Published online Jul 28, 2013. doi: 10.3748/wjg.v19.i28.4545
Revised: April 24, 2013
Accepted: June 1, 2013
Published online: July 28, 2013
AIM: To investigate the differences in the mutation spectra of the SLC25A13 gene mutations from specific regions of China.
METHODS: Genetic analyses of SLC25A13 mutations were performed in 535 patients with neonatal intrahepatic cholestasis from our center over eight years. Unrelated infants with at least one mutant allele were enrolled to calculate the proportion of SLC25A13 mutations in different regions of China. The boundary between northern and southern China was drawn at the historical border of the Yangtze River.
RESULTS: A total of 63 unrelated patients (about 11% of cases with intrahepatic cholestasis) from 16 provinces or municipalities in China had mutations in the SLC25A13 gene, of these 16 (25%) were homozygotes, 28 (44%) were compound heterozygotes and 19 (30%) were heterozygotes. In addition to four well described common mutations (c.851_854del, c.1638_1660dup23, c.615+5G>A and c.1750+72_1751-4dup17insNM_138459.3:2667 also known as IVS16ins3kb), 13 other mutation types were identified, including three novel mutations: c.985_986insT, c.287T>C and c.1349A>G. According to the geographical division criteria, 60 mutant alleles were identified in patients from the southern areas of China, 43 alleles were identified in patients from the border, and 4 alleles were identified in patients from the northern areas of China. The proportion of four common mutations was higher in south region (56/60, 93%) than that in the border region (34/43, 79%, χ2 = 4.621, P = 0.032) and the northern region (2/4, 50%, χ2 = 8.288, P = 0.041).
CONCLUSION: The SLC25A13 mutation spectra among the three regions of China were different, providing a basis for the improvement of diagnostic strategies and interpretation of genetic diagnosis.
Core tip: Genetic testing of SLC25A13 gene was performed in individuals from southern, border and northern regions of China. The proportion of four common mutations was significant higher in southern region than in the border region and the northern region, so mutation screening for the common 4 mutations an appropriate test in the southern region. In the border and northern region, DNA sequencing is probably more practical.
-
Citation: Chen R, Wang XH, Fu HY, Zhang SR, Abudouxikuer K, Saheki T, Wang JS. Different regional distribution of
SLC25A13 mutations in Chinese patients with neonatal intrahepatic cholestasis. World J Gastroenterol 2013; 19(28): 4545-4551 - URL: https://www.wjgnet.com/1007-9327/full/v19/i28/4545.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i28.4545
Citrin deficiency is estimated to be the most common urea cycle disorder in the world. It is an autosomal recessive disorder which includes adult-onset type II citrullinemia (CTLN2; OMIM #60347)[1,2] and neonatal intrahepatic cholestasis caused by citrin deficiency (NICCD; OMIM #605814)[3-5]. Most NICCD patients show symptoms which ameliorate by 1 year of age[6], but some patients may progress to liver failure and even require liver transplantation during infancy[7-10]. Others may develop CTLN2 more than a decade later[11,12]. Dietary treatment has shown to ameliorate symptoms and may prevent the need for transplant[13,14]. Therefore, prompt diagnosis and appropriate management are important for achieving a favorable long term prognosis for this disease.
Citrin deficiency is caused by mutations in the SLC25A13 gene[1,15]. The protein product of the SLC25A13 gene is citrin, a polypeptide of 675 amino acid residues with a molecular weight of 74 kDa. Citrin contains four EF-hand domains and six mitochondrial transmermbrane (TM)-spanning domains, and resides in the mitochondrial inner membrane[1]. Citrin is expressed in the liver and functions as calcium (Ca2+)-stimulated aspartate-glutamate carrier (AGC) for cytosolic glutamate and protons[16]. Over 60 different functional proved mutations in the human SLC25A13 gene have been identified. These show significant differences in their racial distribution[13,17-20]. In China, the carrier frequency of 4 most common known mutations shows significant regional difference[19,20]. The estimated carries in population are 1/48 in south of the Yangtze River and 1/940 are carries of the river in the North[19].
Currently common mutation screening is used for rapid molecular diagnosis[21]. However, the appropriateness of use in specific populations needs to be established in that population. Therefore, we undertook the present study to investigate the regional distribution of SLC25A13 mutations spectrum in Chinese patients with neonatal intrahepatic cholestasis. Our results will facilitate the design of appropriate screening strategies for this disorder in different regions.
