Published online Jun 14, 2013. doi: 10.3748/wjg.v19.i22.3439
Revised: December 22, 2012
Accepted: January 11, 2013
Published online: June 14, 2013
AIM: To compare deep sedation with propofol-fentanyl and midazolam-fentanyl regimens during upper gastrointestinal endoscopy.
METHODS: After obtaining approval of the research ethics committee and informed consent, 200 patients were evaluated and referred for upper gastrointestinal endoscopy. Patients were randomized to receive propofol-fentanyl or midazolam-fentanyl (n = 100/group). We assessed the level of sedation using the observer’s assessment of alertness/sedation (OAA/S) score and bispectral index (BIS). We evaluated patient and physician satisfaction, as well as the recovery time and complication rates. The statistical analysis was performed using SPSS statistical software and included the Mann-Whitney test, χ2 test, measurement of analysis of variance, and the κ statistic.
RESULTS: The times to induction of sedation, recovery, and discharge were shorter in the propofol-fentanyl group than the midazolam-fentanyl group. According to the OAA/S score, deep sedation events occurred in 25% of the propofol-fentanyl group and 11% of the midazolam-fentanyl group (P = 0.014). Additionally, deep sedation events occurred in 19% of the propofol-fentanyl group and 7% of the midazolam-fentanyl group according to the BIS scale (P = 0.039). There was good concordance between the OAA/S score and BIS for both groups (κ = 0.71 and κ = 0.63, respectively). Oxygen supplementation was required in 42% of the propofol-fentanyl group and 26% of the midazolam-fentanyl group (P = 0.025). The mean time to recovery was 28.82 and 44.13 min in the propofol-fentanyl and midazolam-fentanyl groups, respectively (P < 0.001). There were no severe complications in either group. Although patients were equally satisfied with both drug combinations, physicians were more satisfied with the propofol-fentanyl combination.
CONCLUSION: Deep sedation occurred with propofol-fentanyl and midazolam-fentanyl, but was more frequent in the former. Recovery was faster in the propofol-fentanyl group.
- Citation: Santos MELD, Maluf-Filho F, Chaves DM, Matuguma SE, Ide E, Luz GO, Souza TF, Pessorrusso FCS, Moura EGH, Sakai P. Deep sedation during gastrointestinal endoscopy: Propofol-fentanyl and midazolam-fentanyl regimens. World J Gastroenterol 2013; 19(22): 3439-3446
- URL: https://www.wjgnet.com/1007-9327/full/v19/i22/3439.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i22.3439
The routine use of sedation during endoscopic procedures is increasing worldwide[1,2]. In a survey conducted in the United States in 2006, > 98% of endoscopies and colonoscopies were performed under sedation[1,3]. A similar trend has been observed in Switzerland, Germany, and Australia[2,4-7]. The combination of a benzodiazepine and opioid is reportedly used at approximately 75% of all healthcare facilities in the United States[1,3] and considered the combination of choice by most endoscopists worldwide[1,2,4-9]. As a result of its anxiolytic and sedative properties, its ability to provide anterograde amnesia, and its short half-life, midazolam is the most widely used benzodiazepine. Fentanyl is the most widely used opioid, although meperidine is still frequently used[2-4,8,9].
Propofol is a hypnotic agent that induces anesthesia almost immediately and has a short half-life. It also allows the patient to recover rapidly and be discharged. Patient and physician satisfaction is high with propofol. As a result of these properties, the use of propofol has been adopted at endoscopy centers worldwide[1-7,10]. However, its use has also been associated with deep sedation[11-14].
During endoscopy, sedation and analgesia improve the efficiency of the procedure, quality of the results, and comfort of the patient[5,8]. However, sedation is also responsible for the majority of complications related to diagnostic endoscopy[15]. During sedation and analgesia, there is a continuum of states, ranging from mild sedation to general anesthesia. In the middle of this continuum is conscious sedation, which is the target level of sedation for patients undergoing upper or lower gastrointestinal endoscopy[4,5,10,11,13,14,16-19]. A level of sedation deeper than that intended is associated with a higher rate of complications[11,15,20]. The guidelines established in 2002 by the American Society of Anesthesiologists (ASA) task force on sedation and analgesia by non-anesthesiologists, which have also been endorsed by the American Society for Gastrointestinal Endoscopy, recommend that a distinction be made between conscious sedation and deep sedation and that one professional be dedicated to the assessment and monitoring of patients during sedation[11-13].
The level of consciousness is typically assessed with the subjective clinical scale known as the observer’s assessment of alertness/sedation (OAA/S) score, as validated by Chernik et al[21], which ranks sedation as mild, conscious, or deep. Another means of assessing the level of consciousness is the calculation of the bispectral index (BIS). Through the use of a BIS monitor, complex mathematical calculations of electroencephalography waves are transformed into numbers ranging from 0 (no brain activity) to 100 (fully conscious). This provides an objective measure of the level of consciousness. The BIS is considered a viable alternative for monitoring the level of consciousness in patients submitted to general anesthesia. Although its use in endoscopy is controversial, it has been investigated with increasing frequency, and further studies are recommended[22-27].
