Published online Nov 14, 2011. doi: 10.3748/wjg.v17.i42.4711
Revised: June 23, 2011
Accepted: June 30, 2011
Published online: November 14, 2011
AIM: To identify differentially expressed microRNAs (miRNAs) in human colon cancer stem cells (SW1116csc) and study their function in SW1116csc proliferation.
METHODS: SW1116csc were isolated from the human colon cancer cell line, SW1116 and cultured in serum-free medium. A miRNA microarray was used to detect differential expression profiles of miRNAs in SW1116csc and SW1116 cells. Real-time quantitative polymerase chain reaction (PCR) was performed to verify the differential expression of candidate miRNAs obtained from the microarray. Target mRNAs of differentially expressed miRNAs were predicted with target prediction tools. miRNA expression plasmids were transfected into SW1116csc using Lipofectamine 2000 reagent. Cell proliferation curves were generated with trypan blue staining, and the colony formation rate of transfected cells was measured with the soft agar colony formation assay. Expression of target mRNAs and proteins from differentially expressed miRNAs were detected using reverse transcription (RT)-PCR and western blotting.
RESULTS: Compared with expression in SW1116 cells, 35 miRNAs (including hsa-miR-192, hsa-miR-29b, hsa-miR-215, hsa-miR-194, hsa-miR-33a and hsa-miR-32) were upregulated more than 1.5-fold, and 11 miRNAs (including hsa-miR-93, hsa-miR-1231, hsa-miRPlus-F1080, hsa-miR-524-3p, hsa-miR-886-3p and hsa-miR-561) were downregulated in SW1116csc. The miRNA microarray results were further validated with quantitative RT-PCR. miR-93 was downregulated, and its predicted mRNA targets included BAMBI, CCND2, CDKN1A, HDAC8, KIF23, MAP3K9, MAP3K11, MYCN, PPARD, TLE4 and ZDHHC1. Overexpressed miR-93 significantly inhibited cell proliferation and colony formation by SW1116csc. Furthermore, miR-93 negatively regulated the mRNA and protein levels of HDAC8 and TLE4.
CONCLUSION: Some miRNAs were differentially expressed during differentiation of SW1116csc into SW1116 cells. miR-93 may inhibit SW1116csc proliferation and colony formation.
- Citation: Yu XF, Zou J, Bao ZJ, Dong J. miR-93 suppresses proliferation and colony formation of human colon cancer stem cells. World J Gastroenterol 2011; 17(42): 4711-4717
- URL: https://www.wjgnet.com/1007-9327/full/v17/i42/4711.htm
- DOI: https://dx.doi.org/10.3748/wjg.v17.i42.4711
Cancer stem cells (CSC) are a sub-population of cancer cells that possess characteristics associated with normal stem cells, such as self renewal and the ability to differentiate into multiple cell types. CSCs are tumorigenic, in contrast to most cancer cells, which are thought to be non-tumorigenic. The CSC hypothesis infers that if CSCs were eliminated, the tumor would simply regress due to differentiation and cell death. Since the identification and characterization of CSCs in hematological malignancies, an increasing number of studies have described CSCs in solid tumors such as ovarian[1], colon[2], lung[3], breast[4], liver[5], melanoma[6] and pancreatic[7] tumors, raising the possibility that the CSC hypothesis applies to most neoplastic systems. CSCs are the most critical tumor cell type because they are capable of self renewing, differentiating, and maintaining tumor growth and heterogeneity, and thus play an important role in tumorigenesis and therapeutics.
MicroRNAs (miRNAs) are small noncoding RNAs that posttranscriptionally regulate gene expression. Mature miRNAs can specifically bind to 3’ untranslated regions of target cellular mRNA, in turn triggering mRNA degradation or inhibition of translation[8]. To date, thousands of miRNAs have been identified in the human genome, where they act as key regulators of a wide variety of biological processes including development, cell differentiation, apoptosis, metabolism, and signal transduction[9]. Consequently, abnormal patterns of miRNAs have been found in various human diseases, most notably cancer[10]. Recent findings indicate that alterations in the expression of several miRNAs are present in human cancers, suggesting potential roles in carcinogenesis[11]. Expression of some miRNAs, such as let-7 in human lung cancers[12], the miR-15a/miR-16-1 cluster in chronic lymphocytic leukemia[13], and neighboring miR-143/miR-145 in colorectal neoplasia and breast cancer[14,15], is reduced, suggesting potential tumor suppressor activity. In contrast, other miRNAs, such as the miR-17-92 cluster in human B-cell lymphomas[16] and miR-155/BIC in Hodgkin lymphoma[17], are overexpressed, suggesting oncogenic potential.
miRNAs are emerging as important regulators of cellular differentiation and proliferation and have been implicated in the etiology of a variety of cancers. However, the role of miRNAs in human colon cancer stem cells remains poorly understood. In this study, we screened and identified differential miRNA expression profiles in colon cancer stem cells using a miRNA microarray and studied the function of differentially expressed miRNAs in the proliferation of colon cancer stem cells.