Between June 2003 and December 2011, patients with cholestasis who were referred to the pediatric liver center of Children’s Hospital of Fudan University for conjugated hyperbilirubinemia were enrolled. The inclusion criteria included the onset of conjugated jaundice before 6 mo of age; serum total bilirubin < 5 mg/dL and conjugated bilirubin > 1 mg/dL, or total bilirubin > 5 mg/dL and conjugated bilirubin > 20%[22]. We excluded other diseases that may affect the extrahepatic biliary system, such as biliary atresia, choledochal cyst, tumor, inspissated bile, or hemangioma, by imaging the hepatobiliary system. The imaging procedures included ultrasound scanning and hepatobiliary iminodiacetic acid (HIDA) scintigraphy in each case and laparotomic cholangiography in selected cases. Cases (n = 535) met the inclusion criteria and written informed consent was obtained from their parents.
The study protocol conforms to the ethical guidelines of the Declaration of the Helsinki of 1975 and was approved by the Ethics Committee on human research of the Children’s Hospital of Fudan University.
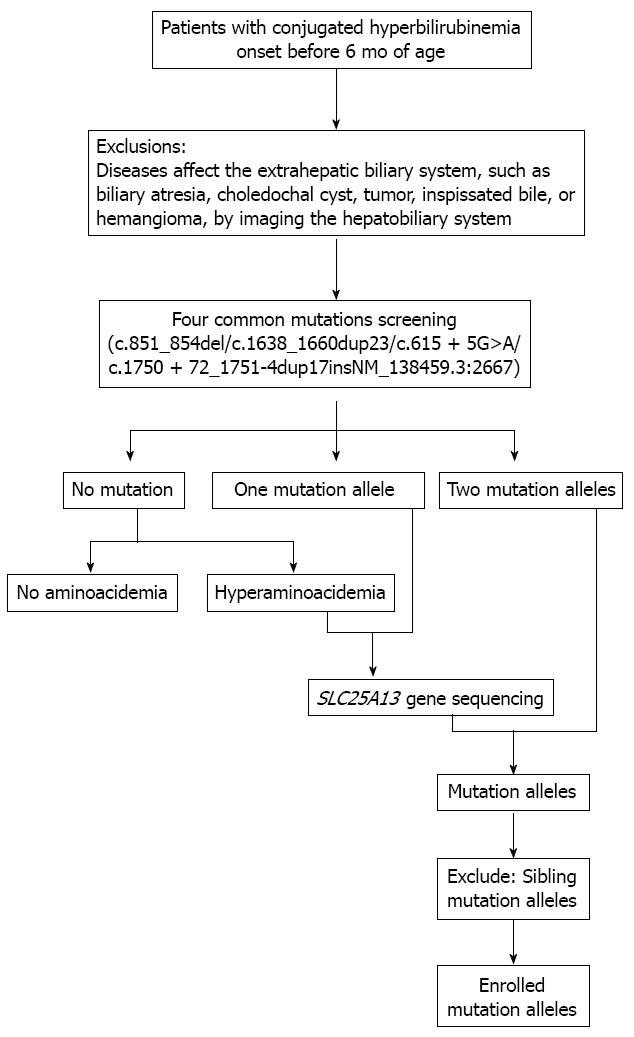
DNA was extracted from peripheral blood samples, which were obtained from each participant and his or her parents using the Tiangen Blood Genomic DNA Isolation Kit according to the manufacturer’s instructions (Tiangen Biotech, Shanghai, China). Four common mutations (c.851_854del, c.1638_1660dup23, c.615+5G>A andc.1750+72_1751-4dup17insNM_138459.3:2667 also known as IVS16ins3kb)[1,20,23] were screened in all subjects. In patients for whom only one mutation was identified by the above screening or who had hyperaminoacidemia were subject to DNA sequencing as described previously[24]. Selection process of patients with mutant allele for analysis is given in Figure 1. The mutation alleles were verified in their parents by the target sequencing to establish segregation. Genomic sequences were obtained at the National Center for Biotechnology Information with RefSeq NM_014251.2 as SLC25A13 reference. Nomenclature of SLC25A13 variants was assigned following the guidelines of Human Genome Variation Society (http://www.hgvs.org/mutnomen)[25].