In the past 10 years, new sedation guidelines have been established. Many of those guidelines state that propofol can be safely administered by an endoscopist or nurse under the supervision of a physician[1,3-5,8,10,12,14,17,25,28,29]. However, because propofol has been associated with deep sedation events and complications, some have recommended that propofol be administered exclusively by anesthesiologists[5,11-13,17].
In view of these facts, we evaluated the use of propofol-fentanyl versus midazolam-fentanyl for sedation of patients undergoing upper gastrointestinal endoscopy. The primary endpoint of this study was to compare the frequency of deep sedation in each group. We also compared the two drug combinations in terms of time to induction, time to recovery, time to discharge, efficacy, and safety, as well as patient and endoscopist satisfaction.
This was a prospective, single-blind, randomized controlled trial carried out between January 2007 and October 2010 at the Gastrointestinal Endoscopy Clinic of the Department of Gastroenterology at the University of São Paulo Medical School - Hospital das Clínicas, Brazil.
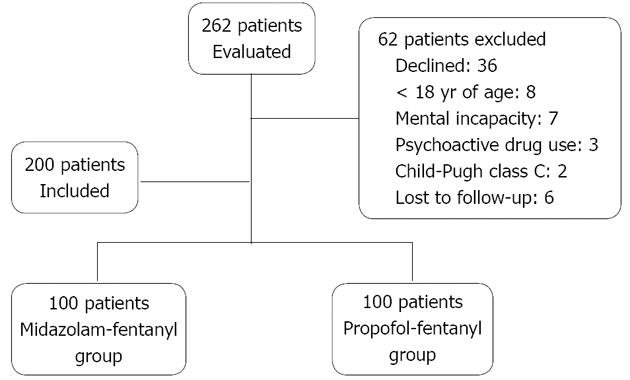
We recruited 262 patients from those scheduled to undergo upper gastrointestinal endoscopy at the Gastrointestinal Endoscopy Unit. The inclusion criteria were age > 18 years, physical status classified as ASA I, II or III, and having a contact telephone number. The exclusion criteria were as follows: pregnancy; a history of allergy to the medications to be administered; a history of allergy to soy or eggs; a psychotic disorder; being under treatment with psychoactive medications; being an illicit drug user or a heavy consumer of alcohol; Child-Pugh class C cirrhosis; presence of chronic kidney disease (being on dialysis); and being submitted to endoscopy as an emergency procedure. Of the 262 patients recruited, 62 were excluded (Figure 1). The final sample comprised 200 patients. Through a drawing of sealed envelopes, patients were randomized to two groups of 100: propofol-fentanyl and midazolam-fentanyl. The endoscopists scheduled to perform the procedures had no access to the envelopes.
Drug infusion was performed by the nursing staff and attending endoscopist. In both groups, the objective was to achieve conscious sedation by using the dose calculated for that purpose (Table 1). The propofol and midazolam were administered by intravenous bolus and supplemented as necessary by the endoscopist to maintain the desired level of sedation. A single dose of fentanyl was used in both groups. The fixed maximum dose for midazolam was 10 mg or 0.1 mg/kg of body weight. If additional sedation became necessary, the endoscopist had the option of suspending the initial regimen and adding propofol.
| Midazolam-fentanyl | |
| Midazolam | |
| Initial dose | |
| ASA I | 3-5 mg |
| ASA II or III | 2-3 mg |
| Maintenance | 0.5-1.0 mg every 2-3 min, up to a maximum cumulative dose of 10 mg or 0.1 mg/kg of body weight |
| Fentanyl | |
| Single dose | |
| ASA I | 50 μg |
| ASA II or III | 20-30 μg |
| Propofol-fentanyl | |
| Propofol | |
| Initial dose | |
| ASA I | 0.5 mg/kg |
| ASA II or III | 0.25 mg/kg |
| Maintenance | 10-20 mg bolus at 60 s intervals |
| Fentanyl | |
| Single dose | |
| ASA I | 50 μg |
| ASA II or III | 20-30 μg |
Prior to the procedure, a clinical history and physical examination was performed for each patient. Additionally, the anesthetic risk was assessed with the ASA classification of physical status, and the patients completed a demographic questionnaire. Continuous monitoring during the procedure included the noninvasive measurement of blood pressure, heart rate, respiratory rate (thoracic excursion measurement), and oxygen saturation (SpO2). We defined the following evaluation time points: “baseline” (immediately before the procedure); “duodenum” (the approximate midpoint of the procedure, when the endoscopist was evaluating the duodenum or the jejunal loop in post-gastrectomy patients); and “recovery” (when the patient was awake and underwent the final evaluation).
During the procedure, the level of sedation was evaluated in two ways. We applied the OAA/S scale[21], which is scored as 1 for deep sedation, 2-4 for conscious sedation, and 5 for mild sedation. In addition, after cleaning the skin with gauze and alcohol, we applied disposable electrodes to the forehead and connected the leads to a BIS monitor (A-2000 BIS XP; Aspect Medical Systems, Newton, MA, United States) (Figure 2). The BIS monitor output was evaluated continuously throughout the procedure and recovery period. BIS ≤ 65 indicates deep sedation, 66-85 indicates conscious sedation, and BIS > 85 indicates mild sedation. The OAA/S scale and BIS were determined simultaneously every 2 min.
