Published online Jul 21, 2005. doi: 10.3748/wjg.v11.i27.4258
Revised: January 6, 2005
Accepted: January 12, 2005
Published online: July 21, 2005
AIM: To summarize the operative experiences for giant leiomyoma of esophagus.
METHODS: Eight cases of giant esophageal leiomyoma (GEL) whose tumors were bigger than 10 cm were treated surgically in our department from June 1980 to March 2004. All of these cases received barium swallow roentgenography and esophagoscopy. Leiomyoma located in upper thirds of the esophagus in one case, middle thirds of the esophagus in five cases, lower thirds of the esophagus in two cases. Resection of tumors was performed successfully in all of these cases. Operative methods included transthoracic extramucosal enucleation and buttressing the muscular defect with pedicled great omental flap (one case), esophagectomy and esophago-gastrostomy above the arch of aorta (three cases), total esophagectomy and esophageal replacement with colon (four cases). Histological examination confirmed that all of these cases were leiomyoma.
RESULTS: All of the eight patients recovered approvingly with no mortality and resumed normal diet after operation. Vomiting during meals occurred in one patient with esophagogastrostomy, and remained 1 mo. Reflux esophagitis occurred in one patient with esophago-gastrostomy and was alleviated with medication. Thoracic colon syndrome (TCS) occurred in one patient with colon replacement at 15 mo postoperatively. No recurrence occurred in follow-up from 6 mo to 8 years.
CONCLUSION: Surgical treatment for GEL is both safe and effective. The choices of operative methods mainly depend on the location and range of lesions. We prefer to treat GEL via esophagectomy combined with esophago-gastrostomy or esophagus replacement with colon. The long-time quality of life is better in the latter.
- Citation: Cheng BC, Chang S, Mao ZF, Li MJ, Huang J, Wang ZW, Wang TS. Surgical treatment of giant esophageal leiomyoma. World J Gastroenterol 2005; 11(27): 4258-4260
- URL: https://www.wjgnet.com/1007-9327/full/v11/i27/4258.htm
- DOI: https://dx.doi.org/10.3748/wjg.v11.i27.4258
Leiomyoma is the most common benign tumor found in the esophagus but it is, however, a rare neoplasm. It is found mainly in the lower and middle thirds of the esophagus and, in most cases are single lesions[1]. Leiomyoma can occur at any age but 90% of cases occur in patients between the ages of 20 and 69 years, with peak incidence in the third to fifth decades[2]. The male-to-female ratio is approximately 2:1. About half the patients with leiomyoma are asymptomatic and malignant change is rare. Transthoracic extramucosal enucleation is a safe and effective procedure for about 96% of esophageal leiomyoma[3]. However, few of esophageal leiomyoma becomes giant-sized gradually (diameter of tumor larger than 10 cm) and show the symptoms caused by compression of tumor, obstruction of esophagus or dysfunction of cardia. The surgical treatment strategy of such GEL is not the same of common esophageal leiomyoma, especially when mucosa is damaged by leiomyoma or lesions have potential sarcomatous change. This report summarized the operative experience of GEL in our department.
Between June 1980 and March 2004, there were 8 cases of GEL whose tumors were larger than 10 cm that underwent surgical treatment in our department. These patients comprised five males and three females, whose age ranged from 25 to 58 years (mean age, 42.5 years). Duration of symptoms was from 5 to 12 years. All of the eight patients had dysphagia and complained of retrosternal pain and backache in five cases, superior belly distention in five cases, weight loss in four cases, heartburn in three cases, cough in two cases, took normal diet in two cases, semi-liquid diet in four cases, liquid diet in two cases. All of the eight patients received barium swallow roentgenography and esophagoscopy. CT scan was performed in five cases, MRI in three cases, endoscopic ultrasonography (EUS) in one case. Leiomyoma located in upper thirds of the esophagus in one case, middle thirds of the esophagus in five cases, lower thirds of the esophagus in two cases. The shapes of tumors were multinode-like in four cases, spiral-like in three cases, horseshoe-like in one case.