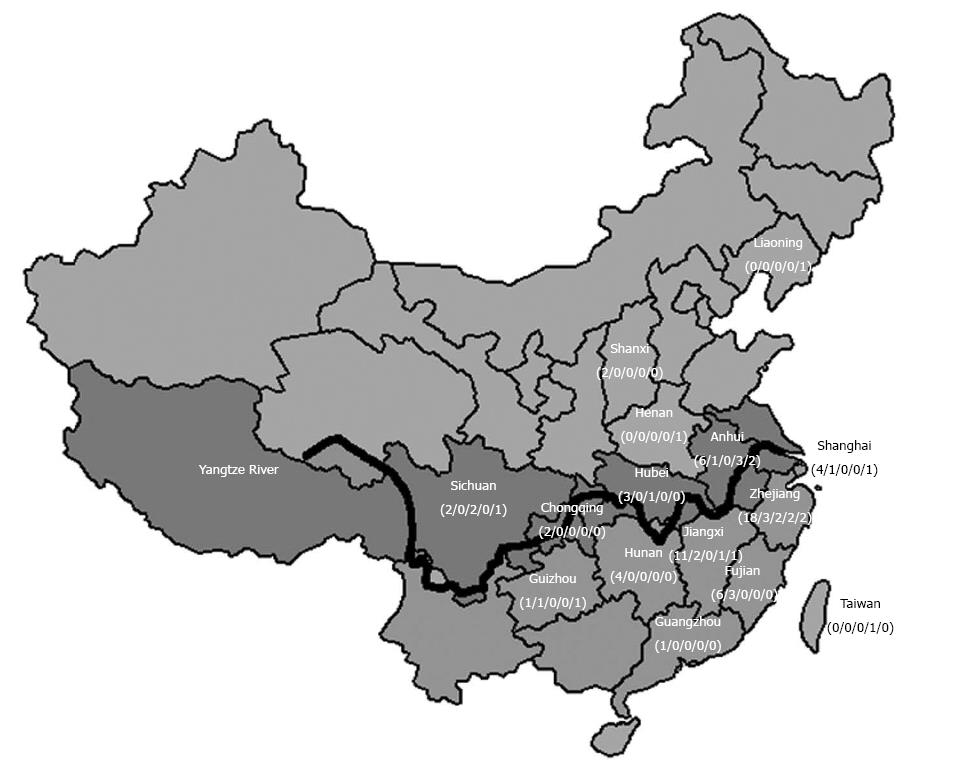
The population boundary between northern and southern China is drawn at the historical border of the Yangtze River during early Neolithic times (3000-7000 years ago)[26]. According to this criteria, Zhejiang, Jiangxi, Fujian, Guangdong, Hunan, Guizhou, Taiwan are classified as southern areas as they are south of Yangtze River; the provinces of Jiangsu, Shanghai, Anhui, Hubei, Sichuan and Chongqing are classified as border areas as they are in the basin of the Yangtze River; and the provinces of Henan, Liaoning, Shanxi, Jilin, Shandong, Hebei and Ningxia are classified as northern areas as they are in north of Yangtze River (Figure 2).
Patients with at least one mutated SLC25A13 allele were selected. To calculate the mutation spectra, the mutations observed in the related family members were counted only once. If the patient was a heterozygote or a compound heterozygote and the parents were from different regions, the parent’s sample was tested to determine the origin of the allele(s). Except for two patients who were born from consanguineous parents, all other infants were to our knowledge unrelated.
The homology between the mutated Citrin protein and the human reference, as well as Citrin from other species, were surveyed using Clustal X software (European Bioinformatics Institute, Hinxton, Saffron Walde, United kingdom). PolyPhen-2 (Polymorphism Phenotyping version2.2.2), which is available at http://genetics.bwh.harvard.edu/pph2/, was used to predict the possible impact of an amino acid substitution on the structure and function of the Citrin protein. MutationTaster was used to evaluate the disease-causing potential of sequence alterations, at http://mutationtaster.org/MutationTaster/index.html. A P value close to 1 indicates a high ‘security’ of the prediction. MutationTaster employs a Bayes classifier to eventually predict the potential of an alteration causing disease. The Bayes classifier is fed with the outcome of all tests and the features of the alterations and calculates probabilities for the alteration to be either disease causing or not.
Statistical tests on the distribution of mutant genotypes in the three areas of China were assessed by performing a 2 × 2 χ2 test with the SPSS version 17.0 software (University of Chicago, Chicago, IL, United States) package. A P value < 0.05 was considered to be statistically significant. When there are small expected values in the 2 × 2 table, the result of fisher’s exact test was used.
Among the 535 patients, 183 originated from the southern area, 291 were from the border area and 61 were from the northern area. Sixty-nine patients with at least one SLC25A13 gene mutation were found, including 6 sibling pairs. These sixty-three unrelated patients, including 25 females and 38 males, were further analyzed. Sixteen (25%) were found to be homozygotes for one mutation, 28 (44%) were compound heterozygotes and 19 (30%) heterozygotes for only one mutation. The distribution of carriers according to the state of origin is depicted in Figure 2.