The human colon cancer cell line (SW1116) was purchased from Cell Bank, Shanghai Institute of Life Science, Chinese Academy of Sciences and maintained in RPMI-1640 medium (Invitrogen, Carlsbad, CA, United States) supplemented with 10% fetal bovine serum (Gibco BRL, United States), penicillin G (1 × 105 U/L), and streptomycin (100 mg/L) in a 50 mL/L CO2 atmosphere at 37 °C. SW1116csc (Figure 1) were isolated previously[18] and maintained in serum-free DMEM/F12 medium supplemented with human recombinant epidermal growth factor (20 μg/L; Invitrogen), human recombinant basic fibroblast growth factor (20 μg/L; Invitrogen), L-glutamine (2 mmol/L), insulin (4 U/L), penicillin G (1 × 105 U/L), and streptomycin (100 mg/L).
Total RNA was extracted from SW1116csc and SW1116 cells using Trizol reagent (Invitrogen). The quantity was measured on a spectrophotometer (Ultraspec 2000, Pharmacia Biotech), and the integrity of the RNA was checked on a 1% agarose gel. Low-molecular-weight RNA (< 200 nt) was separated from the total RNA using mirVana miRNA purification columns (Ambion, Austin, TX, United States) for miRCURY™ array microarray (v.13.0) (Exiqon) analysis according to the manufacturer’s protocol, which uses the LNA probe to detect miRNA expression. LNA is a high-affinity RNA analog with a bicyclic furanose unit locked in an RNA-mimicking sugar conformation, which results in unprecedented hybridization affinity toward complementary single-stranded RNA molecules. This makes LNA-modified DNA probes ideally suited for RNA targeting. Microarray images were analyzed using an Axon GenePix 4000B microarray scanner (Molecular Devices), and GenePix pro V6.0 (Molecular Devices) was used to read the raw intensity of the image. Average values of the replicate spots of each miRNA were background subtracted, normalized, and subjected to further analysis. Normalization was performed using the signal of U6 snRNA on the chip, and the cutoff value was set to 1000.
For polymerase chain reaction (PCR) of miRNAs, cDNA synthesis was performed with 100 ng total RNA using a SuperScript III Reverse Transcriptase kit (Invitrogen) according to the manufacturer’s protocol. The sequences of primers and probes specific for individual miRNAs and the U6 RNA internal control are shown in Table 1. PCR consisted of denaturation at 95 °C followed by 40 cycles of 95 °C for 15 s, 60 °C for 30 s, and 72 °C for 3 s. Thermal cycling and fluorescent monitoring were performed on an ABI 7700 sequence analyzer (PE Biosystems). Relative expression (RE) of the sample gene was calculated using the ΔΔCT method using the formula RE = 2ΔΔCT where CT = PCR cycle in which the sample fluorescent intensity exceeds that of background, ΔCT sample = CT sample - CT U6 sample, ΔCT control = CT control - CT U6 control, and ΔΔCT = ΔCT sample - ΔCT control.
| Primer | Primer sequence (5'→3') |
| hsa-miR-93-RT | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGCTACCT |
| hsa-miR-93-F | ACACTCCAGCTGGGCAAAGTGCTGTTCGTGC |
| hsa-miR-1231-RT | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGGCAGCT |
| hsa-miR-1231-F | ACACTCCAGCTGGGGTGTCTGGGCGGAC |
| hsa-miR-32-RT | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGTGCAAC |
| hsa-miR-32-F | ACACTCCAGCTGGGTATTGCACATTACTAA |
| hsa-miR-33a-RT | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGTGCAAT |
| hsa-miR-33a-F | ACACTCCAGCTGGGGTGCATTGTAGTTGC |
| hsa-miR-194-RT | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGTCCACA |
| hsa-miR-194-F | ACACTCCAGCTGGGTGTAACAGCAACTCCA |
| hsa-miR-215-RT | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGGTCTGT |
| hsa-miR-215-F | ACACTCCAGCTGGGATGACCTATGAATTG |
| hsa-miR-29b-RT | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGAACACT |
| hsa-miR-29b-F | ACACTCCAGCTGGGTAGCACCATTTGAAATC |
| hsa-miR-192-RT | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGGGCTGT |
| hsa-miR-192-F | ACACTCCAGCTGGGCTGACCTATGAATTG |
| URP | TGGTGTCGTGGAGTCG |
| U6 | F-CTCGCTTCGGCAGCACA R-AACGCTTCACGAATTTGCGT |
A list of potential miRNA targets was created by combining predicted targets from the mirBase, TargetScan, miRanda, and PicTar target prediction algorithms. Potential targets were chosen based on gene function, the number of predicted target sites, and target prediction by multiple algorithms.
The human miR-93 precursor (CUGGGGGCUCCAAAGUGCUGUUCGUGCAGGUAGUGUGAUUACCCAACCUACUGCUGAGCUAG CACUUCCCGAGCCCCCGG) was reverse-transcribed and cloned into pSilencer 4.1 (Ambion). The correct plasmid was named pS-miR-93, and the control plasmid (pS-Neg) consisted of a scrambled sequence (Ambion). The sequence of the pS-miR-93 plasmid was confirmed with DNA sequencing. SW1116csc were seeded at 1 × 105 cells/well in a 12-well plate (BD Bioscience). After 48 h, the cells were transfected with 1 μg/well of pS-miR-93 or pS-Neg using 4 μL Lipofectamine 2000 reagent (Invitrogen) per well according to the manufacturer’s protocol. After 24 h, cells were harvested, and total mRNAs and proteins were extracted.