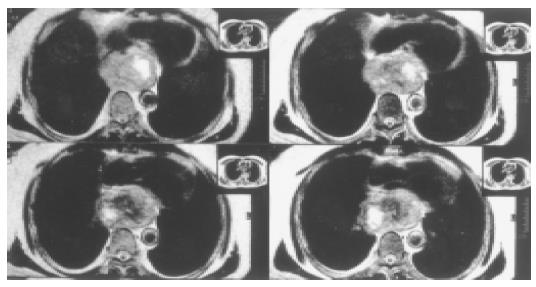
All patients received operation under general anesthesia. One patient underwent extramucosal enucleation of tumor and buttressing of the muscular defect with pedicled greater omental flap. In this case, the tumor located in lower thirds of the esophagus (Figure 1). Left posterolateral thoracotomy was performed through the sixth intercostal space. After removing the tumor, the muscular defect was found too large to allow a tension-free suture. Through a radial incision near esophageal hiatus, we made the great omentum into a pedicle flap (26 cm × 9 cm) and pulled it up into thoracic cavity to cover the muscular defect. Three patients accepted esophagectomy and esophagogastrostomy above the arch of aorta through left posterolateral thoracotomy. Nasogastric tube and plasmatic canal were placed to aid in drainage and nutrition. Another four patients underwent total esophagectomy and esophageal replacement with colon. In these cases, patients were laid in supine position at the beginning. Esophageal interposition was performed with left colon (isoperistaltic, retrosternal) through left cervical abdominal incision. Then the patient’s position was changed to full left lateral decubitus. Tumor and total esophagus were resected through right posterolateral thoracotomy. A feeding jejunostomy was used in these four patients, and enteral feeding was begun after 24 h with 5% dextrose in water and continued with an elemental diet after 48 h. The minimum size of tumor was 10 cm × 10 cm × 6 cm, the maximum was 20 cm × 15 cm × 15 cm. Histological examination confirmed that all of these were leiomyoma.
All of the eight patients recovered approvingly with no mortality and resumed normal diet after operation. Postoperative complications included leakage of cervical esophageal stoma (one case), pulmonary atelectasis (one case), thoracic infection (one case), chylothorax (one case). They all healed with conservative interventions and drainage. Patients were followed up from 6 mo to 8 years. Vomiting after meals occurred in one patient with esophagogastrostomy, and remained for 1 mo. Vomiting after meals was not seen in patients with colonic interposition. Reflux esophagitis occurred in one patient with esophagogastrostomy and was alleviated with medication. Thoracic colon syndrome (TCS) occurred in one patient with colon replacement at 15 mo postoperatively, and recovered with re-operation. No recurrence occurred.
Leiomyoma is the most common benign tumor found in the esophagus but is nonetheless rare. Of all esophageal tumors, benign tumors account for fewer than 10%, of which 4% are leiomyomas[4]. These tumors are usually found in middle age and are almost twice as common in men. They are located mainly in the lower and middle thirds of the esophagus. Leiomyoma may undergo cystic degeneration, calcification occurs infrequently and malignant change is rare[5]. Leiomyoma is a slow growing tumor and the size of the lesion may not change for many years. Approximately one-half of all tumors are smaller than 5 cm, but 5% may be larger than 10 cm. Giant leiomyoma involving the entire esophagus and weighing up to 5 kg have been reported[6]. Giant leiomyoma of esophagus should be removed when diagnosed, but its operative method is not the same of common leiomyoma.
Since most leiomyomas are intramural, eccentric, and well encapsulated, they can be easily shelled out (enucleated) without resorting to esophageal resection. One concern after enucleation of a large leiomyoma is that the myotomy may result functionally in achalasia. It is generally accepted that tumors up to 8 cm can be safely enucleated without significant postoperative dysphagia as long as the mucosa is intact and the myotomy is reapproximated[3,7]. Larger lesions can be removed via enucleation but often result in the muscular defects too large to allow a tension-free suture, requiring a tissue flap to prevent mucosal bulging. We resected a 10 cm × 10 cm × 6 cm esophageal leiomyoma successfully with extramucosal enucleation and buttressing the muscular defect with pedicled greater omental flap. No postoperative dysphagia occurred during follow-up.