A total of 17 mutations, including 14 mutations that had been previously reported by us and others (c.851_854del, c.1638_1660dup23, c.615+5G>A, c.1750+72_1751-4dup17insNM138459.3:2667, c.1019_1177del, c.1801G>A, c.550C>T, c.1078C>T, c.955C>T, c.1754G>A, c.775C>T, c.1092_5delT, c.615+1G>A, c.254T>C)[17,20,24,27-30] and 3 novel mutations (c.985_986insT, c.287T>C, c.1349A>G),were observed in the present investigation (Table 1).
| Mutation | South | Border | North | Nucleotide change | Protein change | Ref. |
| Common | 56 (93) | 34 (79)1 | 2 (50)2 | |||
| 851del4 | 41 (68) | 22 (51) | 2 (50) | c.851_854del | p.M285fsX286 | Kobayashi et al[1] |
| 1638ins23 | 9 (15) | 4 (9) | c.1638_1660dup23 | p.A554fsX570 | Kobayashi et al[1] | |
| IVS6+5G>A | 2 (3) | 4 (9) | c.615 + 5G>A | - | Saheki et al[23] | |
| IVS16ins3kb | 4 (7) | 4 (9) | c. 1750+72_1751-4dup17ins | p.A584fsX585 | Tabata et al[20] | |
| NM_138459.3:2667 | ||||||
| Other | 4 (7) | 9 (21) | 2 (50) | |||
| IVS11+1G>A | 1 (25) | c.1019_1177del | - | Kobayashi et al[1] | ||
| E601K | 1 (2) | 1 (2) | c.1801G>A | p.E601K | Yamaguchi et al[17] | |
| R184X | 1 (2) | c.550C>T | p.R184X | Lu et al[19] | ||
| R360X | 1 (25) | c.1078C>T | p.R360X | Tabata et al[20] | ||
| R319X | 1 (2) | c.955C>T | p.R319X | Song et al[29] | ||
| IVS6+1G>A | 2 (5) | c.615+1G>A | - | Fu et al[24] | ||
| L85P | 1 (2) | c.254T>C | p.L85P | Fu et al[24] | ||
| R585H | 1 (2) | c.1754G>A | p.R585H | Song et al[28] | ||
| 1092_5delT | 1 (2) | c.1092_5delT | p.R319X | Fu et al[24] | ||
| Q259X | 1 (2) | c.775C>T | p.Q259X | Wen et al[30] | ||
| 985insT | 1 (2) | c.985_986insT | p.A329fsX372 | Present study | ||
| F96S | 1 (2) | c.287T>C | p.F96S | Present study | ||
| E450G | 1 (2) | c.1349A>G | p.E450G | Present study |
The c.287T>C mutation in exon 4 is predicted to result in the substitute of phenylalanine to serine at position 96 (p.F96S). This mutation was found in a compound heterozygote state with the mutations c.851_854del. p.F96S is located between the second and third EF-hand domain, which is highly conserved in different species (Table 2). The Polymorphism Phenotyping for the variant amino acid p.F96S from Polyphen 2 is 1.000, indicating that the missense mutation has a high chance of affecting protein function. The P value from MutationTaster is 0.997, suggesting that is most likely a disease-causing mutation.
| F96S | |||||||||||||||||||||||
| Human | L | C | A | P | D | A | L | F | M | V | A | F | Q | L | F | D | K | A | G | K | G | E | V |
| Canis | L | C | A | P | D | A | L | F | M | V | A | F | Q | L | F | D | K | A | G | K | G | E | V |
| Bos | L | C | A | P | D | A | L | F | M | V | A | F | Q | L | F | D | K | A | G | K | G | E | V |
| Equus | L | C | A | P | D | A | L | F | M | V | A | F | Q | L | F | D | K | A | G | K | G | E | V |
| Pan | L | C | A | P | D | A | L | F | M | V | A | F | Q | L | F | D | K | A | G | K | G | E | V |
| Mus | L | C | A | P | D | A | L | F | M | V | A | F | Q | L | F | D | K | A | G | K | G | E | V |
| Gallus | L | C | A | P | D | A | L | F | M | V | A | F | Q | L | F | D | K | A | G | K | G | E | V |
| Xenopus | L | C | A | P | D | A | L | F | M | V | A | F | Q | L | F | D | K | A | G | K | G | E | V |
| E450G | |||||||||||||||||||||||
| Human | G | G | S | Q | V | I | F | T | N | P | L | E | I | V | K | I | R | L | Q | V | A | G | E |
| Canis | G | G | S | Q | V | I | F | T | N | P | L | E | I | V | K | I | R | L | Q | V | A | G | E |
| Bos | G | G | S | Q | V | I | F | T | N | P | L | E | I | V | K | I | R | L | Q | V | A | G | E |
| Equus | G | G | S | Q | V | I | F | T | N | P | L | E | I | V | K | I | R | L | Q | V | A | G | E |
| Pan | G | G | S | Q | V | I | F | T | N | P | L | E | I | V | K | I | R | L | Q | V | A | G | E |
| Mus | G | G | S | Q | V | I | F | T | N | P | L | E | I | V | K | I | R | L | Q | V | A | G | E |
| Gallus | G | M | C | Q | V | V | F | T | N | P | L | E | I | V | K | I | R | L | Q | T | A | G | E |
Mutations c.985_986insT and c.1349A>G were found in compound heterozygote state in a patient. The mutation c.985_986insT was found to be derived from this patient’s paternal allele and predicted to result in a frame shift and the introduction of a premature stop codon at position 372. Mutation c.1349A>G (p.E450G) was derived from the patient’s maternal allele, which is located in the loop between the TM3 and TM4 spanning regions. Conservation analysis in different species indicated that the amino acid in this position is highly conserved (Table 2). The Polymorphism Phenotyping for the variant amino acid p.E450G was 1.000, indicating a high chance of affecting protein function. The P value from MutationTaster is more than 0.999, suggesting that the mutation might affect the protein’s features.