SW1116csc and pS-miR-93-transfected SW1116csc (SWt) cells were seeded at a density of 1 × 104 in 35-mm Petri dishes. Cultured cells stained with trypan blue were observed and counted in triplicate over 6 wk.
Cells were disassociated, suspended in medium containing 0.3% agar and 10% serum, and plated onto a bottom layer containing 0.6% agar. The cells were plated at a density of 3 × 104 cells/6-cm dish, and the number of colonies > 0.5 mm in diameter was counted 14 d later.
Total RNA was extracted, and cDNA was synthesized as above. PCR was performed in a 50-μL volume containing 1 μL Taq DNA polymerase (Hua Mei Co.), 5 μL 10× buffer, 1 μL dNTPs (10 mmol/L), 2 μL primers (10 μmol/L), and 1 μL cDNA (0.1 μg/μL). Amplification was performed in a thermal cycler (Perkin-Elmer Co., United States). The PCR products were analyzed and photographed with a gel documentation system (FR-200, Shanghai Fu Ri Bio Co.). For HDAC8, the forward primer used was F: 5’-TGGGCAGTCGCTGGT-3’, and the reverse primer was R: 5’-GTGGCTGGGCAGTCATAA-3’ (product size: 285 bp). For TLE4, F: 5’-ACTCCCACTGCTGTGCAAG-3’, R: 5’-GTTTCTGGCACAATGCACAG-3’ (194 bp). For glyceraldehyde-3-phosphate dehydrogenase (GAPDH), F: 5’-TTGGTATCGTGGAAGGACTCA-3’, R: 5’-TGTCATCATATTTGGCAGGTT-3’ (270 bp). RT-PCR primers were synthesized by Shanghai Sangon Co.
Cell extracts were prepared in lysis buffer (20 mmol/L Tris-HCl, pH 7.5, 0.1% Triton X-100, 0.5% sodium deoxycholate, 1 mmol/L phenylmethylsulfonyl fluoride, 10 µg/mL aprotinin, and 10 µg/mL leupeptin) and centrifuged at 12 000 ×g at 4 °C. The total protein concentration was measured using a BCA assay. Cellular extracts containing 50 μg total proteins were subjected to 10% SDS-PAGE and transferred electrophoretically to polyvinylidene difluoride membranes (Invitrogen). Blots were probed at 4 °C overnight with primary antibodies in 5% milk/TBST. The antibodies for western blotting were HDAC8, TLE4 and GAPDH (Santa Cruz Biotechnology, Santa Cruz, CA).
All data are presented as the mean ± SE. SPSS for Windows (version 15.0, SPSS Inc., United States) was used for statistical analysis. P values < 0.05 were considered statistically significant.
We used an miRNA microarray to evaluate miRNA expression profiles of colon cancer stem cells (SW1116csc) and differentiated colon cancer cells (SW1116). Analysis of data derived from the miRCURY™ array microarray indicated that compared with SW1116 cells, there were 46 differentially expressed miRNAs in SW1116csc. Among them, 35 miRNAs (including hsa-miR-192, hsa-miR-29b, hsa-miR-215, hsa-miR-194, hsa-miR-33a and hsa-miR-32) were overexpressed more than 1.5-fold and 11 miRNAs (including hsa-miR-93, hsa-miR-1231, hsa-miRPlus-F1080, hsa-miR-524-3p, hsa-miR-886-3p and hsa-miR-561) were downregulated (Table 2). Expression of miR-93 in SW1116csc was decreased by 16.7 times, and we chose this miRNA for further study.