The greater omentum (GO) square is large and allows the covering of various tissue defects. The pedicled GO flap has the advantages of its own blood supply and anti-inflammatory effect[14]. Using the GO to fill or buttress tissular defect is not novel in thoracic surgery[14]. Angiography shows many types of vascular supply for the GO. Therefore, the GO flap exsection technique is very important. The flap transposition through the diaphragm is not difficult. The opening should not be very wide for good great omentum tissue adhesion and too narrow to prevent compression of the vessels.
Majority of GEL may require esophageal resection. The choices of this method depend on several factors: (1) The esophageal leiomyoma is too large to allow a resection without damaging the mucosa; (2) Adhesion between leiomyoma and the esophageal mucosa is tight and extensive; (3) Unrepairable mucosal defect caused by resection of tumor; (4) Large leiomyomas locate in the distal esophagus and extend downward across the cardia; (5) Leiomyosarcoma is suspected. Resection for GEL is typically performed with esophagectomy and gastric pull-up through left posterolateral thoracotomy. In our experience, blood supply of the GEL was abundant, especially at the region posterior to the arch of aorta and upper part of the diaphragm. Careful dissection and hemostasia was necessary. Be cautious not to damage thoracic duct. For this purpose, transfixing the trunk of thoracic duct above the diaphragm between descending aorta and azygos vein was helpful. Gastric pull-up has some postoperative morbidity including reflux esophagitis, stricture formation, dumping, diarrhea, reduced meal capacity, and weight loss[8]. Many of these complications are thought to be caused by division of the vagal nerves and the subsequent loss of parasympathetic innervation to the foregut[9].
The rationale for use of the colon is that there will be fewer complications related to acid reflux than when the stomach is used for esophageal replacement. If the GEL is located in the upper thirds of the esophagus and the mucosa is damaged by GEL, esophageal replacement with colon (ERC) might be the best choice in this situation. Our techniques of ERC are different to others. The retrosternal colon replacement is performed through left cervical abdominal approaches in supine position at first, and then resects tumor and total esophagus through right posterolateral thoracotomy in full left lateral decubitus. We prefer this approach to synchronous abdominoth-oracocervical incision, for the former has allowed better exposure.
Several factors are important to decrease the mortality and postoperative complications of ERC: (1) Choosing suitable colonic segment and ensuring an adequate arterial supply. Before cutting the mesocolon, we usually clamp the communicating branches for 3~5 min in order to ensure that blood supply is enough; (2) Adopting isoperistaltic interposition. Studies show that isoperistaltic interposition is superior to antiperistaltic interposition and the long-time quality of life is better in the former[10,13]; (3) Single-layer esophagocolonic anastomosis. Our studies show that single-layer anastomosis can reduce the morbidity of fistula and stricture[11]; (4) Choosing colonic pull-up approach properly. The upward approach of colonic segment is decided by the condition of individual patient and the experience of the surgeon; (5) Avoiding redundancy of the colonic segment. Colon redundancy can be due to technical error at the time of operation (i.e., leaving redundancy in the interposition graft), intrathoracic herniation of colon, or differential colon growth. Proper attention to the length of the colon segment is important to prevent intrathoracic redundancy[16]. Yildirim et al[17], suggest that the colon is one of the best substitutes for the esophagus, and there is no need to perform a routine pyloroplasty or antireflux procedure as an adjunct to the primary surgery. Our patients healed satisfactorily and the long-time quality of life was good during follow-up and it was coincident with the literature[12,13,15].