The distribution of SLC25A13 mutations in carriers originating from different regions of China is given in Figure 1. Sixty (56%) mutant alleles originated from the southern region with Zhejiang, Jiangxi, Fujian, Guangdong, Hunan, Guizhou, and Taiwan accounting for 27, 15, 9, 1, 4, 3 and 1 mutant allele, respectively. Forty-three mutant alleles (40%) originated from the border region, with Jiangsu, Shanghai, Anhui, Hubei, Sichuan, and Chongqing accounting for 14, 6, 12, 4, 5 and 2, respectively. In the northern region, only four mutant alleles (4%) were found, including 1 from Henan, 1 from Liaoning and 2 from Shanxi provinces.
The SLC25A13 mutation spectra among the three regions of China presented significant differences. The four common mutations exhibited maximal relative frequencies in the southern China (56/60, 93%), and four other mutations were detected, including two novel mutations. On the contrary, in the area bordering the Yangtze River, there was a wide range of mutation types. The four common mutations accounted for 79% (34/43) of the mutant alleles, and eight other mutation types were found, including one novel mutation. In northern China the mutation c.851_854del accounted for 50% (2/4) of the mutant alleles. The other two mutations were c.1019_1177del and c.1078C>T. The proportion of the four common mutations was higher in south region (93%) than that in the border region (79%, χ2 = 4.621, P = 0.032) and that in the north (50%, χ2 = 8.288, P = 0.041) (Table 1).
Among the four common mutations, c.851_854del was the most. Proportion accounts for 68% (41/60) in southern region, 51% (22/43) in the border region and 50% (2/4) in the northern region. The difference of the proportion between the southern and border part of China was marginally significant (χ2 = 3.109, P = 0.078).
The mutation spectra for the SLC25A13 gene differ within the Asian population[18-20]. A different carrier rate for the common mutations between different parts of China has been reported[19,30]. Here we demonstrate that the mutation spectrum of SLC25A13 gene varies considerably among specific regions of China with common mutations having a higher proportion in the southern region than in the border and northern regions.
In the southern region of China, four common mutations accounted for 93% and c.851_854del is the predominant mutation accounting for 68%. This is consistent with the published data[19,30]. The c.851_854del mutation is a common ancestral mutation, and the frequency difference between various regions of China may be associated with ancient migration.
The mutations found in patients from the border region exhibited significant variety. The total mutant allele number in patients from the border was less than that in patients from the southern region (43 vs 60), but the mutation types were much greater than that in the southern area (12 vs 8). In total, seven private mutations were found in patients from the border, compared with three private mutations found in patients from the southern region. This divergence may reflect the ethnic diversity of this area. Previously, the data on SLC25A13 mutations in this region were very limited. This study is the first paper providing an estimate for the border region and significantly increases the data on southern and northern China. Considering the high proportion of uncommon mutations, the previously reported SLC25A13 mutation carrier rate in this region may be underestimated and sequencing may be a prefect method for testing.
The c.851_854del mutation was the only one of the four common mutations detected in the northern region. The explanation for this may be that other common mutations are rare in that part of China. Two mutant c.851_854del alleles were found among the 4 known mutant alleles, suggesting mutation 851del4 is the frequent mutation in this region.
Since variants c.287T>C and c.1349A>G have not undergone functional testing, so we analyzed the data without these and statistical significance was reached even when those two variants were removed from the analysis. The proportion of the four common mutations was also higher in southern region (95%) than the border (81%, χ2 = 4.929, P = 0.048) and the northern region (50%, χ2 = 10.343, P = 0.029). Thus the primary conclusion remains valid.
This paper is the first study conducted the SLC25A13 mutation spectrum in neonatal intrahepatic cholestasis from different parts of China. The previous study evaluated the population frequency for the common mutations and conducted that the carrier frequency in China is 1/79-1/65[17,19]. Conversely only 94% (59/63) of cases with suspected citrin deficiency in our study had the common mutations. This suggests that point mutation testing alone is not sufficient to exclude citrin deficiency even in cases from the southern region but may be a cost effective way of confirming the diagnosis as the first step.