An independent observer was responsible for the monitoring, which included evaluating the level of consciousness, readout of other vital signs, collection of data regarding drugs and doses used, use of benzodiazepine or opioid antagonists, and occurrence of cardiorespiratory events, such as hypoxemia (defined as SpO2< 90% for > 30 s after application of the jaw thrust maneuver), hypotension (defined as ≥ 20% decrease in systolic or diastolic blood pressure), and bradycardia (heart rate < 50 bpm). Hypoxemia was classified as mild if it responded to supplemental oxygen delivered at 3-4 L/min; it was classified as severe if it did not respond to supplemental oxygen and the patient required noninvasive ventilatory support (e.g., bag-mask ventilation) or intubation. The same observer also reported any other adverse events that occurred secondary to sedation. The observer was blinded to the randomization.
As described by Cohen et al[30], we compared the two groups in terms of the time to induction (interval between the first drug bolus administration and initiation of the procedure), time to recovery (interval between removal of the endoscope and final evaluation), and time to discharge (interval between removal of the endoscope and departure from the endoscopy unit). The final evaluation began when the BIS monitor indicated at least 90. Patients were discharged only when they had achieved an OAA/S score of 5 (the maximum), a BIS > 90, and reported no pain or any other type of discomfort.
At discharge, patient satisfaction was assessed with a 10-point visual analog scale (1 = least satisfied and 10 = most satisfied). The patients also completed a satisfaction questionnaire before leaving the facility. At 24 h after discharge, the same observer who was responsible for the monitoring contacted the patients by telephone to administer a questionnaire that evaluated patient satisfaction with the procedure, adverse events and the resumption of domestic activities. All 200 patients completed both questionnaires. The visual analog scale was also applied to the endoscopists who performed the procedures to assess their level of satisfaction with the sedation regimen and was scored as follows: 1-3 = considerable difficulty in performing the procedure; 4-7 = minor difficulty in performing the procedure (patient moved at the beginning or end of the procedure); and 8-10 = no difficulty in performing the procedure.
This work was conducted in accordance with the Declaration of Helsinki (2000) of the World Medical Association. The study was approved by the Hospital das Clínicas Research Ethics Committee, and all participating patients provided written informed consent.
To calculate the sample size, we estimated the proportion of patients who received deep sedation by analyzing the BIS curve. We hypothesized that this proportion would be 10% for the midazolam-fentanyl group and 25% for the propofol-fentanyl group. By adopting an α error tolerance (false-positive risk) of 5% and β error tolerance (false-negative risk) of 20%, we determined that 112 patients per group would be required to provide sufficient power to detect significant differences. It was agreed that we would perform an interim analysis involving 100 patients in each group.
The data were entered into a Microsoft Excel spreadsheet and analyzed with the assistance of the Statistics Sector of the University of São Paulo Medical School, Department of Gastroenterology. The statistical analysis was performed with the Statistical Package for the Social Sciences (SPSS Inc., Chicago, IL, United States). We applied the Mann-Whitney test to evaluate continuous variables and used the χ2 test to evaluate categorical variables. To study the effects of the variable “group” at the various time points, we used repeated measures analyses of variance. We used the κ statistic to evaluate the degree of concordance between the OAA/S scale and BIS.
Data were collected by a researcher who was blinded to each patient’s group. However, such masking was not possible when we assessed endoscopist satisfaction with the sedation regimen.
Most patients presented with a low anesthetic risk (ASA class I or II), although one patient in the propofol-fentanyl group was classified as ASA III. There were no significant differences between the two groups regarding demographics, weight, height, body mass index, level of education, or ASA class (Table 2). All of the patients completed the procedure with adequate sedation throughout. None of the procedures were suspended or halted prematurely.
| Variable | Group | Total | P value | |
| Midazolam-fentanyl | Propofol-fentanyl | |||
| Sex | ||||
| Female | 66 (60.0) | 71 (71.0) | 137 (68.5) | 0.543 |
| Male | 34 (34.0) | 29 (29.0) | 63 (31.5) | |
| ASA | ||||
| I | 63 (63.0) | 55 (55.0) | 118 (59.0) | 0.316 |
| II | 37 (37.0) | 44 (44.0) | 81 (40.5) | |
| III | 0 (0.0) | 1 (0.5) | 1 (0.5) | |
| Age (yr) | 52.14 ± 15.01 | 54.40 ± 15.44 | 0.352 | |
| Weight (kg) | 67.45 ± 11.28 | 70.93 ± 17.64 | 0.242 | |
| Height (cm) | 1.62 ± 0.09 | 1.61 ± 0.10 | 0.546 | |
| BMI (kg/m2) | 25.91 ± 4.54 | 27.39 ± 6.59 | 0.251 | |
| DM proportion | 4.5% | 7.5% | 0.276 | |
| SH proportion | 16.0% | 18.0% | 0.640 | |
| Cardiopathy proportion | 0.5% | 0.0% | 0.999 | |
| Other proportion | 11.5% | 9.0% | 0.404 | |
Patients in the midazolam-fentanyl group received midazolam and fentanyl at mean doses of 5.25 ± 1.7 mg and 43.1 ± 9.87 μg, respectively. Patients in the propofol-fentanyl group received propofol and fentanyl at mean doses of 70.3 ± 38.9 mg and 41.0 ± 10.25 μg, respectively. Sixty minutes after the end of the procedure, a patient in the midazolam-fentanyl group presented with persistent drowsiness, despite normal cardiorespiratory function, and was given 0.2 mg flumazenil.