Science Editor Guo SY Language Editor Elsevier HK
Co-first-author: Sheng Chang
| 1. | Hatch GF, Wertheimer-Hatch L, Hatch KF, Davis GB, Blanchard DK, Foster RS, Skandalakis JE. Tumors of the esophagus. World J Surg. 2000;24:401-411. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 102] [Cited by in F6Publishing: 77] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 2. | Lee LS, Singhal S, Brinster CJ, Marshall B, Kochman ML, Kaiser LR, Kucharczuk JC. Current management of esophageal leiomyoma. J Am Coll Surg. 2004;198:136-146. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 94] [Cited by in F6Publishing: 82] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 3. | Bonavina L, Segalin A, Rosati R, Pavanello M, Peracchia A. Surgical therapy of esophageal leiomyoma. J Am Coll Surg. 1995;181:257-262. [PubMed] [Cited in This Article: ] |
| 4. | Jesić R, Randjelović T, Gerzić Z, Zdravković Dj, Krstić M, Milinić N, Pavlović A, Svejić T, Bulajić M. Leiomyoma of the esophagus. Case report. Srp Arh Celok Lek. 1997;125:113-115. [PubMed] [Cited in This Article: ] |
| 5. | Nagashima R, Takeda H, Motoyama T, Tsukamoto O, Takahashi T. Coexistence of superficial esophageal carcinoma and leiomyoma: case report of an endoscopic resection. Endoscopy. 1997;29:683-684. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 14] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 6. | Aurea P, Grazia M, Petrella F, Bazzocchi R. Giant leiomyoma of the esophagus. Eur J Cardiothorac Surg. 2002;22:1008-1010. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 33] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 7. | Roviaro GC, Maciocco M, Varoli F, Rebuffat C, Vergani C, Scarduelli A. Videothoracoscopic treatment of oesophageal leiomyoma. Thorax. 1998;53:190-192. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 39] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 8. | Watson TJ, Peters JH, DeMeester TR. Esophageal replacement for end-stage benign esophageal disease. Surg Clin North Am. 1997;77:1099-1113. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 20] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 9. | Banki F, Mason RJ, DeMeester SR, Hagen JA, Balaji NS, Crookes PF, Bremner CG, Peters JH, DeMeester TR. Vagal-sparing esophagectomy: a more physiologic alternative. Ann Surg. 2002;236:324-35; discussion 335-6. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 124] [Cited by in F6Publishing: 128] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 10. | Cheng BC, Lu SQ, Gao SZ, Tu ZF, Lin DM, Wang TS. Colon replacement from esophagus. Clinical experience from 240 cases. Chin Med J (Engl). 1994;107:216-218. [PubMed] [Cited in This Article: ] |
| 11. | Gao SZ, Wang TS, Yao Z, Cheng BC, Tu ZF, Ling DM, Peng SY. Experimental study and clinical application of a single-row suturing esophagogastrostomy. J Surg Oncol. 1990;43:167-171. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 3] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 12. | Cheng BC, Gao SZ, Tu ZF, Lu DT, Zhou SG, Cai XT, Huang ZR, Wang CX. The clinical study on the route of colon seg-ment for replacement of the esophagus. Chin J Thorac Cardiovasc Surg. 2000;16:283-285. [Cited in This Article: ] |
| 13. | Cheng BC, Shao K. Evaluation of the quality of life after esophageal replacement with colon. Chin J Clin Rehabilitation. 2002;6:2676-2677. [Cited in This Article: ] |
| 14. | Levashev YN, Akopov AL, Mosin IV. The possibilities of greater omentum usage in thoracic surgery. Eur J Cardiothorac Surg. 1999;15:465-468. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 53] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 15. | Thomas P, Fuentes P, Giudicelli R, Reboud E. Colon interposition for esophageal replacement: current indications and long-term function. Ann Thorac Surg. 1997;64:757-764. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 94] [Cited by in F6Publishing: 100] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 16. | Domreis JS, Jobe BA, Aye RW, Deveney KE, Sheppard BC, Deveney CW. Management of long-term failure after colon interposition for benign disease. Am J Surg. 2002;183:544-546. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 27] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 17. | Yildirim S, Köksal H, Celayir F, Erdem L, Oner M, Baykan A. Colonic interposition vs. gastric pull-up after total esophagectomy. J Gastrointest Surg. 2004;8:675-678. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 32] [Article Influence: 1.6] [Reference Citation Analysis (0)] |