There were limitations of this study. Firstly, only a small number of patients came from the north of the Yangtze River, and only limited cases were reported from that area, so the sampling bias needs to be considered. The current literature has not shown a significant difference between SLC25A13 mutation types and the phenotype observed, so the smaller sample size is not likely to lead to referral bias in favor of null or missense mutations in this study. Secondly, for 19 of the 126 alleles, we could not find any mutations. One possible explanation could be that the patients with one detected mutant allele are carriers and this may be a risk factor for cholestasis or they may have an alternate cause for cholestasis. Alternatively, as previously described[31,32] they may have a second mutation not detected by Sanger sequencing or the targeted test for the IVS16ins3kb rearrangement such as intronic mutations or large rearrangements.
In conclusion, the mutation spectra of the SLC25A13 gene are significantly different among patients with neonatal intrahepatic cholestasis from different parts of China. These differences should be considered when establishing a molecular diagnostic strategy or interpreting their results.
We thank David P Dimmock for helpful discussions and comments on an initial version of the manuscript. We also thank the patients and their parents for their kind cooperation as well as the physicians who referred the patients.
SLC25A13 gene mutations lead to Citrin deficiency, which includes adult-onset type II citrullinemia and neonatal intrahepatic cholestasis caused by citrin deficiency. The carrier frequency is high in Asian populations, and the mutation spectrum of SLC25A13 gene in Chinese population (most came from southern China) was found to be different from that of other population groups in East Asia.
The mutant alleles of SLC25A13 gene was conducted in southern, border and northern regions of China, providing a basis for the improvement of diagnostic strategies and interpretation of genetic diagnosis.
The proportion of four common mutations was higher in the southern region (56/60, 93%) than that in the border region (34/43, 79%, χ2 = 4.621, P = 0.032) and the northern region (2/4, 50%, χ2 = 8.288, P = 0.041). Three novel mutations were found, which has expanded the SLC25A13 mutation spectrum.
The mutation spectra of the SLC25A13 gene are significantly different among patients with neonatal intrahepatic cholestasis from different parts of China. These differences should be considered when establishing a molecular diagnostic strategy or interpreting their results.
An allele is a single copy of a gene. For autosomal genes, an individual inherits two alleles at each locus, with one from each parent. Genotypes are described as homozygous if the two alleles are the same and as heterozygous if the alleles are different. The mutant allele is the mutated form of a gene.
This is a retrospective study aimed at investigating the regional distribution of SLC25A13 mutations in Chinese patients with neonatal intrahepatic cholestasis. The topic is relevant, since biochemical diagnosis of citrin deficiency is not widely available and mutation analysis of the SLC25A13 gene is crucial to diagnosis. The study was well-conducted and the manuscript is reasonably well written with good scientific value.
P- Reviewers CarvalhoRJ, Qin P S- Editor Zhai HH L- Editor A E- Editor Zhang DN
| 1. | Kobayashi K, Sinasac DS, Iijima M, Boright AP, Begum L, Lee JR, Yasuda T, Ikeda S, Hirano R, Terazono H. The gene mutated in adult-onset type II citrullinaemia encodes a putative mitochondrial carrier protein. Nat Genet. 1999;22:159-163. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 293] [Cited by in F6Publishing: 310] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 2. | Yasuda T, Yamaguchi N, Kobayashi K, Nishi I, Horinouchi H, Jalil MA, Li MX, Ushikai M, Iijima M, Kondo I. Identification of two novel mutations in the SLC25A13 gene and detection of seven mutations in 102 patients with adult-onset type II citrullinemia. Hum Genet. 2000;107:537-545. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 96] [Cited by in F6Publishing: 103] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 3. | Ohura T, Kobayashi K, Tazawa Y, Nishi I, Abukawa D, Sakamoto O, Iinuma K, Saheki T. Neonatal presentation of adult-onset type II citrullinemia. Hum Genet. 2001;108:87-90. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 92] [Cited by in F6Publishing: 99] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 4. | Tazawa Y, Kobayashi K, Ohura T, Abukawa D, Nishinomiya F, Hosoda Y, Yamashita M, Nagata I, Kono Y, Yasuda T. Infantile cholestatic jaundice associated with adult-onset type II citrullinemia. J Pediatr. 