbert JP, Legido J, García-Sanz I, Pajares JM. Helicobacter pylori and perforated peptic ulcer prevalence of the infection and role of non-steroidal anti-inflammatory drugs. Dig Liver Dis. 2004;36:116-120. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 39] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 8. | Kurata JH, Nogawa AN. Meta-analysis of risk factors for peptic ulcer. Nonsteroidal antiinflammatory drugs, Helicobacter pylori, and smoking. J Clin Gastroenterol. 1997;24:2-17. [PubMed] [Cited in This Article: ] |
| 9. | Manfredini R, De Giorgio R, Smolensky MH, Boari B, Salmi R, Fabbri D, Contato E, Serra M, Barbara G, Stanghellini V. Seasonal pattern of peptic ulcer hospitalizations: analysis of the hospital discharge data of the Emilia-Romagna region of Italy. BMC Gastroenterol. 2010;10:37. [PubMed] [Cited in This Article: ] |
| 10. | Janik J, Chwirot P. Perforated peptic ulcer--time trends and patterns over 20 years. Med Sci Monit. 2000;6:369-372. [PubMed] [Cited in This Article: ] |
| 11. | Svanes C, Sothern RB, Sørbye H. Rhythmic patterns in incidence of peptic ulcer perforation over 5.5 decades in Norway. Chronobiol Int. 1998;15:241-264. [PubMed] [Cited in This Article: ] |
| 12. | Paimela H, Tuompo PK, Peräkyl T I, Höckerstedt K, Kivilaakso E. Peptic ulcer surgery during the H2-receptor antagonist era: a population-based epidemiological study of ulcer surgery in Helsinki from 1972 to 1987. Br J Surg. 1991;78:28-31. [PubMed] [Cited in This Article: ] |
| 13. | Christensen A, Bousfield R, Christiansen J. Incidence of perforated and bleeding peptic ulcers before and after the introduction of H2-receptor antagonists. Ann Surg. 1988;207:4-6. [PubMed] [Cited in This Article: ] |
| 14. | Hermansson M, Ekedahl A, Ranstam J, Zilling T. Decreasing incidence of peptic ulcer complications after the introduction of the proton pump inhibitors, a study of the Swedish population from 1974-2002. BMC Gastroenterol. 2009;9:25. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 54] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 15. | Svanes C, Lie RT, Kvåle G, Svanes K, Søreide O. Incidence of perforated ulcer in western Norway, 1935-1990: cohort- or period-dependent time trends? Am J Epidemiol. 1995;141:836-844. [PubMed] [Cited in This Article: ] |
| 16. | Paimela H, Oksala NK, Kivilaakso E. Surgery for peptic ulcer today. A study on the incidence, methods and mortality in surgery for peptic ulcer in Finland between 1987 and 1999. Dig Surg. 2004;21:185-191. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 47] [Cited by in F6Publishing: 50] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 17. | Higham J, Kang JY, Majeed A. Recent trends in admissions and mortality due to peptic ulcer in England: increasing frequency of haemorrhage among older subjects. Gut. 2002;50:460-464. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 141] [Cited by in F6Publishing: 161] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 18. | Sonnenberg A. Time trends of ulcer mortality in Europe. Gastroenterology. 2007;132:2320-2327. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 67] [Cited by in F6Publishing: 75] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 19. | Lassen A, Hallas J, Schaffalitzky de Muckadell OB. Complicated and uncomplicated peptic ulcers in a Danish county 1993-2002: a population-based cohort study. Am J Gastroenterol. 2006;101:945-953. [PubMed] [Cited in This Article: ] |
| 20. | Svanes C, Søreide JA, Skarstein A, Fevang BT, Bakke P, Vollset SE, Svanes K, Søreide O. Smoking and ulcer perforation. Gut. 1997;41:177-180. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 39] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 21. | Andersen IB, Jørgensen T, Bonnevie O, Grønbaek M, Sørensen TI. Smoking and alcohol intake as risk factors for bleeding and perforated peptic ulcers: a population-based cohort study. Epidemiology. 2000;11:434-439. [PubMed] [Cited in This Article: ] |