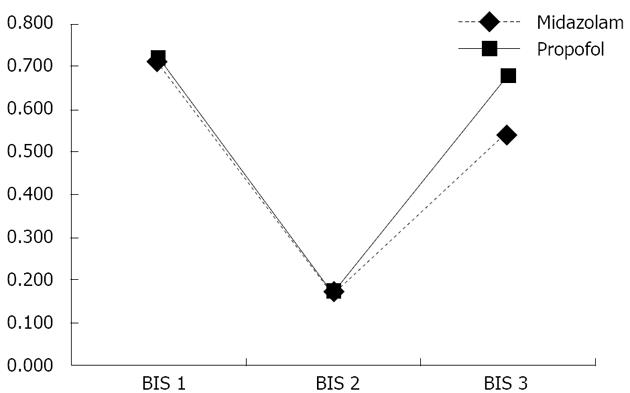
As seen in Table 3, the OAA/S classification of sedation in the midazolam-fentanyl group was mild in 1%, conscious in 88%, and deep in 11%, compared with 2%, 73%, and 25%, respectively, in the propofol-fentanyl group. There was a statistically significant difference between the two groups in terms of the OAA/S score (P = 0.014). Based on the BIS values, sedation in the midazolam-fentanyl and propofol-fentanyl groups was classified as mild in 18% and 14%, as conscious in 75% and 67%, and as deep in 7% and 19%, respectively (P = 0.039). Comparing the BIS values obtained before, during, and after the procedure, we found that there was a trend toward a return to its initial value more rapidly in the propofol-fentanyl group than in the midazolam-fentanyl group (Figure 3). The OAA/S score showed good concordance with the BIS in the midazolam-fentanyl group (κ= 0.635, P < 0.001), the propofol-fentanyl group (κ= 0.710, P < 0.001), and the sample as a whole (κ= 0.696, P < 0.001).
| Variable | Group | Total | P value | |
| Midazolam-fentanyl | Propofol-fentanyl | |||
| OAA/S score | ||||
| 1 | 11 (11.0) | 25 (25.0) | 36 (18.0) | 0.014 |
| 2-4 | 88 (88.0) | 73 (73.0) | 161 (80.5) | |
| 5 | 1(1.0) | 2 (2.0) | 3 (1.5) | |
| BIS | ||||
| ≤ 65 | 7 (7.0) | 19 (19.0) | 26 (13.0) | 0.039 |
| 66-85 | 75 (75.0) | 67 (67.0) | 142 (71.0) | |
| > 85 | 18 (18.0) | 14 (14.0) | 32 (16.0) | |
The mean score on the visual analog scale of patient satisfaction was 9.84 ± 0.4 in the midazolam-fentanyl group and 9.64 ± 0.8 in the propofol-fentanyl group (P = 0.178). The mean score on the visual analog scale for endoscopist satisfaction was 8.90 ± 1.2 for the midazolam-fentanyl regimen and 9.30 ± 0.9 for the propofol-fentanyl regimen (P = 0.012). The time to induction was significantly shorter in the propofol-fentanyl group (2.63 ± 1.62 min vs 2.96 ± 1.5 min, P = 0.012). The times to recovery and discharge were also shorter in the propofol-fentanyl group (P < 0.001 and P < 0.001, respectively).
We found no statistically significant differences between the two groups in terms of the patient-reported quality of sedation or pain/discomfort related to the procedure. The proportion of patients who remembered being awake at the beginning, middle, and end of the procedure was greater in the propofol-fentanyl group than the midazolam-fentanyl group (P < 0.001 for all three time points). According to the results of the post-discharge questionnaire, none of the patients experienced any adverse reactions within the first 24 h after discharge. On average, the patients in the propofol-fentanyl group reported having resumed their domestic activities 60 min after discharge compared with 80 min after discharge for the midazolam-fentanyl group (P < 0.001).
No serious complications were observed in either group. During the procedure, 42% of the propofol-fentanyl group patients developed mild transient hypoxemia (SpO2 of 85%-90% for > 30 s after the jaw thrust maneuver), which also occurred in 26% of the midazolam-fentanyl group patients (P = 0.025). In all of those cases, the hypoxemia responded to supplemental oxygen delivered by nasal cannula at 3-4 L/min. There were no instances of arrhythmia. Systolic hypotension was observed in 5% of the midazolam-fentanyl group and 10% of the propofol-fentanyl group, whereas diastolic hypotension was observed in 6% of the midazolam-fentanyl group and 16% of the propofol-fentanyl group. All of the variations in arterial blood pressure, heart rate, and respiratory rate were transient and required no mechanical or pharmacological intervention. There were no cases of perforation, bleeding, or death, and none of the patients required invasive ventilatory support, or hospitalization.