2001;138:735-740. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 81] [Cited by in F6Publishing: 84] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 5. | Tomomasa T, Kobayashi K, Kaneko H, Shimura H, Fukusato T, Tabata M, Inoue Y, Ohwada S, Kasahara M, Morishita Y. Possible clinical and histologic manifestations of adult-onset type II citrullinemia in early infancy. J Pediatr. 2001;138:741-743. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 75] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 6. | Ohura T, Kobayashi K, Tazawa Y, Abukawa D, Sakamoto O, Tsuchiya S, Saheki T. Clinical pictures of 75 patients with neonatal intrahepatic cholestasis caused by citrin deficiency (NICCD). J Inherit Metab Dis. 2007;30:139-144. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 108] [Cited by in F6Publishing: 111] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 7. | Tamamori A, Okano Y, Ozaki H, Fujimoto A, Kajiwara M, Fukuda K, Kobayashi K, Saheki T, Tagami Y, Yamano T. Neonatal intrahepatic cholestasis caused by citrin deficiency: severe hepatic dysfunction in an infant requiring liver transplantation. Eur J Pediatr. 2002;161:609-613. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 74] [Cited by in F6Publishing: 75] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 8. | Shigeta T, Kasahara M, Kimura T, Fukuda A, Sasaki K, Arai K, Nakagawa A, Nakagawa S, Kobayashi K, Soneda S. Liver transplantation for an infant with neonatal intrahepatic cholestasis caused by citrin deficiency using heterozygote living donor. Pediatr Transplant. 2010;14:E86-E88. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 9. | Xing YZ, Qiu WJ, Ye J, Han LS, Xu SS, Zhang HW, Gao XL, Wang Y, Gu XF. Studies on the clinical manifestation and SLC25A13 gene mutation of Chinese patients with neonatal intrahepatic cholestasis caused by citrin deficiency. Zhonghua Yixue Yichuanxue Zazhi. 2010;27:180-185. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 11] [Reference Citation Analysis (0)] |
| 10. | Song YZ, Deng M, Chen FP, Wen F, Guo L, Cao SL, Gong J, Xu H, Jiang GY, Zhong L. Genotypic and phenotypic features of citrin deficiency: five-year experience in a Chinese pediatric center. Int J Mol Med. 2011;28:33-40. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 11. | Saheki T, Kobayashi K. Mitochondrial aspartate glutamate carrier (citrin) deficiency as the cause of adult-onset type II citrullinemia (CTLN2) and idiopathic neonatal hepatitis (NICCD). J Hum Genet. 2002;47:333-341. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 212] [Cited by in F6Publishing: 222] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 12. | Saheki T, Kobayashi K, Iijima M, Moriyama M, Yazaki M, Takei Y, Ikeda S. Metabolic derangements in deficiency of citrin, a liver-type mitochondrial aspartate-glutamate carrier. Hepatol Res. 2005;33:181-184. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 44] [Cited by in F6Publishing: 47] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 13. | Dimmock D, Maranda B, Dionisi-Vici C, Wang J, Kleppe S, Fiermonte G, Bai R, Hainline B, Hamosh A, O’Brien WE. Citrin deficiency, a perplexing global disorder. Mol Genet Metab. 2009;96:44-49. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 65] [Cited by in F6Publishing: 56] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 14. | Dimmock D, Kobayashi K, Iijima M, Tabata A, Wong LJ, Saheki T, Lee B, Scaglia F. Citrin deficiency: a novel cause of failure to thrive that responds to a high-protein, low-carbohydrate diet. Pediatrics. 2007;119:e773-e777. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 76] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 15. | Sinasac DS, Crackower MA, Lee JR, Kobayashi K, Saheki T, Scherer SW, Tsui LC. Genomic structure of the adult-onset type II citrullinemia gene, SLC25A13, and cloning and expression of its mouse homologue. Genomics. 1999;62:289-292. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 31] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 16. | Palmieri L, Pardo B, Lasorsa FM, del Arco A, Kobayashi K, Iijima M, Runswick MJ, Walker JE, Saheki T, Satrústegui J. Citrin and aralar1 are Ca(2+)-stimulated aspartate/glutamate transporters in mitochondria. EMBO J. 2001;20:5060-5069. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 365] [Cited by in F6Publishing: 360] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 17. | Yamaguchi N, Kobayashi K, Yasuda T, Nishi I, Iijima M, Nakagawa M, Osame M, Kondo I, Saheki T. Screening of SLC25A13 mutations in early and late onset patients with citrin deficiency and in the Japanese population: Identification of two novel mutations and establishment of multiple DNA diagnosis methods for nine mutations. Hum Mutat. 