| 22. | Søreide JA, Søndenaa K, Ulvestad E. Helicobacter pylori Status after Treatment for Perforated Gastroduodenal Ulcer: A Cross-Sectional Study Utilizing Serology. Dig Surg. 1997;14:366-370. [Cited in This Article: ] |
| 23. | Available from: http://www.ssb.no (Accessed September 6, 2012). [Cited in This Article: ] |
| 24. | Svanes C, Øvrebø K, Søreide O. Ulcer bleeding and perforation: non-steroidal anti-inflammatory drugs or Helicobacter pylori. Scand J Gastroenterol Suppl. 1996;220:128-131. [PubMed] [Cited in This Article: ] |
| 25. | Available from: http: //www.apotek.no (Accessed July 6, 2012). [Cited in This Article: ] |
| 26. | Henry D, Robertson J. Nonsteroidal anti-inflammatory drugs and peptic ulcer hospitalization rates in New South Wales. Gastroenterology. 1993;104:1083-1091. [PubMed] [Cited in This Article: ] |
| 27. | Møller MH, Engebjerg MC, Adamsen S, Bendix J, Thomsen RW. The Peptic Ulcer Perforation (PULP) score: a predictor of mortality following peptic ulcer perforation. A cohort study. Acta Anaesthesiol Scand. 2012;56:655-662. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 58] [Cited by in F6Publishing: 67] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 28. | Hemmer PH, de Schipper JS, van Etten B, Pierie JP, Bonenkamp JJ, de Graaf PW, Karsten TM. Results of surgery for perforated gastroduodenal ulcers in a Dutch population. Dig Surg. 2011;28:360-366. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 29. | Critchley AC, Phillips AW, Bawa SM, Gallagher PV. Management of perforated peptic ulcer in a district general hospital. Ann R Coll Surg Engl. 2011;93:615-619. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 30. | Bae S, Shim KN, Kim N, Kang JM, Kim DS, Kim KM, Cho YK, Jung SW. Incidence and short-term mortality from perforated peptic ulcer in Korea: a population-based study. J Epidemiol. 2012;22:508-516. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 31. | Møller MH, Adamsen S, Thomsen RW, Møller AM. Multicentre trial of a perioperative protocol to reduce mortality in patients with peptic ulcer perforation. Br J Surg. 2011;98:802-810. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 101] [Cited by in F6Publishing: 94] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 32. | Forsmo HM, Glomsaker T, Vandvik PO. Perforated peptic ulcer--a 12-year material. Tidsskr Nor Laegeforen. 2005;125:1822-1824. [PubMed] [Cited in This Article: ] |
| 33. | Eiriksson K, Oddsdottir M, Magnusson J. Peptic ulcer perforations, University Hospital of Iceland 1989-1995. Laeknabladid. 1998;84:466-473. [PubMed] [Cited in This Article: ] |
| 34. | Bertleff MJ, Halm JA, Bemelman WA, van der Ham AC, van der Harst E, Oei HI, Smulders JF, Steyerberg EW, Lange JF. Randomized clinical trial of laparoscopic versus open repair of the perforated peptic ulcer: the LAMA Trial. World J Surg. 2009;33:1368-1373. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 114] [Cited by in F6Publishing: 125] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 35. | Wysocki A, Budzyński P, Kulawik J, Drożdż W. Changes in the localization of perforated peptic ulcer and its relation to gender and age of the patients throughout the last 45 years. World J Surg. 2011;35:811-816. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 36. | Adler J, Ingram D, House T. Perforated peptic ulcer--a seasonal disease? Aust N Z J Surg. 1984;54:59-61. [PubMed] [Cited in This Article: ] |
| 37. | Christensen A, Hansen CP, Thagaard C, Lanng C. Seasonal periodicity of perforated gastric ulcer. Dan Med Bull. 1988;35:281-282. [PubMed] [Cited in This Article: ] |
| 38. | Svanes C, Salvesen H, Stangeland L, Svanes K, Søreide O. Perforated peptic ulcer over 56 years. Time trends in patients and disease characteristics. Gut. 1993;34:1666-1671. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 57] [Article Influence: 1.8] [Reference Citation Analysis (0)] |