Although there are abundant data in the literature and various guidelines on sedation during endoscopy[1-6,10,12,17,31], few studies have compared propofol with midazolam for conscious sedation in patients undergoing diagnostic upper gastrointestinal endoscopy, especially when using the incidence of deep sedation events as the primary outcome. In the present study, we used the BIS and OAA/S scale in an innovative manner and assessed the frequency of deep sedation events for two sedation regimens frequently used during endoscopy[11,12,16,17,32,33]. In the midazolam-fentanyl group, deep sedation, defined according to the BIS and OAA/S score, occurred in 11% and 7% of the patients, respectively, compared with 25% and 19% of those in the propofol-fentanyl group. It was clear that despite the use of doses targeting conscious sedation, deep sedation events were common in the midazolam-fentanyl group. These findings are in agreement with those of Patel et al[20], who evaluated the use of a benzodiazepine-opioid combination with the objective of achieving conscious sedation in patients undergoing diagnostic upper gastrointestinal endoscopy. The authors found that deep sedation, as evaluated by a modified OAA/S scale, occurred in 26% of the patients. In the present study, the overall rates of deep sedation were 18% and 13% when assessed by the OAA/S score and BIS, respectively. However, we found that the frequency of clinically relevant complications was negligible, which is likely attributable to the relatively good overall health status of the patients. It is notable that the adverse effects arising from deep sedation were easily reversed with simple clinical maneuvers. There was a trend toward the BIS returning to its initial value more rapidly in the propofol-fentanyl group, which suggests faster recovery in those patients.
The principal complication that occurred in our study was hypoxemia, which was observed in 42% of the patients in the propofol-fentanyl group and 26% of the midazolam-fentanyl group. In all of those patients, hypoxemia responded to supplemental oxygen delivery. The close monitoring of the patients during sedation could explain the relatively high rates of transient mild hypoxemia found in both of the groups. These findings again raise the controversy regarding giving supplemental oxygen routinely during upper gastrointestinal endoscopy, which is our current practice. In addition, the small size of our sample prevented us from effectively evaluating the incidence of severe complications, which are rare in endoscopy.
In the pre-endoscopy, pre-medication period, patients typically have a BIS > 93, whereas a BIS of 60-70 is indicative of deep sedation[24]. There are some discrepancies between the data in the literature and information provided by the manufacturer in terms of the relationship between the numerical values and levels of consciousness[23,24,26,27]. Some authors define deep sedation as a BIS of 60-70 and conscious sedation as > 70[22], while others define deep sedation as BIS < 75 in the presence of an OAA/S score of 1 or 2[27]. To improve the sensitivity and specificity of our evaluations, we defined deep sedation as BIS ≤ 65 and an OAA/S score of 1 and conscious sedation as BIS > 65 and an OAA/S score > 1. Bower et al[24] have suggested that a BIS of 75-85 indicates conscious sedation, which is an appropriate level of sedation for endoscopic procedures. In assessing the level of consciousness of patients undergoing endoscopic procedures, those authors demonstrated a strong temporal correlation between the BIS and OAA/S score (r = 0.59, P < 0.0001). In the present study, we observed good concordance between the BIS and OAA/S score (κ= 0.7, P < 0.001). However, the BIS has obvious advantages over the OAA/S. The primary advantage is that the BIS is a much simpler and more continuous measure. The BIS allows objective measurements of sedation in patients undergoing endoscopic procedures[26].
The use of the BIS in monitoring the level of sedation of patients undergoing endoscopic procedures is controversial because its impact remains unclear[11,13,17,27]. In a study of patients undergoing colonoscopy, the sedation was administered by nurses under the supervision of gastroenterologists, and there was no reduction in the propofol dose used or the time to recovery[34]. Other authors have shown that BIS monitoring leads to the use of a lower mean dose of propofol in endoscopic retrograde cholangiopancreatography[23]. In a study of BIS monitoring during sedation for endoscopic submucosal dissection, the propofol dose was not reduced, although there was increased satisfaction on the part of the patients and endoscopists[25]. Nevertheless, some authors question the accuracy of BIS monitoring in detecting deep sedation[27]. There is no evidence to support the routine use of BIS monitoring in the ambulatory setting of diagnostic endoscopic procedures. In the future, it may prove beneficial for more complex therapeutic endoscopic procedures.
The present study had some limitations. The fact that physical status was classified as ASA I or II in 99.5% of the patients might have limited the external validity of the study. However, the exclusion of patients with poor physical status allowed us to focus more closely on the relationship between propofol and deep sedation in the clinical setting most often encountered in endoscopy clinics. In fact, the inclusion of patients with more comorbidity who were undergoing endoscopic procedures of greater complexity would have shifted the discussion to the efficacy and safety of sedation in complex situations. Although that is an important topic and is currently being investigated by other authors[23,35], it was not the focus of the present study. There were also some potential biases related to the difficulty of achieving full blinding of the sedation regimen. However, double blinding was achieved for the collection of data, such as the level of sedation (OAA/S and BIS), the time to induction, recovery, and discharge, and the level of patient satisfaction. All procedures were performed consecutively, respecting the sedation regimens initially proposed and with the observer present.