2002;19:122-130. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 82] [Cited by in F6Publishing: 86] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 18. | Kobayashi K, Bang Lu Y, Xian Li M, Nishi I, Hsiao KJ, Choeh K, Yang Y, Hwu WL, Reichardt JK, Palmieri F. Screening of nine SLC25A13 mutations: their frequency in patients with citrin deficiency and high carrier rates in Asian populations. Mol Genet Metab. 2003;80:356-359. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 60] [Cited by in F6Publishing: 60] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 19. | Lu YB, Kobayashi K, Ushikai M, Tabata A, Iijima M, Li MX, Lei L, Kawabe K, Taura S, Yang Y. Frequency and distribution in East Asia of 12 mutations identified in the SLC25A13 gene of Japanese patients with citrin deficiency. J Hum Genet. 2005;50:338-346. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 81] [Cited by in F6Publishing: 85] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 20. | Tabata A, Sheng JS, Ushikai M, Song YZ, Gao HZ, Lu YB, Okumura F, Iijima M, Mutoh K, Kishida S. Identification of 13 novel mutations including a retrotransposal insertion in SLC25A13 gene and frequency of 30 mutations found in patients with citrin deficiency. J Hum Genet. 2008;53:534-545. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 86] [Cited by in F6Publishing: 94] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 21. | Kikuchi A, Arai-Ichinoi N, Sakamoto O, Matsubara Y, Saheki T, Kobayashi K, Ohura T, Kure S. Simple and rapid genetic testing for citrin deficiency by screening 11 prevalent mutations in SLC25A13. Mol Genet Metab. 2012;105:553-558. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 22. | De Bruyne R, Van Biervliet S, Vande Velde S, Van Winckel M. Clinical practice: neonatal cholestasis. Eur J Pediatr. 2011;170:279-284. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 55] [Cited by in F6Publishing: 60] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 23. | Saheki T, Kobayashi K, Iijima M, Horiuchi M, Begum L, Jalil MA, Li MX, Lu YB, Ushikai M, Tabata A. Adult-onset type II citrullinemia and idiopathic neonatal hepatitis caused by citrin deficiency: involvement of the aspartate glutamate carrier for urea synthesis and maintenance of the urea cycle. Mol Genet Metab. 2004;81 Suppl 1:S20-S26. [PubMed] [Cited in This Article: ] |
| 24. | Fu HY, Zhang SR, Wang XH, Saheki T, Kobayashi K, Wang JS. The mutation spectrum of the SLC25A13 gene in Chinese infants with intrahepatic cholestasis and aminoacidemia. J Gastroenterol. 2011;46:510-518. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 25. | den Dunnen JT, Antonarakis SE. Mutation nomenclature extensions and suggestions to describe complex mutations: a discussion. Hum Mutat. 2000;15:7-12. [PubMed] [Cited in This Article: ] |
| 26. | Zhao TM, Lee TD. Gm and Km allotypes in 74 Chinese populations: a hypothesis of the origin of the Chinese nation. Hum Genet. 1989;83:101-110. [PubMed] [Cited in This Article: ] |
| 27. | Takaya J, Kobayashi K, Ohashi A, Ushikai M, Tabata A, Fujimoto S, Yamato F, Saheki T, Kobayashi Y. Variant clinical courses of 2 patients with neonatal intrahepatic cholestasis who have a novel mutation of SLC25A13. Metabolism. 2005;54:1615-1619. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 28. | Song YZ, Ushikai M, Sheng JS, Iijima M, Kobayashi K. SLC25A13 gene mutation analysis in a pedigree of neonatal intrahepatic cholestasis caused by citrin deficiency. Zhonghua Erke Zazhi. 2007;45:408-412. [PubMed] [Cited in This Article: ] |
| 29. | Song YZ, Sheng JS, Ushikai M, Hwu WL, Zhang CH, Kobayashi K. Identification and diagnosis of three novel mutations in SLC25A13 gene of neonatal intrahepatic cholestasis caused by citrin deficiency. Zhonghua Erke Zazhi. 2008;46:411-415. [PubMed] [Cited in This Article: ] |
| 30. | Wen PQ, Wang GB, Chen ZL, Cui D, Yuan Q, Song P, Chen SL, Liao JX, Li CR. SLC25A13 gene analysis in neonates with intrahepatic cholestasis caused by citrin deficiency. Zhongguo Dangdai Erke Zazhi. 2011;13:303-308. [PubMed] [Cited in This Article: ] |
| 31. | Fu HY, Zhang SR, Yu H, Wang XH, Zhu QR, Wang JS. Most common SLC25A13 mutation in 400 Chinese infants with intrahepatic cholestasis. World J Gastroenterol. 2010;16:2278-2282. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 6] [Cited by in F6Publishing: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 32. | Ben-Shalom E, Kobayashi K, Shaag A, Yasuda T, Gao HZ, Saheki T, Bachmann C, Elpeleg O. Infantile citrullinemia caused by citrin deficiency with increased dibasic amino acids. Mol Genet Metab. 2002;77:202-208. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 47] [Article Influence: 2.1] [Reference Citation Analysis (0)] |