| miRNA name | SW1116csc | SW1116 | Fold |
| image intensity | image intensity | change | |
| hsa-miR-93 | 0.46 | 8.13 | 0.06 |
| hsa-miR-1231 | 0.02 | 0.08 | 0.22 |
| hsa-miRPlus-F1080 | 0.01 | 0.05 | 0.23 |
| hsa-miR-524-3p | 0.05 | 0.16 | 0.34 |
| hsa-miR-886-3p | 3.34 | 8.37 | 0.40 |
| hsa-miR-561 | 0.02 | 0.04 | 0.45 |
| hsa-miR-497 | 0.01 | 0.03 | 0.51 |
| hsa-miR-23a | 11.92 | 22.51 | 0.53 |
| hsa-miR-886-5p | 2.89 | 5.18 | 0.56 |
| hsa-miRPlus-A1087 | 0.47 | 0.79 | 0.59 |
| hsa-miRPlus-E1170 | 30.71 | 49.93 | 0.61 |
| hsa-miRPlus-E1102 | 0.42 | 0.28 | 1.50 |
| hsa-miR-138-2 | 0.39 | 0.26 | 1.50 |
| hsa-miR-31 | 0.45 | 0.30 | 1.52 |
| hsa-miR-17 | 2.50 | 1.64 | 1.52 |
| hsa-miR-374a | 5.40 | 3.55 | 1.52 |
| hsa-miR-424 | 3.38 | 2.21 | 1.53 |
| hsa-miRPlus-F1181 | 0.64 | 0.42 | 1.53 |
| hsa-miRPlus-E1238 | 0.28 | 0.18 | 1.54 |
| hsa-miR-542-3p | 0.38 | 0.24 | 1.56 |
| hsa-miR-582-3p | 0.27 | 0.17 | 1.57 |
| hsa-miR-584 | 0.28 | 0.17 | 1.61 |
| hsa-miR-522 | 0.29 | 0.18 | 1.64 |
| hsa-miR-590-5p | 0.41 | 0.25 | 1.67 |
| hsa-miR-487b | 2.88 | 1.72 | 1.68 |
| hsa-miR-29c | 1.53 | 0.89 | 1.72 |
| hsa-miR-96 | 2.39 | 1.34 | 1.78 |
| hsa-miR-193a-3p | 22.24 | 12.50 | 1.78 |
| hsa-miR-20a | 0.74 | 0.41 | 1.79 |
| hsa-miR-301a | 2.28 | 1.27 | 1.80 |
| hsa-miRPlus-E1106 | 0.56 | 0.31 | 1.82 |
| hsa-miR-30e | 1.21 | 0.65 | 1.86 |
| hsa-miR-874 | 0.20 | 0.10 | 1.99 |
| hsa-miR-19a | 17.38 | 8.71 | 2.00 |
| hsa-miR-519a | 1.73 | 0.77 | 2.24 |
| hsa-miRPlus-A1065 | 0.30 | 0.13 | 2.32 |
| hsa-miR-521 | 0.94 | 0.40 | 2.36 |
| hsa-miR-876-5p | 0.23 | 0.10 | 2.37 |
| hsa-miR-493 | 0.28 | 0.11 | 2.53 |
| hsa-miR-101 | 4.92 | 1.28 | 3.85 |
| hsa-miR-32 | 1.89 | 0.35 | 5.48 |
| hsa-miR-33a | 3.97 | 0.64 | 6.16 |
| hsa-miR-194 | 0.79 | 0.11 | 7.11 |
| hsa-miR-215 | 0.97 | 0.11 | 9.09 |
| hsa-miR-29b | 3.00 | 0.33 | 9.20 |
| hsa-miR-192 | 1.47 | 0.14 | 10.67 |
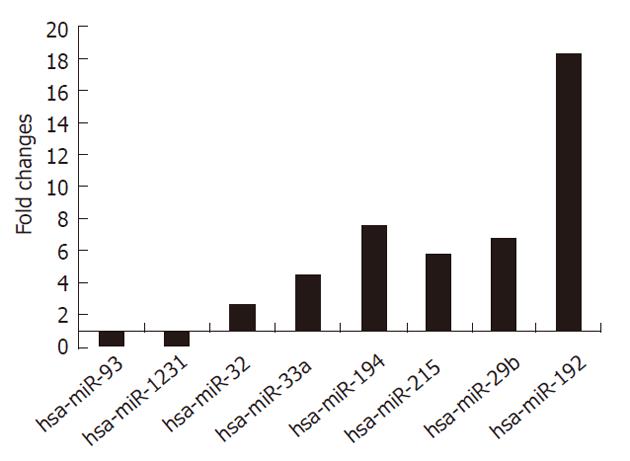
To validate our microarray results, we performed quantitative PCR with some of the miRNAs that were differentially expressed in SW1116csc according to the microarray data. We selected miR-93, miR-1231, miR-32, miR-33a, miR-194, miR-215, miR-29b and miR-192 for quantitative PCR. The quantitative PCR results indicated that the expression of miR-93 and miR-1231 was decreased, whereas the expression of miR-32, miR-33a, miR-194, miR-215, miR-29b and miR-192 was significantly increased in SW1116csc cells (Figure 2). The quantitative PCR result was in agreement with that of the microarray.
By combining predicted targets generated with the mirBase, TargetScan, miRanda and PicTar target prediction algorithms, potential targets of miR-93 were chosen. The stem relevant mRNA targets included BAMBI, CCND2, CDKN1A, HDAC8, KIF23, MAP3K9, MAP3K11, MYCN, PPARD, TLE4, and ZDHHC1.