In the present study, we demonstrated that the times to induction, recovery, and discharge were significantly shorter in the propofol-fentanyl group. These findings replicate the results obtained by other authors who have demonstrated that propofol allows patients to resume their work activities sooner, thereby also increasing overall productivity[3,5,9,19,30,31,33,35-38]. We found no significant difference between the two regimens in terms of patient satisfaction, although there was a difference in terms of satisfaction on the part of endoscopists. The endoscopists expressed a preference for the propofol-fentanyl combination. This finding is in keeping with the global trend toward the use of propofol sedation by gastroenterologists and endoscopists[2,3].
Our findings show that although the use of the midazolam-fentanyl regimen results in deep sedation less often than the propofol-fentanyl regimen does, the difference is not clinically relevant. In our opinion, there is little evidence to support the position that propofol should be administered only by anesthesiologists or that the use of propofol is disproportionately associated with the occurrence of unwanted deep sedation. In fact, deep sedation can also occur when the midazolam-fentanyl regimen is used. This underscores the importance of monitoring the vital signs of patients under sedation. We could also add that both drug dosage and titration are crucial for the success of the sedation regimen.
In our opinion, patients classified as ASA I or II, if properly evaluated and monitored, can be safely subjected to diagnostic upper gastrointestinal endoscopy under sedation with the propofol-fentanyl combination at doses targeting conscious sedation. We also believe that the presence of an anesthesiologist is not mandatory in this setting. The use of this regimen can increase physician satisfaction and productivity.
The routine use of sedation during endoscopic procedures is increasing worldwide. The combination of a benzodiazepine (e.g., midazolam) and opioid (e.g., fentanyl) is reportedly used at approximately 75% of all healthcare facilities in the United States and considered the combination of choice by most endoscopists worldwide. Propofol is a hypnotic agent and its use has been adopted at endoscopy centers worldwide. However, its use has also been associated with deep sedation. In this study, authors evaluated the use of propofol-fentanyl versus midazolam-fentanyl for sedation of patients undergoing upper gastrointestinal endoscopy, comparing the frequency of deep sedation in each group. They also compared the two drug combinations in terms of time to induction, time to recovery, time to discharge, efficacy, and safety, as well as patient and endoscopist satisfaction.
Nowadays, propofol is only used routinely by anesthesiologists. The research hotspots are to find a secure way to provide safety and lower complications of propofol use by non-anesthesiologists.
Although there are abundant data in the literature and various guidelines on sedation during endoscopy, few studies have compared propofol with midazolam for conscious sedation in patients undergoing diagnostic upper gastrointestinal endoscopy. In the present study, authors used the bispectral index (BIS) monitor and observer’s assessment of alertness/sedation (OAA/S) scale in an innovative manner and assessed the frequency of deep sedation events for two sedation regimens frequently used during endoscopy.
The results suggest that patients classified as low anesthetic risk, if properly evaluated and monitored, can be safely subjected to diagnostic upper gastrointestinal endoscopy under sedation with the propofol-fentanyl combination at doses targeting conscious sedation. Authors also believe that the presence of an anesthesiologist is not mandatory in this setting. The use of this regimen can increase physician satisfaction and productivity.
Deep sedation is represented by a BIS ≤ 65 and/or OAA/S scale = 1. The BIS monitors cerebral activity. BIS ≤ 65 indicates deep sedation, 66-85 indicates conscious sedation, and > 85 indicates mild sedation. OAA/S scale was developed to measure the level of alertness in subjects who are sedated. It is scored as 1 for deep sedation, 2-4 for conscious sedation, and 5 for mild sedation.
An interesting study that assessed the differences and reliability between upper gastrointestinal endoscopies performed with midazolam-fentanyl versus propofol-fentanyl sedation. The main findings of the paper were that deep sedation was more frequent in the propofol-fentanyl group, however, this group had a more rapid recovery after the procedure.
P- Reviewers Familiari L, Galloro G, Lakatos PL S- Editor Gou SX L- Editor A E- Editor Xiong L
| 1. | McQuaid KR, Laine L. A systematic review and meta-analysis of randomized, controlled trials of moderate sedation for routine endoscopic procedures. Gastrointest Endosc. 2008;67:910-923. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 332] [Cited by in F6Publishing: 354] [Article Influence: 22.1] [Reference Citation Analysis (1)] |
| 2. | Benson AA, Cohen LB, Waye JD, Akhavan A, Aisenberg J. Endoscopic sedation in developing and developed countries. Gut Liver. 2008;2:105-112. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 3. | Cohen LB, Wecsler JS, Gaetano JN, Benson AA, Miller KM, Durkalski V, Aisenberg J. Endoscopic sedation in the United States: results from a nationwide survey. Am J Gastroenterol. 2006;101:967-974. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 331] [Cited by in F6Publishing: 332] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 4. | Thomson A, Andrew G, Jones DB. Optimal sedation for gastrointestinal endoscopy: review and recommendations. J Gastroenterol Hepatol. 2010;25:469-478. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 37] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 5. | Cohen LB, Ladas SD, Vargo JJ, Paspatis GA, Bjorkman DJ, Van der Linden P, Axon AT, Axon AE, Bamias G, Despott E. Sedation in digestive endoscopy: the Athens international position statements. Aliment Pharmacol Ther. 2010;32:425-442. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 53] [Cited by in F6Publishing: 58] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 6. | Heuss LT, Peter S. Propofol use by gastroenterologists-the European experience. Gastrointest Endosc Clin N Am. 2008;18:727-38, ix. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 13] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 7. | Riphaus A, Rabofski M, Wehrmann T. Endoscopic sedation and monitoring practice in Germany: results from the first nationwide survey. Z Gastroenterol. 2010;48:392-397. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 43] [Cited by in F6Publishing: 38] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 8. | Meining A, Semmler V, Kassem AM, Sander R, Frankenberger U, Burzin M, Reichenberger J, Bajbouj M, Prinz C, Schmid RM. The effect of sedation on the quality of upper gastrointestinal endoscopy: an investigator-blinded, randomized study comparing propofol with midazolam. Endoscopy. 2007;39:345-349. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 56] [Cited by in F6Publishing: 58] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 9. | Sipe BW, Rex DK, Latinovich D, Overley C, Kinser K, Bratcher L, Kareken D. Propofol versus midazolam/meperidine for outpatient colonoscopy: administration by nurses supervised by endoscopists. Gastrointest Endosc. 2002;55:815-825. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 227] [Cited by in F6Publishing: 226] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 10. | Fanti L, Testoni PA. Sedation and analgesia in gastrointestinal endoscopy: what’s new? World J Gastroenterol. 2010;16:2451-2457. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 18] [Cited by in F6Publishing: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 11. | American Society of Anesthesiologists Task Force on Sedation and Analgesia by Non-Anesthesiologists. Practice guidelines for sedation and analgesia by non-anesthesiologists. Anesthesiology. 2002;96:1004-1017. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1504] [Cited by in F6Publishing: 1263] [Article Influence: 57.4] [Reference Citation Analysis (0)] |
| 12. | Faigel DO, Baron TH, Goldstein JL, Hirota WK, Jacobson BC, Johanson JF, Leighton JA, Mallery JS, Peterson KA, Waring JP. Guidelines for the use of deep sedation and anesthesia for GI endoscopy. Gastrointest Endosc. 2002;56:613-617. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 142] [Cited by in F6Publishing: 127] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 13. | Lichtenstein DR, Jagannath S, Baron TH, Anderson MA, Banerjee S, Dominitz JA, Fanelli RD, Gan SI, Harrison ME, Ikenberry SO. Sedation and anesthesia in GI endoscopy. Gastrointest Endosc. 2008;68:815-826. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 266] [Cited by in F6Publishing: 271] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 14. | Dumonceau JM, Riphaus A, Aparicio JR, Beilenhoff U, Knape JT, Ortmann M, Paspatis G, Ponsioen CY, Racz I, Schreiber F. European Society of Gastrointestinal Endoscopy, European Society of Gastroenterology and Endoscopy Nurses and Associates, and the European Society of Anaesthesiology Guideline: Non-anesthesiologist administration of propofol for GI endoscopy. Endoscopy. 2010;42:960-974. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 121] [Cited by in F6Publishing: 145] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 15. | Cohen LB. Patient monitoring during gastrointestinal endoscopy: why, when, and how? Gastrointest Endosc Clin N Am. 2008;18:651-63, vii. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 16. | Carvalho PHB, Posso IP, Matuguma SE. Tratado de Endoscopia Digestiva, Diagnóstica e Terapêutica - Intestino delgado, cólon e reto. Vol. 4. Sao Paulo: Atheneu 2008; 37-48. [Cited in This Article: ] |
| 17. | Waring JP, Baron TH, Hirota WK, Goldstein JL, Jacobson BC, Leighton JA, Mallery JS, Faigel DO. Guidelines for conscious sedation and monitoring during gastrointestinal endoscopy. Gastrointest Endosc. 2003;58:317-322. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 232] [Cited by in F6Publishing: 252] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 18. | Tan G, Irwin MG. Recent advances in using propofol by non-anesthesiologists. F1000 Med Rep. 2010;2:79. [PubMed] [Cited in This Article: ] |