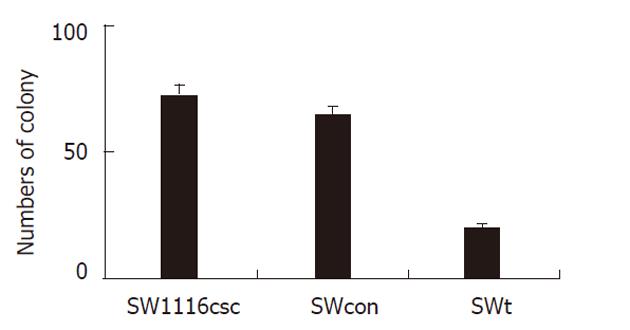
We tested for differences in the proliferation rate between SW1116csc, pS-Neg-transfected SW1116csc (SWcon), and pS-miR-93-transfected SW1116csc (SWt). The cells were examined from week 1 to week 7 after seeding. As shown in Figure 3, there was a difference in the growth rate between SWcon and SWt cells. SWt cells grew slowly and showed growth inhibition after week 3. The self-renewing capacity of SWt cells was also examined with the colony formation assay. When plated at a density of 100 cells/well, SW1116csc and SWcon cells generated a greater mean number of tumor spheres (72.3 ± 4.2 and 64.5 ± 3.6, respectively) than did SWt cells (19.6 ± 2.1) (Figure 4).
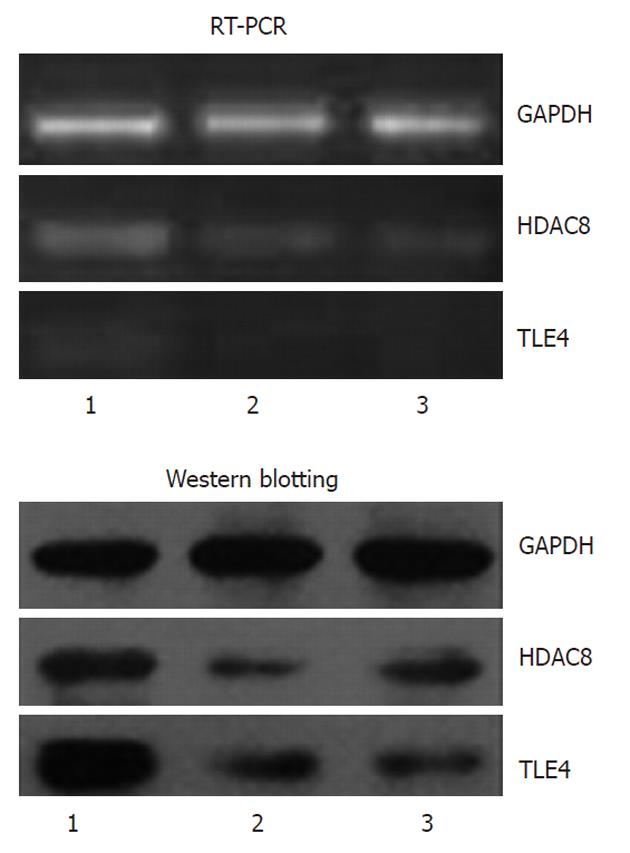
To provide additional evidence for the role of miR-93 in inhibition of SW1116csc proliferation and colony formation, we examined the effect of miR-93 on the expression of HDAC8 and TLE4. As shown in Figure 5, miR-93 mimics significantly attenuated the mRNA and protein levels of HDAC8 and TLE4.
Mature functional miRNAs of approximately 22 nucleotides that are generated from long primary miRNA transcripts control gene expression at the posttranscriptional level by degrading or repressing target mRNAs. Some miRNAs aberrantly expressed in cancer have been well documented[19,20]. These miRNAs regulate the expression of signaling molecules such as cytokines, growth factors, transcription factors, and proapoptotic and antiapoptotic molecules. Recently, miRNAs were found to play a role in the differentiation of stem cells. Proper regulation of differentiation of stem cells is crucial to normal development and the avoidance of cancer[21]. However, the differential expression of miRNAs in human colon cancer stem cells has not been addressed.
Here we analyzed the differential expression of miRNAs in human colon cancer stem cells and differentiated colon cancer cells. We identified 46 differentially expressed miRNAs in SW1116csc. We observed miRNAs that were upregulated and others that were downregulated. The miR-93 expression level was significantly lower in SW1116csc than in SW1116 cells, indicating that miR-93 may be involved in the development or replication of colon CSCs.
One of the key characteristics of stem cells is their ability to divide for long periods of time when most other cells are quiescent[22]. Because the function of miR-93 in colon CSCs was unknown, we identified the biological effects of miR-93 on colon CSCs when its expression in these cells was upregulated by examining the role of miR-93 on cellular growth and proliferation of transfected SW1116csc. Overexpression of miR-93 consistently inhibited cell proliferation and colony formation of SW1116csc. To the best of our knowledge, this study is the first to demonstrate the differential expression of miRNAs in colon CSCs and the effect of miR-93 on colon CSCs. However, the underlying mechanisms of these effects are not completely understood. Using miRNA target prediction tools and RT-PCR, we found that inhibition of overexpressed miR-93 probably affects cell proliferation and colony formation of colon cancer stem cells by targeting HDAC8 and TLE4.
Histone deacetylase (HDAC) enzymes are a family of proteins with complex, multifunctional roles in vivo, including transcriptional regulation, regulation of tubulin and cytoskeletal function, control of cardiac cell growth, regulation of thymocyte development, and facilitation of DNA repair[23]. HDAC enzymes function in part to control the acetylation state of nucleosomal histones, thereby regulating transcription. More recently, however, it has been shown that HDAC enzymes have many non-histone acetylation targets as well, including tubulin, heat-shock proteins, and a variety of transcription factors such as p53 and NF-kB subunit p65[24,25].