| 19. | Rex DK, Overley C, Kinser K, Coates M, Lee A, Goodwine BW, Strahl E, Lemler S, Sipe B, Rahmani E. Safety of propofol administered by registered nurses with gastroenterologist supervision in 2000 endoscopic cases. Am J Gastroenterol. 2002;97:1159-1163. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 133] [Cited by in F6Publishing: 153] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 20. | Patel S, Vargo JJ, Khandwala F, Lopez R, Trolli P, Dumot JA, Conwell DL, Zuccaro G. Deep sedation occurs frequently during elective endoscopy with meperidine and midazolam. Am J Gastroenterol. 2005;100:2689-2695. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 128] [Cited by in F6Publishing: 132] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 21. | Chernik DA, Gillings D, Laine H, Hendler J, Silver JM, Davidson AB, Schwam EM, Siegel JL. Validity and reliability of the Observer‘s Assessment of Alertness/Sedation Scale: study with intravenous midazolam. J Clin Psychopharmacol. 1990;10:244-251. [PubMed] [Cited in This Article: ] |
| 22. | DeWitt JM. Bispectral index monitoring for nurse-administered propofol sedation during upper endoscopic ultrasound: a prospective, randomized controlled trial. Dig Dis Sci. 2008;53:2739-2745. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 23. | Paspatis GA, Chainaki I, Manolaraki MM, Vardas E, Theodoropoulou A, Tribonias G, Konstantinidis K, Karmiris K, Chlouverakis G. Efficacy of bispectral index monitoring as an adjunct to propofol deep sedation for ERCP: a randomized controlled trial. Endoscopy. 2009;41:1046-1051. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 46] [Cited by in F6Publishing: 45] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 24. | Bower AL, Ripepi A, Dilger J, Boparai N, Brody FJ, Ponsky JL. Bispectral index monitoring of sedation during endoscopy. Gastrointest Endosc. 2000;52:192-196. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 96] [Cited by in F6Publishing: 100] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 25. | Imagawa A, Fujiki S, Kawahara Y, Matsushita H, Ota S, Tomoda T, Morito Y, Sakakihara I, Fujimoto T, Taira A. Satisfaction with bispectral index monitoring of propofol-mediated sedation during endoscopic submucosal dissection: a prospective, randomized study. Endoscopy. 2008;40:905-909. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 52] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 26. | Moses PL, Vargo JJ, Mitty RD, Pleskow DK, Walker JA, Rex DK. BIS values correlate with clinical sedation scores during midazolam/narcotic or propofol sedation for endoscopy. Gastrointest Endosc. 2004;59:130. [DOI] [Cited in This Article: ] |
| 27. | Qadeer MA, Vargo JJ, Patel S, Dumot JA, Lopez AR, Trolli PA, Conwell DL, Stevens T, Zuccaro G. Bispectral index monitoring of conscious sedation with the combination of meperidine and midazolam during endoscopy. Clin Gastroenterol Hepatol. 2008;6:102-108. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 40] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 28. | Dinis-Ribeiro M, Vargo JJ. Sedation by non-anesthesiologists: are opioids and benzodiazepines outdated? Digestion. 2010;82:100-101. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 4] [Cited by in F6Publishing: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 29. | Chen SC, Rex DK. Review article: registered nurse-administered propofol sedation for endoscopy. Aliment Pharmacol Ther. 2004;19:147-155. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 39] [Cited by in F6Publishing: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 30. | Cohen LB, Hightower CD, Wood DA, Miller KM, Aisenberg J. Moderate level sedation during endoscopy: a prospective study using low-dose propofol, meperidine/fentanyl, and midazolam. Gastrointest Endosc. 2004;59:795-803. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 157] [Cited by in F6Publishing: 157] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 31. | Vargo JJ, Cohen LB, Rex DK, Kwo PY. Position statement: nonanesthesiologist administration of propofol for GI endoscopy. Gastrointest Endosc. 2009;70:1053-1059. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 90] [Cited by in F6Publishing: 81] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 32. | de Azevedo MP. Sedação e anestesia em endoscopia digestiva. Medicina perioperatória. 2005;81:709-724. [Cited in This Article: ] |
| 33. | Available from: http: //www.portalmedico.org.br/resolucoes/cfm. [Cited in This Article: ] |
| 34. | Drake LM, Chen SC, Rex DK. Efficacy of bispectral monitoring as an adjunct to nurse-administered propofol sedation for colonoscopy: a randomized controlled trial. Am J Gastroenterol. 2006;101:2003-2007. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 66] [Cited by in F6Publishing: 66] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 35. | Correia LM, Bonilha DQ, Gomes GF, Brito JR, Nakao FS, Lenz L, Rohr MR, Ferrari AP, Libera ED. Sedation during upper GI endoscopy in cirrhotic outpatients: a randomized, controlled trial comparing propofol and fentanyl with midazolam and fentanyl. Gastrointest Endosc. 2011;73:45-51, 51.e1. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 48] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 36. | Cohen LB, Dubovsky AN, Aisenberg J, Miller KM. Propofol for endoscopic sedation: A protocol for safe and effective administration by the gastroenterologist. Gastrointest Endosc. 2003;58:725-732. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 108] [Cited by in F6Publishing: 119] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 37. | Rex DK. Review article: moderate sedation for endoscopy: sedation regimens for non-anaesthesiologists. Aliment Pharmacol Ther. 2006;24:163-171. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 61] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 38. | Cohen LB, Delegge MH, Aisenberg J, Brill JV, Inadomi JM, Kochman ML, Piorkowski JD. AGA Institute review of endoscopic sedation. Gastroenterology. 2007;133:675-701. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 280] [Cited by in F6Publishing: 309] [Article Influence: 18.2] [Reference Citation Analysis (0)] |