HDAC8 is a newly identified HDAC that was cloned and characterized in 2000. It is a novel marker of smooth muscle differentiation and is expressed at low levels in normal white blood cells but overexpressed in some malignant hematological cell lines[26]. cDNA microarray analysis suggests that there is differential expression of HDAC8 between mammary tumors and normal lactating mammary glands and that it may play a key regulatory role in mammary gland tumorigenesis[27]. Here we found that the expression level of HDAC8 in human colon cancer cells was high. Thus, accumulating data increase our understanding of the role of abnormally elevated HDAC8 activity in the pathogenesis of tumors.
Groucho (Gro) or Transducin-like Enhancer of Split (TLE) proteins constitute a family of highly conserved cofactors for transcription. They act as non-DNA-binding corepressors and are recruited to promoters via interaction with a DNA binding partner. Gro corepressor proteins interact with multiple transcription factors and thus affect different signaling pathways[28]. They contact histones and recruit HDACs, thereby altering local chromatin structure. Gro corepressor proteins confer repressing functions on binding partners with an activating potential[29].
The Gro/TLE family of corepressors interacts with at least five families of transcription factors and plays critical roles in Drosophila and vertebrate development. During B-lymphocyte differentiation, TLEs mediate the repressive effect of Pax5 via recruitment by Pu.1, limiting alternative cell fates[30]. However, the ability of Gro/TLEs to interact with other signaling pathways suggests a potentially broader role for Gro/TLEs in both normal and malignant hematopoiesis. Members of the Gro/TLE family of corepressors bind to all known Tcf/LEF complexes and act as inhibitors of Wnt/β-catenin signaling[31,32], a pathway implicated in expansion and self-renewal of the hematopoietic stem cell compartment[33]. Similarly, Gro/TLEs inhibit NF-κB signaling[34], a pathway constitutively activated in acute myeloblastic leukemia and thought to play an important role in hematopoietic cell proliferation, survival, and chemoresistance. Gro/TLE gene family members are also key effectors of Notch signaling, a pathway implicated in HSC fate determination and self-renewal[35]. The high expression of TLE4 in human colon CSCs indicates interactions with Wnt, NF-κB, or Notch signaling and suggests that TLE4 may be involved in the proliferation and differentiation of colon CSCs.
In summary, our study suggests that during the course of colon CSC differentiation towards colon cancer cells, some miRNAs are differentially expressed. miR-93 is one of the miRNAs that was downregulated, and overexpressed miR-93 significantly inhibited cell proliferation and colony formation by colon CSCs. Furthermore, overexpression of miR-93 negatively regulated mRNA and protein expression of HDAC8 and TLE4. The inhibition of cell proliferation by miR-93 in colon CSCs may occur by targeting HDAC8 and TLE4.
The cancer stem cell (CSC) hypothesis is currently at the center of a rapidly evolving field, involving a change of perspective on the development and treatment of cancers. However, research has been hampered by the lack of distinct molecular markers of CSCs.
Since the identification and characterization of CSCs in hematological malignancies, an increasing number of studies have described CSCs in solid tumors such as ovarian, colon, lung, breast, liver, melanoma and pancreatic tumors, suggesting that the CSC hypothesis applies to most neoplastic systems.
Previously isolated SW1116csc were compared with SW1116 cells. The authors identified 35 miRNAs that were upregulated and 11 miRNAs that were downregulated in SW1116csc. Overexpressed miR-93 significantly inhibited cell proliferation and colony formation of SW1116csc. Furthermore, miR-93 negatively regulated the mRNA and protein expression levels of HDAC8 and TLE4.
The study of CSCs has important implications for future cancer treatment and therapies. The CSC hypothesis states that if the CSCs were eliminated, the tumor would simply regress due to differentiation and cell death. By selectively targeting CSCs, it may be possible to treat patients with aggressive, non-resectable tumors and prevent the tumor from metastasizing.
CSCs are a sub-population of cancer cells that possess characteristics associated with normal stem cells, such as self renewal and the ability to differentiate into multiple cell types. CSCs are tumorigenic, in contrast to most cancer cells, which are thought to be non-tumorigenic. CSCs persist in tumors as a distinct population and cause relapse and metastasis by giving rise to new tumors.
The authors presented an original work about colon CSCs and identified a potentially new pathway that could be targeted for colon cancer management.
Peer reviewer: Dr. Jose Perea, Department of Surgery, Rosas De Aravaca, 82a, Madrid 28023, Spain
S- Editor Yang XC L- Editor Logan S E- Editor Xiong L
| 1. | Peng S, Maihle NJ, Huang Y. Pluripotency factors Lin28 and Oct4 identify a sub-population of stem cell-like cells in ovarian cancer. Oncogene. 2010;29:2153-2159. [PubMed] [Cited in This Article: ] |
| 2. | Ricci-Vitiani L, Lombardi DG, Pilozzi E, Biffoni M, Todaro M, Peschle C, De Maria R. Identification and expansion of human colon-cancer-initiating cells. Nature. 2007;445:111-115. [PubMed] [Cited in This Article: ] |
| 3. | Eramo A, Lotti F, Sette G, Pilozzi E, Biffoni M, Di Virgilio A, Conticello C, Ruco L, Peschle C, De Maria R. Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ. 2008;15:504-514. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1158] [Cited by in F6Publishing: 1212] [Article Influence: 71.3] [Reference Citation Analysis (0)] |
| 4. | Shimono Y, Zabala M, Cho RW, Lobo N, Dalerba P, Qian D, Diehn M, Liu H, Panula SP, Chiao E. Downregulation of miRNA-200c links breast cancer stem cells with normal stem cells. Cell. 2009;138:592-603. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 908] [Cited by in F6Publishing: 932] [Article Influence: 62.1] [Reference Citation Analysis (0)] |
| 5. | Ji J, Yamashita T, Budhu A, Forgues M, Jia HL, Li C, Deng C, Wauthier E, Reid LM, Ye QH. Identification of microRNA-181 by genome-wide screening as a critical player in EpCAM-positive hepatic cancer stem cells. Hepatology. 2009;50:472-480. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 397] [Cited by in F6Publishing: 436] [Article Influence: 29.1] [Reference Citation Analysis (0)] |
| 6. | Schatton T, Murphy GF, Frank NY, Yamaura K, Waaga-Gasser AM, Gasser M, Zhan Q, Jordan S, Duncan LM, Weishaupt C. Identification of cells initiating human melanomas. Nature. 2008;451:345-349. [PubMed] [Cited in This Article: ] |
| 7. | Ji Q, Hao X, Zhang M, Tang W, Yang M, Li L, Xiang D, Desano JT, Bommer GT, Fan D. MicroRNA miR-34 inhibits human pancreatic cancer tumor-initiating cells. PLoS One. 2009;4:e6816. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 516] [Cited by in F6Publishing: 522] [Article Influence: 34.8] [Reference Citation Analysis (0)] |
| 8. | Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281-297. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25833] [Cited by in F6Publishing: 26776] [Article Influence: 1338.8] [Reference Citation Analysis (0)] |
| 9. | Ambros V. The functions of animal microRNAs. Nature. 2004;431:350-355. [PubMed] [Cited in This Article: ] |
| 10. | Lu J, Getz G, Miska EA, Alvarez-Saavedra E, Lamb J, Peck D, Sweet-Cordero A, Ebert BL, Mak RH, Ferrando AA. MicroRNA expression profiles classify human cancers. Nature. 2005;435:834-838. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7124] [Cited by in F6Publishing: 7177] [Article Influence: 377.7] [Reference Citation Analysis (0)] |
| 11. | Zhang B, Pan X, Cobb GP, Anderson TA. microRNAs as oncogenes and tumor suppressors. Dev Biol. 2007;302:1-12. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1765] [Cited by in F6Publishing: 1891] [Article Influence: 105.1] [Reference Citation Analysis (0)] |
| 12. | Meng F, Henson R, Wehbe-Janek H, Smith H, Ueno Y, Patel T. The MicroRNA let-7a modulates interleukin-6-dependent STAT-3 survival signaling in malignant human cholangiocytes. J Biol Chem. 2007;282:8256-8264. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 145] [Cited by in F6Publishing: 153] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 13. | Cimmino A, Calin GA, Fabbri M, Iorio MV, Ferracin M, Shimizu M, Wojcik SE, Aqeilan RI, Zupo S, Dono M. miR-15 and miR-16 induce apoptosis by targeting BCL2. Proc Natl Acad Sci USA. 2005;102:13944-13949. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2701] [Cited by in F6Publishing: 2623] [Article Influence: 138.1] [Reference Citation Analysis (0)] |
| 14. | Iorio MV, Ferracin M, Liu CG, Veronese A, Spizzo R, Sabbioni S, Magri E, Pedriali M, Fabbri M, Campiglio M. MicroRNA gene expression deregulation in human breast cancer. Cancer Res. 2005;65:7065-7070. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2930] [Cited by in F6Publishing: 2983] [Article Influence: 157.0] [Reference Citation Analysis (0)] |
| 15. | Michael MZ, O' Connor SM, van Holst Pellekaan NG, Young GP, James RJ. Reduced accumulation of specific microRNAs in colorectal neoplasia. Mol Cancer Res. 2003;1:882-891. [PubMed] [Cited in This Article: ] |
| 16. | He L, Thomson JM, Hemann MT, Hernando-Monge E, Mu D, Goodson S, Powers S, Cordon-Cardo C, Lowe SW, Hannon GJ. A microRNA polycistron as a potential human oncogene. Nature. 2005;435:828-833. [PubMed] [Cited in This Article: ] |
| 17. | Kluiver J, Poppema S, de Jong D, Blokzijl T, Harms G, Jacobs S, Kroesen BJ, van den Berg A. BIC and miR-155 are highly expressed in Hodgkin, primary mediastinal and diffuse large B cell lymphomas. J Pathol. 2005;207:243-249. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 524] [Cited by in F6Publishing: 512] [Article Influence: 26.9] [Reference Citation Analysis (0)] |
| 18. | Zou J, Yu XF, Bao ZJ, Dong J. Proteome of human colon cancer stem cells: a comparative analysis. World J Gastroenterol. 2011;17:1276-1285. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 15] [Cited by in F6Publishing: 21] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 19. | Johnson CD, Esquela-Kerscher A, Stefani G, Byrom M, Kelnar K, Ovcharenko D, Wilson M, Wang X, Shelton J, Shingara J. The let-7 microRNA represses cell proliferation pathways in human cells. Cancer Res. 2007;67:7713-7722. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 937] [Cited by in F6Publishing: 971] [Article Influence: 57.1] [Reference Citation Analysis (0)] |
| 20. | Shi L, Cheng Z, Zhang J, Li R, Zhao P, Fu Z, You Y. hsa-mir-181a and hsa-mir-181b function as tumor suppressors in human glioma cells. Brain Res. 2008;1236:185-193. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 325] [Cited by in F6Publishing: 329] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 21. | Zwaka TP, Thomson JA. Differentiation of human embryonic stem cells occurs through symmetric cell division. Stem Cells. 2005;23:146-149. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 41] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 22. | Hatfield SD, Shcherbata HR, Fischer KA, Nakahara K, Carthew RW, Ruohola-Baker H. Stem cell division is regulated by the microRNA pathway. Nature. 2005;435:974-978. [PubMed] [Cited in This Article: ] |
| 23. | de Ruijter AJ, van Gennip AH, Caron HN, Kemp S, van Kuilenburg AB. Histone deacetylases (HDACs): characterization of the classical HDAC family. Biochem J. 2003;370:737-749. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2200] [Cited by in F6Publishing: 2217] [Article Influence: 105.6] [Reference Citation Analysis (0)] |
| 24. | Glozak MA, Sengupta N, Zhang X, Seto E. Acetylation and deacetylation of non-histone proteins. Gene. 2005;363:15-23. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1183] [Cited by in F6Publishing: 1199] [Article Influence: 63.1] [Reference Citation Analysis (0)] |
| 25. | Kim SC, Sprung R, Chen Y, Xu Y, Ball H, Pei J, Cheng T, Kho Y, Xiao H, Xiao L. Substrate and functional diversity of lysine acetylation revealed by a proteomics survey. Mol Cell. 2006;23:607-618. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1168] [Cited by in F6Publishing: 1182] [Article Influence: 65.7] [Reference Citation Analysis (0)] |
| 26. | Van den Wyngaert I, de Vries W, Kremer A, Neefs J, Verhasselt P, Luyten WH, Kass SU. Cloning and characterization of human histone deacetylase 8. FEBS Lett. 2000;478:77-83. [PubMed] [Cited in This Article: ] |
| 27. | Thakur A, Xu H, Wang Y, Bollig A, Biliran H, Liao JD. The role of X-linked genes in breast cancer. Breast Cancer Res Treat. 2005;93:135-143. [PubMed] [Cited in This Article: ] |
| 28. | Gasperowicz M, Otto F. Mammalian Groucho homologs: redundancy or specificity? J Cell Biochem. 2005;95:670-687. [PubMed] [Cited in This Article: ] |
| 29. | Cai Y, Brophy PD, Levitan I, Stifani S, Dressler GR. Groucho suppresses Pax2 transactivation by inhibition of JNK-mediated phosphorylation. EMBO J. 2003;22:5522-5529. [PubMed] [Cited in This Article: ] |
| 30. | Nutt SL, Eberhard D, Horcher M, Rolink AG, Busslinger M. Pax5 determines the identity of B cells from the beginning to the end of B-lymphopoiesis. Int Rev Immunol. 2001;20:65-82. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 76] [Cited by in F6Publishing: 92] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 31. | Huelsken J, Behrens J. The Wnt signalling pathway. J Cell Sci. 2002;115:3977-3978. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 366] [Cited by in F6Publishing: 380] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 32. | Brantjes H, Roose J, van De Wetering M, Clevers H. All Tcf HMG box transcription factors interact with Groucho-related co-repressors. Nucleic Acids Res. 2001;29:1410-1419. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 283] [Cited by in F6Publishing: 282] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 33. | Willert K, Brown JD, Danenberg E, Duncan AW, Weissman IL, Reya T, Yates JR, Nusse R. Wnt proteins are lipid-modified and can act as stem cell growth factors. Nature. 2003;423:448-452. [PubMed] [Cited in This Article: ] |
| 34. | Tetsuka T, Uranishi H, Imai H, Ono T, Sonta S, Takahashi N, Asamitsu K, Okamoto T. Inhibition of nuclear factor-kappaB-mediated transcription by association with the amino-terminal enhancer of split, a Groucho-related protein lacking WD40 repeats. J Biol Chem. 2000;275:4383-4390. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 78] [Cited by in F6Publishing: 80] [Article Influence: 3.3] [Reference Citation Analysis (0)] |